
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 15, Problem 19PP
PRACTICE PROBLEM 15.19
Birch reduction of toluene leads to a product with the molecular formula
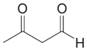 and
and 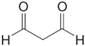 . What is the structure of the Birch reduction product.
. What is the structure of the Birch reduction product.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Using the following two half-reactions, determine the pH range in which $NO_2^-\ (aq)$ cannot be found as the predominant chemical species in water.* $NO_3^-(aq)+10H^+(aq)+8e^-\rightarrow NH_4^+(aq)+3H_2O(l),\ pE^{\circ}=14.88$* $NO_2^-(aq)+8H^+(aq)+6e^-\rightarrow NH_4^+(aq)+2H_2O(l),\ pE^{\circ}=15.08$
Indicate characteristics of oxodec acid.
What is the final product when hexanedioic acid reacts with 1º PCl5 and 2º NH3.
Chapter 15 Solutions
Organic Chemistry
Ch. 15 - PRACTICE PROBLEM 15.1
Show how loss of a proton...Ch. 15 - Prob. 2PPCh. 15 - PRACTICE PROBLEM 15.3
Outline all steps in a...Ch. 15 - PRACTICE PROBLEM 15.4 Provide a mechanism that...Ch. 15 - Prob. 5PPCh. 15 - Prob. 6PPCh. 15 - Prob. 7PPCh. 15 - PRACTICE PROBLEM 15.8 Write resonance structures...Ch. 15 - PRACTICE PROBLEM 15.9
Provide a mechanism for the...Ch. 15 - PRACTICE PROBLEM 15.10 The trifluoromethyl group...
Ch. 15 - PRACTICE PROBLEM 15.11
Predict the major products...Ch. 15 - PRACTICE PROBLEM 15.12 Predict the major product...Ch. 15 - PRACTICE PROBLEM 15.13
Write mechanisms for the...Ch. 15 - Prob. 14PPCh. 15 - PRACTICE PROBLEM 15.15
Suppose you needed to...Ch. 15 - PRACTICE PROBLEM 15.16 1-Fluoro-2,4-dinitrobenzene...Ch. 15 - Prob. 17PPCh. 15 - PRACTICE PROBLEM 15.18
When...Ch. 15 - PRACTICE PROBLEM 15.19 Birch reduction of toluene...Ch. 15 - Prob. 20PCh. 15 - Prob. 21PCh. 15 - What monobromination product (or products) would...Ch. 15 - 15.23 Predict the major products of the following...Ch. 15 - Prob. 24PCh. 15 - 15.25 Starting with styrene, outline a synthesis...Ch. 15 - Prob. 26PCh. 15 - 15.27 Starting with aniline, outline a synthesis...Ch. 15 - Prob. 28PCh. 15 - Propose structures for compounds GI:Ch. 15 - 2,6-Dichlorophenol has been isolated from the...Ch. 15 - Prob. 31PCh. 15 - 15.32 Give structures (including stereochemistry...Ch. 15 - Provide a detailed mechanism for each of the...Ch. 15 - 15.34 Provide a detailed mechanism for the...Ch. 15 - Prob. 35PCh. 15 - Many polycyclic aromatic compounds have been...Ch. 15 - Prob. 37PCh. 15 - Prob. 38PCh. 15 - Prob. 39PCh. 15 - Prob. 40PCh. 15 - Predict the product of the following reaction.Ch. 15 - 15.42 When m-chlorotoluene is treated with sodium...Ch. 15 - Prob. 43PCh. 15 - Prob. 44PCh. 15 - Prob. 45PCh. 15 - Prob. 46PCh. 15 - 15.47 Provide structures for compounds A and B:
Ch. 15 - Prob. 48PCh. 15 - 15.49 Treating cyclohexene with acetyl chloride...Ch. 15 - 15.50 The tert-butyl group can be used as a...Ch. 15 - 15.51 When toluene is sulfonated (concentrated )...Ch. 15 - Prob. 52PCh. 15 - 2-Methylnaphthalene can be synthesized from...Ch. 15 - Prob. 54PCh. 15 - Prob. 55PCh. 15 - Prob. 56PCh. 15 - Prob. 57PCh. 15 - Prob. 58PCh. 15 - Furan undergoes electrophilic aromatic...Ch. 15 - A C-D bond is harder to break than a C-H bond,...Ch. 15 - 15.61 Acetanilide was subjected to the following...Ch. 15 - Prob. 62PCh. 15 - Prob. 63PCh. 15 - Prob. 64PCh. 15 - When compound C, which is often used to model a...Ch. 15 - Open the molecular model file for benzyne and...Ch. 15 - The structure of thyroxine, a thyroid hormone that...Ch. 15 - Prob. 2LGPCh. 15 - 3. Deduce the structures of compounds E–L in the...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Why does a one-step growth curve differ in shape from that of a bacterial growth curve?
Brock Biology of Microorganisms (15th Edition)
APPLY 1.2 Express the following quantities in scientific notation
using fundamental SI units of mass and lengt...
Chemistry (7th Edition)
11.57 Draw the cis and trans isomers for each of the following: (11.6)
a. 2-pentene
b. 3-hexene
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
PRACTICE PROBLEM 18.10 How would you use the acetoacetic ester synthesis to prepare the following?
Organic Chemistry
41. A reaction in which A, B, and C react to form products is first order in A, second order in B, and zero ord...
Chemistry: Structure and Properties (2nd Edition)
Which type of cartilage is most plentiful in the adult body?
Anatomy & Physiology (6th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the final product when D-galactose reacts with hydroxylamine?arrow_forwardIndicate the formula of the product obtained by reacting methyl 5-chloro-5-oxopentanoate with 1 mole of 4-penten-1-ylmagnesium bromide.arrow_forwardIn the two chair conformations of glucose, the most stable is the one with all the OH groups in the equatorial position. Is this correct?arrow_forward
- please help me with my homeworkarrow_forwardhelparrow_forwardThe temperature on a sample of pure X held at 1.25 atm and -54. °C is increased until the sample boils. The temperature is then held constant and the pressure is decreased by 0.42 atm. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 2 0 0 200 400 temperature (K) Xarrow_forward
- QUESTION: Answer Question 5: 'Calculating standard error of regression' STEP 1 by filling in all the empty green boxes *The values are all provided in the photo attached*arrow_forwardpressure (atm) 3 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 0 0 200 temperature (K) 400 аarrow_forwarder your payment details | bar xb Home | bartleby x + aleksogi/x/isl.exe/1o u-lgNskr7j8P3jH-1Qs_pBanHhviTCeeBZbufuBYT0Hz7m7D3ZcW81NC1d8Kzb4srFik1OUFhKMUXzhGpw7k1 O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 1 3- 0- 0 200 Explanation Check temperature (K) 400 X Q Search L G 2025 McGraw Hill LLC. All Rights Reserved Terms of Use Privacy Cearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY