
(a)
Interpretation: For the given set of compounds the location of the partial charges that results due to inductive effect should be identified.
Concept Introduction: The nature of the bond depends on the electronegativity values of the shared electron pair of the involved atoms.
Electronegativity is the important chemical property of the elements in the periodic table. It is the tendency of the atoms to attract electrons towards it.
If the difference in electronegativity is between 0.5 and 1.7, a bond between two different electronegative atoms becomes polar. Most electronegative atoms get partial negative charge because they attract electrons from least electronegative atom towards it. The least electronegative atoms get partial positive charge because it loses electrons towards most electronegative atoms.
The process of the attraction of electrons from electron-donating atoms (less electronegative atoms) towards electron-withdrawing atoms (most electronegative atoms) is called induction. It can be represented by the following arrow:
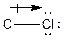
Here, chlorine atom has more electronegative than carbon atom. Chlorine attracts electrons towards it. This difference in electron density is called inductive effect. It can be shown by Greek symbol delta (
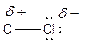
To find: Categorize all polar covalent bonds in the given compound (a)
(b)
Interpretation: For the given set of compounds the location of the partial charges that results due to inductive effect should be identified.
Concept Introduction: The nature of the bond depends on the electronegativity values of the shared electron pair of the involved atoms.
Electronegativity is the important chemical property of the elements in the periodic table. It is the tendency of the atoms to attract electrons towards it.
If the difference in electronegativity is between 0.5 and 1.7, a bond between two different electronegative atoms becomes polar. Most electronegative atoms get partial negative charge because they attract electrons from least electronegative atom towards it. The least electronegative atoms get partial positive charge because it loses electrons towards most electronegative atoms.
The process of the attraction of electrons from electron-donating atoms (less electronegative atoms) towards electron-withdrawing atoms (most electronegative atoms) is called induction. It can be represented by the following arrow:
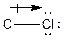
Here, chlorine atom has more electronegative than carbon atom. Chlorine attracts electrons towards it. This difference in electron density is called inductive effect. It can be shown by Greek symbol delta (
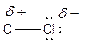
To find: Categorize all polar covalent bonds in the given compound (b)
(c)
Interpretation: For the given set of compounds the location of the partial charges that results due to inductive effect should be identified.
Concept Introduction: The nature of the bond depends on the electronegativity values of the shared electron pair of the involved atoms.
Electronegativity is the important chemical property of the elements in the periodic table. It is the tendency of the atoms to attract electrons towards it.
If the difference in electronegativity is between 0.5 and 1.7, a bond between two different electronegative atoms becomes polar. Most electronegative atoms get partial negative charge because they attract electrons from least electronegative atom towards it. The least electronegative atoms get partial positive charge because it loses electrons towards most electronegative atoms.
The process of the attraction of electrons from electron-donating atoms (less electronegative atoms) towards electron-withdrawing atoms (most electronegative atoms) is called induction. It can be represented by the following arrow:
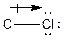
Here, chlorine atom has more electronegative than carbon atom. Chlorine attracts electrons towards it. This difference in electron density is called inductive effect. It can be shown by Greek symbol delta (
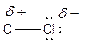
To find: Categorize all polar covalent bonds in the given compound (c)
(d)
Interpretation: For the given set of compounds the location of the partial charges that results due to inductive effect should be identified.
Concept Introduction: The nature of the bond depends on the electronegativity values of the shared electron pair of the involved atoms.
Electronegativity is the important chemical property of the elements in the periodic table. It is the tendency of the atoms to attract electrons towards it.
If the difference in electronegativity is between 0.5 and 1.7, a bond between two different electronegative atoms becomes polar. Most electronegative atoms get partial negative charge because they attract electrons from least electronegative atom towards it. The least electronegative atoms get partial positive charge because it loses electrons towards most electronegative atoms.
The process of the attraction of electrons from electron-donating atoms (less electronegative atoms) towards electron-withdrawing atoms (most electronegative atoms) is called induction. It can be represented by the following arrow:
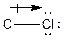
Here, chlorine atom has more electronegative than carbon atom. Chlorine attracts electrons towards it. This difference in electron density is called inductive effect. It can be shown by Greek symbol delta (
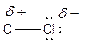
To find: Categorize all polar covalent bonds in the given compound (d)
(e)
Interpretation: For the given set of compounds the location of the partial charges that results due to inductive effect should be identified.
Concept Introduction: The nature of the bond depends on the electronegativity values of the shared electron pair of the involved atoms.
Electronegativity is the important chemical property of the elements in the periodic table. It is the tendency of the atoms to attract electrons towards it.
If the difference in electronegativity is between 0.5 and 1.7, a bond between two different electronegative atoms becomes polar. Most electronegative atoms get partial negative charge because they attract electrons from least electronegative atom towards it. The least electronegative atoms get partial positive charge because it loses electrons towards most electronegative atoms.
The process of the attraction of electrons from electron-donating atoms (less electronegative atoms) towards electron-withdrawing atoms (most electronegative atoms) is called induction. It can be represented by the following arrow:
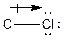
Here, chlorine atom has more electronegative than carbon atom. Chlorine attracts electrons towards it. This difference in electron density is called inductive effect. It can be shown by Greek symbol delta (
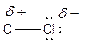
To find: Categorize all polar covalent bonds in the given compound (e)
(f)
Interpretation: For the given set of compounds the location of the partial charges that results due to inductive effect should be identified.
Concept Introduction: The nature of the bond depends on the electronegativity values of the shared electron pair of the involved atoms.
Electronegativity is the important chemical property of the elements in the periodic table. It is the tendency of the atoms to attract electrons towards it.
If the difference in electronegativity is between 0.5 and 1.7, a bond between two different electronegative atoms becomes polar. Most electronegative atoms get partial negative charge because they attract electrons from least electronegative atom towards it. The least electronegative atoms get partial positive charge because it loses electrons towards most electronegative atoms.
The process of the attraction of electrons from electron-donating atoms (less electronegative atoms) towards electron-withdrawing atoms (most electronegative atoms) is called induction. It can be represented by the following arrow:
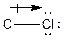
Here, chlorine atom has more electronegative than carbon atom. Chlorine attracts electrons towards it. This difference in electron density is called inductive effect. It can be shown by Greek symbol delta (
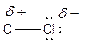
To find: Categorize all polar covalent bonds in the given compound (f)
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Student Study Guide and Solutions Manual T/A Organic Chemistry
- Which of the following is true for a particular reaction if ∆G° is -40.0 kJ/mol at 290 K and –20.0 kJ/mol at 390 K?arrow_forwardWhat is the major product of the following reaction? O O OH OH 1. BH 2. H₂O₂, NaOH OH OHarrow_forwardDraw the products formed when each ester is hydrolyzed with water and sulfuric acid.arrow_forward
- Draw the products of the hydrolysis reaction between the ester molecule and water. Determine the products of the following reaction.arrow_forwardWhat is the unsaturation number for compounds with the formula C₂H₁₂Cl₂? O õ õ o o 4 3arrow_forwardIndicate the product obtained (formula). F3C. CF3 Br NH2 NH OMe K2CO3, DABCO, DMFarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





