
Concept explainers
(a)
Interpretation:
The product for the reaction of ethylbenzene with bromine gas and
Concept introduction:
An electrophilic
Answer to Problem 16.35AP
No product will be formed in the reaction of ethylbenzene with bromine gas and
Explanation of Solution
Ethylbenzene is ortho and para directing group, the electrophile will substitute on either ortho or para position of the benzene ring. In the dark no bromine free radical will be generated, therefore, no free radical reaction will occur. Carbon tetrachloride does not facilitate the production of positively charged
No reaction takes place between ethylbenzene with bromine gas and
(b)
Interpretation:
The product for the reaction ethylbenzene with
Concept introduction:
An electrophilic aromatic substitution reaction is a type of reaction in which an electrophile substitutes a hydrogen atom of the aromatic ring. Aromatic ring does not easily undergo nucleophilic substitution reaction; however, it undergoes electrophilic substitution reaction easily.
Answer to Problem 16.35AP
The products for the reaction ethylbenzene with
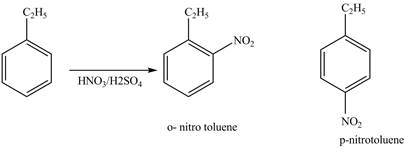
Explanation of Solution
Ethylbenzene is ortho and para directing group, the electrophile will substitute on either ortho or para position of the benzene ring. The nitronium ion
The corresponding
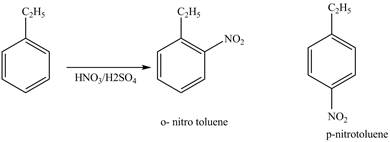
Figure 1
The products for the reaction ethylbenzene with
(c)
Interpretation:
The product for the reaction ethylbenzene with concentrated
Concept introduction:
An electrophilic aromatic substitution reaction is a type of reaction in which an electrophile substitutes a hydrogen atom of the aromatic ring. Aromatic ring does not easily undergo nucleophilic substitution reaction; however, it undergoes electrophilic substitution reaction easily.
Answer to Problem 16.35AP
The products for the reaction ethylbenzene with concentrated
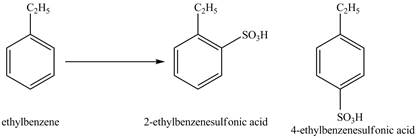
Explanation of Solution
Ethylbenzene is ortho and para directing group, the electrophile will substitute on either ortho or para position of the benzene ring. In the presence of concentrated sulphuric acid, the
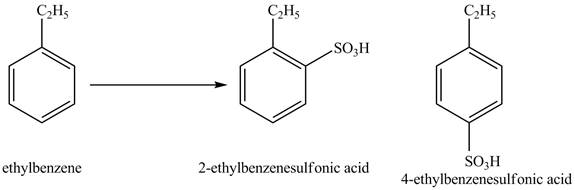
Figure 2
The products for the reaction ethylbenzene with concentrated
(d)
Interpretation:
The product for the reaction ethylbenzene with propionyl chloride and
Concept introduction:
Friedel Craft acylation is an electrophilic aromatic substitution reaction. In this reaction, the synthesis of the monoacylated product takes place from the reaction between aromatic rings and acyl chlorides.
Answer to Problem 16.35AP
The products for the reaction of ethylbenzene with propionyl chloride and
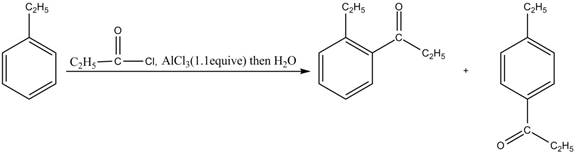
Explanation of Solution
Friedel-Crafts alkylation permits the synthesis of alkylated products by the reaction of arenes with alkyl chlorides in the presence of aluminum chloride (Lewis acid). This alkylation reaction comes under the category of electrophilic aromatic substitution. Ethylbenzene is ortho and para directing group, the electrophile will substitute on either ortho or para position of the benzene ring. The acyl group attacks either ortho or para position of ethylbenzene to form the product. The corresponding chemical reaction is shown below.
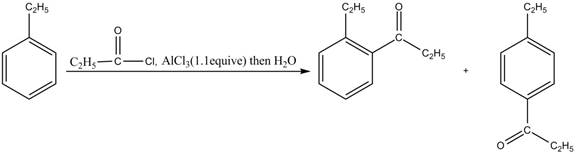
Figure 3
The products for the reaction ethylbenzene with propionyl chloride and
(e)
Interpretation:
The product for the exothermic reaction of ethylbenzene with
Concept introduction:
An electrophilic aromatic substitution reaction is a type of reaction in which an electrophile substitutes a hydrogen atom of the aromatic ring. Aromatic ring does not easily undergo nucleophilic substitution reaction; however, it undergoes electrophilic substitution reaction easily.
Answer to Problem 16.35AP
The product for the exothermic reaction of ethylbenzene with
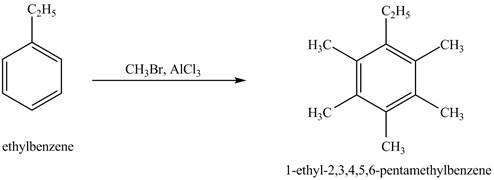
Explanation of Solution
Ethylbenzene is ortho and para directing group, the electrophile will substitute on either ortho or para position of the benzene ring. In the presence of
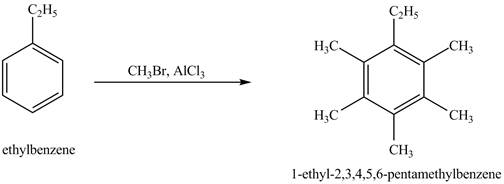
Figure 4
The product for the exothermic reaction of ethylbenzene with
(f)
Interpretation:
The product for the exothermic reaction of ethylbenzene with bromine gas and
Concept introduction:
An electrophilic aromatic substitution reaction is a type of reaction in which an electrophile substitutes a hydrogen atom of the aromatic ring. Aromatic ring does not easily undergo nucleophilic substitution reaction; however, it undergoes electrophilic substitution reaction easily.
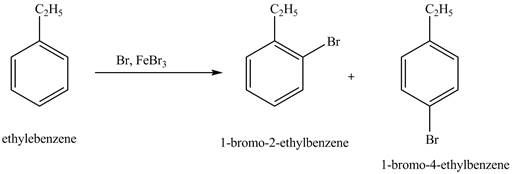
Answer to Problem 16.35AP
The products for the exothermic reaction of ethylbenzene with bromine gas and
Explanation of Solution
Ethylbenzene is ortho and para directing group, the electrophile will substitute on either ortho or para position of the benzene ring. In this reaction, positively charged
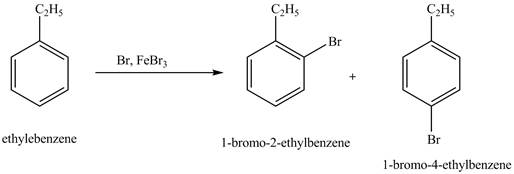
Figure 5
The products for the exothermic reaction ethylbenzene with bromine gas and
Want to see more full solutions like this?
Chapter 16 Solutions
Organic Chemistry, Ebook And Single-course Homework Access
- Draw the structure of serine at pH 6.8. Click and drag to start drawing a structure. : d كarrow_forwardTake a look at this molecule, and then answer the questions in the table below it. CH2OH H H H OH OH OH CH2OH H H H H OH H H OH H OH Is this a reducing sugar? yes α β ロ→ロ no ☑ yes Does this molecule contain a glycosidic bond? If you said this molecule does contain a glycosidic bond, write the symbol describing it. O no 0+0 If you said this molecule does contain a glycosidic bond, write the common names (including anomer and enantiomer labels) of the molecules that would be released if that bond were hydrolyzed. If there's more than one molecule, separate each name with a comma. ☐arrow_forwardAnswer the questions in the table below about this molecule: H₂N-CH₂ -C—NH–CH–C—NH–CH—COO- CH3 CH CH3 What kind of molecule is this? 0= CH2 C If you said the molecule is a peptide, write a description of it using 3-letter codes separated ☐ by dashes. polysaccharide peptide amino acid phospolipid none of the above Хarrow_forward
- Draw a Haworth projection of a common cyclic form of this monosaccharide: CH₂OH C=O HO H H -OH H OH CH₂OH Click and drag to start drawing a structure. : ☐ Х S '☐arrow_forwardNucleophilic Aromatic Substitution 22.30 Predict all possible products formed from the following nucleophilic substitution reactions. (a) (b) 9 1. NaOH 2. HCI, H₂O CI NH₁(!) +NaNH, -33°C 1. NaOH 2. HCl, H₂Oarrow_forwardSyntheses 22.35 Show how to convert toluene to these compounds. (a) -CH,Br (b) Br- -CH3 22.36 Show how to prepare each compound from 1-phenyl-1-propanone. 1-Phenyl-1-propanone ہتی. Br. (b) Br (racemic) 22.37 Show how to convert ethyl benzene to (a) 2,5-dichlorobenzoic acid and (b) 2,4-dichlorobenzoic acid. 22.38 Show reagents and conditions to bring about the following conversions. (a) 9 NH2 8 CO₂H NH2 CO₂Et (d) NO2 NH2 S NH₂ NO2 CHS CHarrow_forward
- ive the major organic product(s) of each of the following reactions or sequences of reactions. Show all rant stereochemistry. [10 only] A. B. NaN3 1. LiAlH4, ether Br 2. H₂O CH3 HNO3 H₂/Pt H₂SO ethanol C. 0 0 CH3CC1 NaOH NHCCH AICI H₂O . NH₂ CH3CH2 N CH2CH3 + HCI CH₂CH 3 1. LIAIH, THE 2. H₂Oarrow_forwardCalculate the stoichiometric amount of CaCl2 needed to convert all of the CuSO4 into CuCl2.arrow_forwardH CH تنی Cl 1. NaCN, DMF 2. LIAIH4, ether H₂O pyridine N NH₂ 5 CH H 1 HNO, H₂SO 2. Nal NH2 Br Br HNO₂ CuCl H₂SO HCI CH3 H3C NN HSO KCN CuCN 1. HNO₂, H₂SO O₂N NH2 2. OH ཀ་ལས། །ས་ཅན་ :i་དེ་མ་མ་སེ་ NH₂ CH3 1. HNO₂, H₂SO4 2. H3PO₂ 1 HNO2, H2SO4 2. Nalarrow_forward
- ive the major organic product(s) of each of the following reactions or sequences of reactions. Show all rant stereochemistry. [10 only] A. B. NaN3 1. LiAlH4, ether Br 2. H₂O CH3 HNO3 H₂/Pt H₂SO ethanol C. 0 0 CH3CC1 NaOH NHCCH AICI H₂O . NH₂ CH3CH2 N CH2CH3 + HCI CH₂CH 3 1. LIAIH, THE 2. H₂Oarrow_forwardIf a pharmacy chain sold 65 million 500-mg tablets of aspirin, how many US tons of aspirin does this represent? Report your answer to 2 significant figures.arrow_forwardHere are the options: reducing a monosaccharide a non reducing disaccharide amylopectin cellulose 1,4' beta- glycosidearrow_forward
