
EBK CHEMICAL PRINCIPLES
8th Edition
ISBN: 9781305856745
Author: DECOSTE
Publisher: CENGAGE LEARNING - CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 15, Problem 38E
Experimental data for the reaction
have been plotted in the following three different ways (with concentration units in
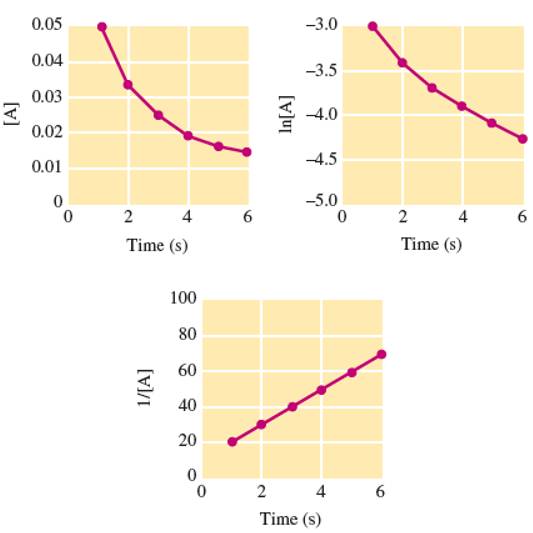
- What is the order of the reaction with respect to A, and what is the initial concentration of A? What is the concentration of A after
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
3.
a.
Use the MS to propose at least two possible molecular formulas.
For an unknown compound:
101.
27.0
29.0
41.0
50.0
52.0
55.0
57.0
100
57.5
58.0
58.5
62.0
63.0
64.0
65.0
74.0
40
75.0
76.0
20
20
40
60
80
100
120
140
160
180
200 220
m/z
99.5
68564810898409581251883040
115.0
116.0
77404799
17417M
117.0
12.9
118.0
33.5
119.0
36
133 0
1.2
157.0
2.1
159.0
16
169.0
219
170.0
17
171.0
21.6
172.0
17
181.0
1.3
183.0
197.0
100.0
198.0
200.
784
Relative Intensity
2
2
8
ō (ppm)
6
2
Solve the structure and assign each of the following spectra (IR and C-NMR)
1.
For an unknown compound with a molecular formula of C8H100:
a.
What is the DU? (show your work)
b.
Solve the structure and assign each of the following spectra.
8
6
2
ō (ppm)
4
2
0
200
150
100
50
ō (ppm)
LOD
D
4000
3000
2000
1500
1000
500
HAVENUMBERI -11
Chapter 15 Solutions
EBK CHEMICAL PRINCIPLES
Ch. 15 - Prob. 1DQCh. 15 - Prob. 2DQCh. 15 - a graph of [A] versus time for zero-, first-, and...Ch. 15 - Prob. 4DQCh. 15 - Prob. 5DQCh. 15 - Prob. 6DQCh. 15 - Prob. 7DQCh. 15 - Prob. 8DQCh. 15 - Provide a conceptual rationale for the differences...Ch. 15 - Prob. 10E
Ch. 15 - Consider the general reaction aA+bBcC and the...Ch. 15 - Prob. 12ECh. 15 - Prob. 13ECh. 15 - Prob. 14ECh. 15 - Prob. 15ECh. 15 - The hydroxyl radical (OH) is an important...Ch. 15 - Prob. 17ECh. 15 - The reaction 2NO(g)+Cl2(g)2NOCl(g) was studied at...Ch. 15 - Prob. 19ECh. 15 - The following data were obtained for the gas-phase...Ch. 15 - Prob. 21ECh. 15 - Prob. 22ECh. 15 - Prob. 23ECh. 15 - Prob. 24ECh. 15 - Prob. 25ECh. 15 - Prob. 26ECh. 15 - Prob. 27ECh. 15 - Prob. 28ECh. 15 - If the half-life for a reaction is 20. seconds,...Ch. 15 - A certain reaction has the following general form:...Ch. 15 - Prob. 31ECh. 15 - Prob. 32ECh. 15 - The decomposition of hydrogen peroxide was studied...Ch. 15 - Prob. 34ECh. 15 - Prob. 35ECh. 15 - Prob. 36ECh. 15 - At 500K in the presence of a copper surface,...Ch. 15 - Experimental data for the reaction A2B+C have been...Ch. 15 - The reaction NO(g)+O3(g)NO2(g)+O2(g) was studied...Ch. 15 - Determine the forms of the integrated and the...Ch. 15 - Prob. 41ECh. 15 - Prob. 42ECh. 15 - Prob. 43ECh. 15 - Prob. 44ECh. 15 - Prob. 45ECh. 15 - Prob. 46ECh. 15 - Prob. 47ECh. 15 - Prob. 48ECh. 15 - Prob. 49ECh. 15 - Prob. 50ECh. 15 - Prob. 51ECh. 15 - Prob. 52ECh. 15 - Prob. 53ECh. 15 - Prob. 54ECh. 15 - Prob. 55ECh. 15 - Define each of the following. elementary step...Ch. 15 - Define what is meant by unimolecular and...Ch. 15 - What two requirements must be met to call a...Ch. 15 - Prob. 59ECh. 15 - Prob. 60ECh. 15 - A proposed mechanism for a reaction is...Ch. 15 - Is the mechanism NO+Cl2k1NOCl2NOCl2+NOk22NOCl...Ch. 15 - The reaction 2NO(g)+O2(g)2NO2(g) exhibits the rate...Ch. 15 - Prob. 64ECh. 15 - The reaction...Ch. 15 - Prob. 66ECh. 15 - Prob. 67ECh. 15 - Prob. 68ECh. 15 - The following mechanism is proposed for the...Ch. 15 - The following mechanism has been proposed to...Ch. 15 - Consider the hypothetical reaction BE+F which is...Ch. 15 - How is the rate of a reaction affected by each of...Ch. 15 - The central idea of the collision model is that...Ch. 15 - Prob. 74ECh. 15 - Prob. 75ECh. 15 - Consider the following potential energy plots Rank...Ch. 15 - Prob. 77ECh. 15 - Prob. 78ECh. 15 - Prob. 79ECh. 15 - Prob. 80ECh. 15 - Prob. 81ECh. 15 - Chemists commonly use a rule of thumb that an...Ch. 15 - Prob. 83ECh. 15 - Prob. 84ECh. 15 - Prob. 85ECh. 15 - Prob. 86ECh. 15 - For the following reaction profiles, indicate the...Ch. 15 - Prob. 88ECh. 15 - Prob. 89ECh. 15 - Prob. 90ECh. 15 - Prob. 91ECh. 15 - Prob. 92ECh. 15 - The decomposition of NH3 to N2 and H2 was studied...Ch. 15 - One pathway for the destruction of ozone in the...Ch. 15 - Prob. 95ECh. 15 - Prob. 96ECh. 15 - Prob. 97ECh. 15 - Prob. 98ECh. 15 - Prob. 99ECh. 15 - Prob. 100AECh. 15 - Prob. 101AECh. 15 - Prob. 102AECh. 15 - Prob. 103AECh. 15 - Prob. 104AECh. 15 - Prob. 105AECh. 15 - Prob. 106AECh. 15 - Prob. 107AECh. 15 - Prob. 108AECh. 15 - Prob. 109AECh. 15 - The decomposition of NO2(g) occurs by the...Ch. 15 - Prob. 111AECh. 15 - Prob. 112AECh. 15 - Prob. 113AECh. 15 - Prob. 114AECh. 15 - Prob. 115AECh. 15 - Prob. 116AECh. 15 - The compound NO2Cl is thought to decompose to NO2...Ch. 15 - Prob. 118AECh. 15 - Prob. 119AECh. 15 - Prob. 120AECh. 15 - Prob. 121AECh. 15 - Prob. 122AECh. 15 - Prob. 123AECh. 15 - Prob. 124AECh. 15 - Prob. 125AECh. 15 - Prob. 126AECh. 15 - Consider the following reaction: CH3X+YCH3Y+X At...Ch. 15 - The following data were collected in two studies...Ch. 15 - Prob. 129CPCh. 15 - For the reaction 2A+Bproducts afriend proposes the...Ch. 15 - Consider the hypothetical reaction A+B+2C2D+3E In...Ch. 15 - A reaction represented by the equation...Ch. 15 - Prob. 133CPCh. 15 - You are studying the kinetics of the reaction...Ch. 15 - Prob. 135CPCh. 15 - Prob. 136MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 16. The proton NMR spectral information shown in this problem is for a compound with formula CioH,N. Expansions are shown for the region from 8.7 to 7.0 ppm. The normal carbon-13 spec- tral results, including DEPT-135 and DEPT-90 results, are tabulated: 7 J Normal Carbon DEPT-135 DEPT-90 19 ppm Positive No peak 122 Positive Positive cus и 124 Positive Positive 126 Positive Positive 128 No peak No peak 4° 129 Positive Positive 130 Positive Positive (144 No peak No peak 148 No peak No peak 150 Positive Positive してしarrow_forward3. Propose a synthesis for the following transformation. Do not draw an arrow-pushing mechanism below, but make sure to draw the product of each proposed step (3 points). + En CN CNarrow_forwardShow work..don't give Ai generated solution...arrow_forward
- Label the spectrum with spectroscopyarrow_forwardQ1: Draw the most stable and the least stable Newman projections about the C2-C3 bond for each of the following isomers (A-C). Are the barriers to rotation identical for enantiomers A and B? How about the diastereomers (A versus C or B versus C)? enantiomers H Br H Br (S) CH3 H3C (S) (R) CH3 H3C H Br A Br H C H Br H3C (R) B (R)CH3 H Br H Br H3C (R) (S) CH3 Br H D identicalarrow_forwardLabel the spectrumarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Kinetics: Initial Rates and Integrated Rate Laws; Author: Professor Dave Explains;https://www.youtube.com/watch?v=wYqQCojggyM;License: Standard YouTube License, CC-BY