
Concept explainers
(a)
Interpretation:
The products should be identified by the reaction of anisaldehyde with
Concept Introduction:
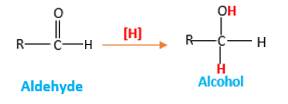
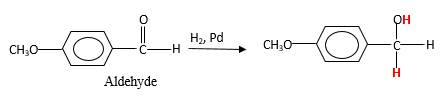
Addition of H2gas to a multiple bond is known as hydrogenation. In the presence of palladium metal as the catalyst H2 molecules react with
Answer to Problem 76P
Explanation of Solution
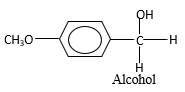
When an aldehyde reacts with H2 gas in the presence of palladium metalresulting product is an alcohol of the initial aldehyde molecule. Palladium metal act as a catalyst to the reaction that provides a surface to bind both the H2 and carbonyl compound which reduce the activation energy of the reaction.
Hydrogen atoms in the alcohol molecule shown below, which are indicated in red color are the added H during the hydrogenation reaction.
(b)
Interpretation:
The products should be identified by the reaction of anisaldehyde with
Concept Introduction:
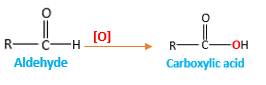
Addition of an O atom in to a molecule is known as oxidation. The hydrogen atom directly connected to the carbonyl C in an aldehyde will oxidized in the presence of an oxidizing agent such as
Oxygen atom in the carboxylic acid molecule shown below, which is indicated in red color is the added O during the oxygenation reaction.
Answer to Problem 76P
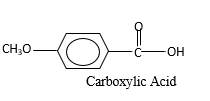
Explanation of Solution
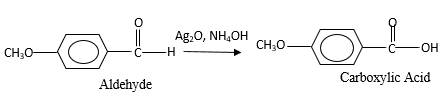
When an aldehyde reacts with
Oxygen atom in the carboxylic acid molecule shown below, which is indicated in red color is the added O during the oxygenation reaction.
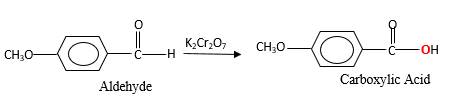
(c)
Interpretation:
The products should be identified by the reaction of anisaldehyde with
Concept Introduction:
Addition of an O atom in to a molecule is known as oxidation. The hydrogen atom directly connected to the carbonyl C in an aldehyde will oxidized in the presence of an oxidizing agent such as (
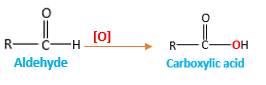
Answer to Problem 76P
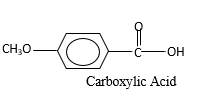
Explanation of Solution
When an aldehyde reacts with
Aldehyde's
(d)
Interpretation:
The products should be identified by the reaction of anisaldehyde with
Concept Introduction:
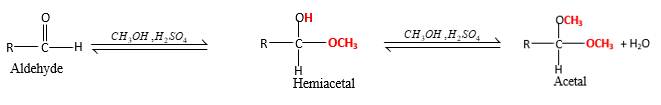
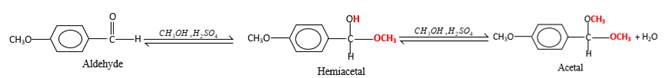
In the presence of alcohol in the acidic medium, aldehydes undergo addition reactions and give acetal in two steps.
Hydrogen atom and CH3 groups in the acetal and hemiacetal molecules shown below, which are indicated in red color are the added molecules during the reaction.
Answer to Problem 76P
Explanation of Solution
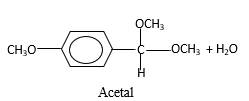
In the presence of alcohol in the acidic medium, aldehydes undergo addition reactions and give acetal in two steps. In the first step aldehydes form hemiacetals and during the second step it converts to an acetal molecule of the respective aldehyde molecule.
Addition of one molecule of alcohol in to an aldehyde forms a hemiacetal, one bond of the
(e)
Interpretation:
The products should be identified by the reaction of anisaldehyde with
Concept Introduction:
In the presence of alcohol in the acidic medium, aldehydes undergo addition reactions and give acetal in two steps.
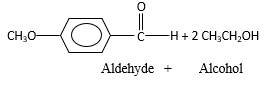
Hydrogen atom and CH2CH3 groups in the acetal and hemiacetal molecules shown below, which are indicated in red color are the added molecules during the reaction.
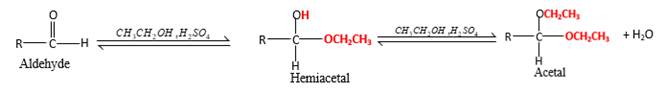
Answer to Problem 76P
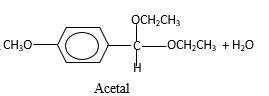
Explanation of Solution
In the presence of alcohol in the acidic medium, aldehydes undergo addition reactions and give acetal in two steps. In the first step aldehydes form hemiacetals and during the second step it converts to an acetal molecule of the respective aldehyde molecule.
Addition of one molecule of alcohol in to an aldehyde forms a hemiacetal, one bond of the
Hydrogen atom and CH2CH3 groups in the acetal and hemiacetal molecules shown below, which are indicated in red color are the added molecules during the reaction.
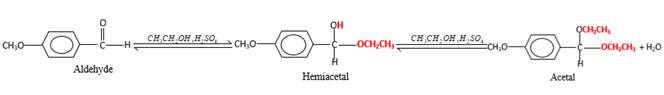
(f)
Interpretation:
The products should be identified by the reaction of
Concept Introduction:
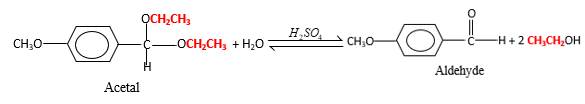
In the presence of waterandacid, acetals undergo hydrolysis reaction and produce aldehydes.
OR' groups in the acetal molecule shown below, which are indicated in red color are the molecules which becomes alcohol molecules during the hydrolysis.
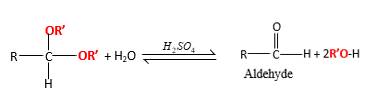
Answer to Problem 76P
Explanation of Solution
Acetals are stable molecules, but their bonds can cleave by a reaction with water and produce aldehydes.
In the acetal molecule, two bonds of the
CH2CH3 groups in the acetal molecule shown below, which are indicated in red color are the molecules which becomes alcohol molecules during the hydrolysis.
Want to see more full solutions like this?
Chapter 16 Solutions
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
- Write all of Me Possible Products For each Of the Following reactions. In each case identity all pains of enantiomers, all digsterzoners and all Meso compounds 9. 11-60 11-0-11 V-G Η Η H ~ C-11 +HB+ - 1 H b. पन्ना 171-0-11 H-C-H Н C-C=c-call +HBr Perendez ==arrow_forwardHow can i draw the mechanisms for this molecule?arrow_forwarda. Discuss and explain he difference IN Stability between the Chai and Boat Гольцу от судомехане b. For the Following Molecule draw both possible Clain conformations and explain which one is more stable and for what Reason. H. CH₂ CH₂ H "Harrow_forward
- Please provide the mechanism for this reacitonarrow_forwardQuestion 5: Name the following compound in two ways using side chain and using prefix amine (Common name and IUPAC name both) CH3NH2 CH3CH2NHCH3 CH₂CH₂N(CH3)2 Draw the structure of diethyl methyl amine Question 6. Write the balanced combustion reaction for: a. Hexane b. Propyne c. 2-pentene Question 7: Write the following electrophilic substitution reactions of benzene: Hint: Use notes if you get confused a. Halogenation reaction: b. Nitration reaction : c. Sulphonation reaction: d. Alkylation reaction: e. Aceylation reaction:arrow_forwardQuestion 4. Name the following structures ○ CH3-C-N-H H CH3CH2-C-N-H H CH3CH2-C-N-CH3 Harrow_forward

 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning


