
Concept explainers
(a)
Interpretation:
Relationship between two given Fischer projections by means of stereoisomerism (identical or enantiomers) should be determined.
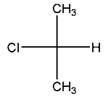 and
and 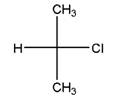
Concept Introduction:
Identical molecules are the ones with no isomers, neither constitutional isomers nor stereoisomers. Identical molecules have the same structural arrangement of atoms and the same three-dimensional arrangement.
Isomers are the molecules with the same formula but either with different structural connectivity (constitutional isomers) or different three-dimensional arrangement (stereoisomers).
A tetrahedral carbon atom bonded to four different groups is called a chiral center. A Molecule having at least one chiral center is a chiral molecule. When the mirror images of a chiral molecule are not superimposable, those mirror images become stereoisomers called enantiomers.
Fischer Projection is a method of drawing 3-D structures of organic molecules using cross formula. In this method, all non-terminal bonds are depicted as horizontal or vertical lines.
In the Fischer projection, horizontal bonds represent groups coming forward (drawn as wedges) and vertical bonds represent groups going backward (drawn as dashed wedges).
(b)
Interpretation:
Relationship between two given Fischer projections by means of stereoisomerism (identical or enantiomers) should be determined.
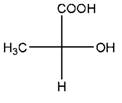 and
and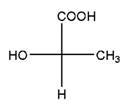
Concept Introduction:
Identical molecules are the ones with no isomers, neither constitutional isomers nor stereoisomers. Identical molecules have the same structural arrangement of atoms and the same three-dimensional arrangement.
Isomers are the molecules with the same formula but either with different structural connectivity (constitutional isomers) or different three-dimensional arrangement (stereoisomers).
A tetrahedral carbon atom bonded to four different groups is called a chiral center. A Molecule having at least one chiral center is a chiral molecule. When the mirror images of a chiral molecule are not superimposable, those mirror images become stereoisomers called enantiomers.
Fischer Projection is a method of drawing 3-D structures of organic molecules using cross formula. In this method, all non-terminal bonds are depicted as horizontal or vertical lines.
In the Fischer projection, horizontal bonds represent groups coming forward (drawn as wedges) and vertical bonds represent groups going backward (drawn as dashed wedges).
(c)
Interpretation:
Relationship between two given Fischer projections by means of stereoisomerism (identical or enantiomers) should be determined.
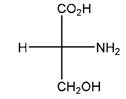 and
and 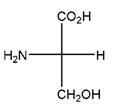
Concept Introduction:
Identical molecules are the ones with no isomers, neither constitutional isomers nor stereoisomers. Identical molecules have the same structural arrangement of atoms and the same three-dimensional arrangement.
Isomers are the molecules with the same formula but either with different structural connectivity (constitutional isomers) or different three-dimensional arrangement (stereoisomers).
A tetrahedral carbon atom bonded to four different groups is called a chiral center. A Molecule having at least one chiral center is a chiral molecule. When the mirror images of a chiral molecule are not superimposable, those mirror images become stereoisomers called enantiomers.
Fischer Projection is a method of drawing 3-D structures of organic molecules using cross formula. In this method, all non-terminal bonds are depicted as horizontal or vertical lines.
In the Fischer projection, horizontal bonds represent groups coming forward (drawn as wedges) and vertical bonds represent groups going backward (drawn as dashed wedges).
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Connect One Semester Access Card for General, Organic, & Biological Chemistry
- Can you please help me with drawing the Lewis structure of each molecular formula?I truly appreciate you!arrow_forwardPlease draw and explainarrow_forwardDescribe each highlighted bond in terms of the overlap of atomic orbitals. (a) Н Н H H [References] HIC H H C H H-C-CC-N: H σ character n character (b) HIC H H H H-C-C-C HIC H Н H O-H σ character n character Submit Answer Try Another Version 3 item attempts remainingarrow_forward
- 11 Naming and drawing alcohols Write the systematic (IUPAC) name for each of the following organic molecules: structure OH HO OH Explanation Check name ☐arrow_forwardwhat is the drawn mechanism for diethyl carbonate and 4 - bromo - N, N -dimethylaniline to create crystal violet?arrow_forwardWhich of the following compounds are constitutional isomers of each other? I and II O II and III O III and IV OI and IV O II and IV CI H CI H CI H H CI H-C-C-CI C-C-C-CI H-C-C-CI H-C-C-CI H CI Ĥ ĆI A A Ĥ ĆI || IVarrow_forward
- Please correct answer and don't used hand raitingarrow_forwardQ1: Curved Arrows, Bronsted Acids & Bases, Lewis Acids & Bases Considering the following reactions: a) Predict the products to complete the reactions. b) Use curved electron-pushing arrows to show the mechanism for the reaction in the forward direction. Redraw some of the compounds to explicitly illustrate all bonds that are broken and all bonds that are formed. c) Label Bronsted acids and bases in the left side of the reactions. Label conjugate acids and bases in the right side of the reactions. d) Label Lewis acids and bases, nucleophiles and electrophiles in the left side of the reactions. A. + OH CH30: OH B. + HBr C. H₂SO4 D. CF 3. CH 3 + HCI N H fluoxetine antidepressant 1↓ JDownloadarrow_forwardDon't used Ai solutionarrow_forward
- Part 3: AHm,system Mass of 1.00 M HCI Vol. of 1.00 M HCI Mass of NaOH(s) Total Mass in Calorimeter Mole product if HCI limiting reactant Trial 1 62.4009 1.511g Mole product if NaOH limiting reactant Limiting reactant Initial Temperature Final Temperature 23.8°C 37.6°C Change in Temperature AHm,system (calculated) Average AHm,system (calculated) (calculated) (calculated) Trial 2 64.006g 1.9599 (calculated) (calculated) (calculated) (calculated) (calculated) (calculated) 24.7°C 41.9°C (calculated) (calculated) (2 pts. each)arrow_forwardDon't used Ai solutionarrow_forwardWhat is the numerical value of the slope using the equation y=-1.823x -0.0162 please show calculationsarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning



