
Concept explainers
Write structural formulas for the products formed when propanal reacts with each of the followin
(a)
(b)
(c)
(d)
(e)
(f)
(g)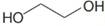 and HA
and HA
(h)
(i)
(j) Hydroxylamine
(k) Phenylhydrazine
(l) Cold dilute
(m)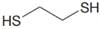 , HA
, HA
(n)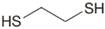 , HA, then Raney nickel
, HA, then Raney nickel
(o) m CPBA
Trending nowThis is a popular solution!

Chapter 16 Solutions
EBK ORGANIC CHEMISTRY
Additional Science Textbook Solutions
Chemistry: Structure and Properties (2nd Edition)
College Physics: A Strategic Approach (3rd Edition)
Human Biology: Concepts and Current Issues (8th Edition)
Microbiology with Diseases by Body System (5th Edition)
Biology: Life on Earth (11th Edition)
Campbell Essential Biology with Physiology (5th Edition)
- For the condensation reaction between Alamine and histamine, please help me write the amididation reaction mechanism. Then write the three letter code for the product of the reaction, then write the one letter code for the product of the reaction. arrow_forwardHow to draw the reaction mechasnism belowarrow_forwardName the following molecules with IUpacarrow_forward
- What is the molecular orbital for cyclopropenyl anion and is it aromatic, antiaromatic or nonaromatic?arrow_forwardUsing the chart describe the change from cystine to tyrosine and its impact on the protein. Using the chart describe the change from histidine to aspartic acid and its impact on the protein.arrow_forwardHow to get the predicted product of this reaction belowarrow_forward
- Please help me fill out the chart then using the chart describe the change from cystine to tyrosine and its impact on the protein. Then using the chart describe the change from histidine to aspartic acid.arrow_forwardWrite the Esterification reaction mechanism for acetic acid, and one propanol to make propanol ethanoate (molecule that gives peas its odor in flavor)arrow_forwardProvide solutionsarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

