
CHEMISTRY-MASTERINGCHEMISTRY W/ETEXT
8th Edition
ISBN: 9780135204634
Author: Robinson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 13, Problem 13.16A
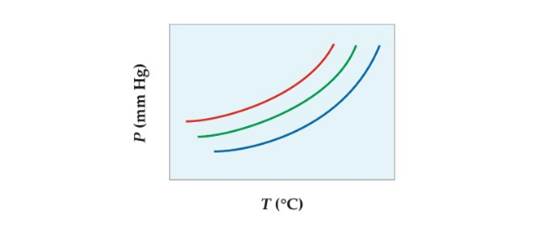
Conceptual APPLY 12.18 The following diagram shows a close-up view of part of the vapor-pressure curves for two pure liquids and a mixture of the two. Which curves represent pure liquids, and which represents the mixture?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Using reaction free energy to predict equilibrium composition
Consider the following equilibrium:
2NOCI (g) 2NO (g) + Cl2 (g) AGº =41. kJ
Now suppose a reaction vessel is filled with 4.50 atm of nitrosyl chloride (NOCI) and 6.38 atm of chlorine (C12) at 212. °C. Answer the following questions
about this system:
?
rise
Under these conditions, will the pressure of NOCI tend to rise or fall?
x10
fall
Is it possible to reverse this tendency by adding NO?
In other words, if you said the pressure of NOCI will tend to rise, can that
be changed to a tendency to fall by adding NO? Similarly, if you said the
pressure of NOCI will tend to fall, can that be changed to a tendency to
rise by adding NO?
yes
no
If you said the tendency can be reversed in the second question, calculate
the minimum pressure of NO needed to reverse it.
Round your answer to 2 significant digits.
0.035 atm
✓
G
00.
18
Ar
Highlight each glycosidic bond in the molecule below. Then answer the questions in the table under the drawing area.
HO-
HO-
-0
OH
OH
HO
NG
HO-
HO-
OH
OH
OH
OH
NG
OH
€
+
Suppose the molecule in the drawing area below were reacted with H₂ over a platinum catalyst. Edit the molecule to show what would happen to it. That is, turn
it into the product of the reaction.
Also, write the name of the product molecule under the drawing area.
Name: ☐
H
C=0
X
H-
OH
HO-
H
HO-
-H
CH₂OH
×
Chapter 13 Solutions
CHEMISTRY-MASTERINGCHEMISTRY W/ETEXT
Ch. 13 - Prob. 13.1PCh. 13 - Prob. 13.2ACh. 13 - Prob. 13.3PCh. 13 - Prob. 13.4ACh. 13 - PRACTICE 12.5 A 50.0 mL sample of drinking water...Ch. 13 - APPLY 12.6 The legal limit for human exposure to...Ch. 13 - PRACTICE 12.7 What mass in grams of a 0.500 m...Ch. 13 - APPLY 12.8 What is the molality of a solution...Ch. 13 - PRACTICE 12.9 The density at 20°C of a 0.500 M...Ch. 13 - APPLY 12.10 The density at 20°C of a 0.258 m...
Ch. 13 - ThesolubilityofCO2inwateris 3.2102 M at 25 °C...Ch. 13 - APPLY 12.12 Use the Henry’s law constant you...Ch. 13 - PRACTICE 12.13 What is the vapor pressure in mm Hg...Ch. 13 - APPLY 12.14 A solution made by dissolving 8.110 g...Ch. 13 - PRACTICE 12.17 What is the vapor pressure of the...Ch. 13 - Conceptual APPLY 12.18 The following diagram shows...Ch. 13 - What is the normal boiling point in °C of an...Ch. 13 - APPLY 12.20 The following phase diagram shows a...Ch. 13 - Prob. 13.19PCh. 13 - APPLY 12.22 Cells in the human eye have an osmotic...Ch. 13 - PRACTICE 12.23 A solution prepared by dissolving...Ch. 13 - Prob. 13.22ACh. 13 - PROBLEM 12.25 What is the difference between a...Ch. 13 - PROBLEM 12.26 Urea has a high solubility in blood...Ch. 13 - Use Table 13.5 to calculate the osmotic pressure...Ch. 13 - Prob. 13.26PCh. 13 - Many people take vitamin supplements to promote...Ch. 13 - Prob. 13.28CPCh. 13 - Prob. 13.29CPCh. 13 - Prob. 13.30CPCh. 13 - Prob. 13.31CPCh. 13 - Prob. 13.32CPCh. 13 - Prob. 13.33CPCh. 13 - Prob. 13.34CPCh. 13 - Prob. 13.35CPCh. 13 - Prob. 13.36SPCh. 13 - 12.41 Why do ionic substances with higher lattice...Ch. 13 - Prob. 13.38SPCh. 13 - Prob. 13.39SPCh. 13 - Classify the strongest type of intermolecular...Ch. 13 - Classify the strongest type of intermolecular...Ch. 13 - Prob. 13.42SPCh. 13 - Prob. 13.43SPCh. 13 - Br2 is much more soluble in tetrachloromethane,...Ch. 13 - Predict whether the solubility of formaldehyde,...Ch. 13 - Prob. 13.46SPCh. 13 - Prob. 13.47SPCh. 13 - Arrange the following compounds in order of their...Ch. 13 - Prob. 13.49SPCh. 13 - Prob. 13.50SPCh. 13 - Prob. 13.51SPCh. 13 - Prob. 13.52SPCh. 13 - Prob. 13.53SPCh. 13 - The dissolution of CaCl2(s) in water is...Ch. 13 - The dissolution of NH4ClO4(s) in water is...Ch. 13 - Assuming that seawater is an aqueous solution of...Ch. 13 - Prob. 13.57SPCh. 13 - Propranolol°C16H21NO2) a so-called beta-blocker...Ch. 13 - Prob. 13.59SPCh. 13 - How would you prepare each of the following...Ch. 13 - Prob. 13.61SPCh. 13 - Prob. 13.62SPCh. 13 - Prob. 13.63SPCh. 13 - Prob. 13.64SPCh. 13 - Which of the following solutions has the higher...Ch. 13 - What is the mass percent concentration of the...Ch. 13 - Prob. 13.67SPCh. 13 - Prob. 13.68SPCh. 13 - Prob. 13.69SPCh. 13 - What is the concentration of each of the following...Ch. 13 - Prob. 13.71SPCh. 13 - The density of a 16.0 mass % solution of sulfuric...Ch. 13 - Prob. 13.73SPCh. 13 - What is the molality of the 40.0 mass % ethylene...Ch. 13 - Prob. 13.75SPCh. 13 - Prob. 13.76SPCh. 13 - Prob. 13.77SPCh. 13 - Prob. 13.78SPCh. 13 - Prob. 13.79SPCh. 13 - Look at the solubility graph in Figure 13.7, and...Ch. 13 - Prob. 13.81SPCh. 13 - Prob. 13.82SPCh. 13 - Prob. 13.83SPCh. 13 - Fish generally need an O2 concentration in water...Ch. 13 - At an altitude of 10, 000 ft, the partial pressure...Ch. 13 - Prob. 13.86SPCh. 13 - Prob. 13.87SPCh. 13 - Prob. 13.88SPCh. 13 - When solid CaCl2 is added to liquid water, the...Ch. 13 - Rank the following aqueous solutions from lowest...Ch. 13 - Which of the following aqueous solutions has the...Ch. 13 - What is the vapor pressure in mm Hg of a solution...Ch. 13 - What is the normal boiling point in oC of a...Ch. 13 - What is the freezing point in °C of a solution...Ch. 13 - Assuming complete dissociation, what is the...Ch. 13 - When 9.12 g of HCI was dissolved in 190 g of...Ch. 13 - Prob. 13.97SPCh. 13 - When 1 mol of NaCI is added to 1 L of water, the...Ch. 13 - Prob. 13.99SPCh. 13 - Draw a phase diagram showing how the phase...Ch. 13 - Prob. 13.101SPCh. 13 - What is the vapor pressure in mm Hg of the...Ch. 13 - What is the vapor pressure in mm Hg of a solution...Ch. 13 - What is the boiling point in oC of each of the...Ch. 13 - What is the freezing point in oC of each of the...Ch. 13 - Prob. 13.106SPCh. 13 - The van’t Hoff factor for KCl is i=1.85. What is...Ch. 13 - Hepatane (C7H16) and octane (C8H18) are...Ch. 13 - Prob. 13.109SPCh. 13 - Acetone, C3H6O , and ethyl acetate, C4H8O2, are...Ch. 13 - Prob. 13.111SPCh. 13 - Prob. 13.112SPCh. 13 - What is the mole fraction of each component in the...Ch. 13 - Prob. 13.114SPCh. 13 - A solution prepared by dissolving 3.00 g of...Ch. 13 - Prob. 13.116SPCh. 13 - Prob. 13.117SPCh. 13 - Prob. 13.118SPCh. 13 - What osmotic presure in mm Hg would you expect for...Ch. 13 - Prob. 13.120SPCh. 13 - Prob. 13.121SPCh. 13 - Prob. 13.122SPCh. 13 - If cost per gram were not a concern, which of the...Ch. 13 - Prob. 13.124SPCh. 13 - Met-enkephalin is one of the so-called endorphins,...Ch. 13 - Prob. 13.126SPCh. 13 - Prob. 13.127SPCh. 13 - Prob. 13.128SPCh. 13 - Prob. 13.129SPCh. 13 - How many grams of naphthalene, C10H8 (commonly...Ch. 13 - Prob. 13.131SPCh. 13 - Assuming that seawater is a 3.5 mass % solution of...Ch. 13 - There’s actually much more in seawater than just...Ch. 13 - Prob. 13.134SPCh. 13 - What is the van’t Hoff factor for K2SO4 in an...Ch. 13 - If the van’t Hoff factor for Lid in a 0.62 m...Ch. 13 - What is the value of the van’t Hoff factor for KCI...Ch. 13 - A solid mixture of KCI, KNO3, and Ba(N03)2 is...Ch. 13 - Prob. 13.139SPCh. 13 - An aqueous solution of a certain organic compound...Ch. 13 - Prob. 13.141SPCh. 13 - Prob. 13.142SPCh. 13 - A solution of 0.250 g of naphthalene (mothballs)...Ch. 13 - Prob. 13.144SPCh. 13 - Prob. 13.145SPCh. 13 - The steroid hormone estradiol contains only C, H,...Ch. 13 - Many acids are partially dissociated into ions in...Ch. 13 - Addition of 50.00 mL of 2.238 H2SO4 (solution...Ch. 13 - Prob. 13.149MPCh. 13 - Prob. 13.150MPCh. 13 - Combustion analysis of a 36.72-mg sample of the...Ch. 13 - Prob. 13.152MPCh. 13 - Prob. 13.153MPCh. 13 - Prob. 13.154MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the Haworth projection of the disaccharide made by joining D-glucose and D-mannose with a ẞ(1-4) glycosidic bond. If the disaccharide has more than one anomer, you can draw any of them. Click and drag to start drawing a structure. Xarrow_forwardEpoxides can be opened in aqueous acid or aqueous base to produce diols (molecules with two OH groups). In this question, you'll explore the mechanism of epoxide opening in aqueous acid. 2nd attempt Be sure to show all four bonds at stereocenters using hash and wedge lines. 0 0 Draw curved arrows to show how the epoxide reacts with hydronium ion. 100 +1: 1st attempt Feedback Be sure to show all four bonds at stereocenters using hash and wedge lines. See Periodic Table See Hint H A 5 F F Hr See Periodic Table See Hintarrow_forward03 Question (1 point) For the reaction below, draw both of the major organic products. Be sure to consider stereochemistry. > 1. CH₂CH₂MgBr 2. H₂O 3rd attempt Draw all four bonds at chiral centers. Draw all stereoisomers formed. Draw the structures here. e 130 AN H See Periodic Table See Hint P C Brarrow_forward
- You may wish to address the following issues in your response if they are pertinent to the reaction(s) you propose to employ:1) Chemoselectivity (why this functional group and not another?) 2) Regioselectivity (why here and not there?) 3) Stereoselectivity (why this stereoisomer?) 4) Changes in oxidation state. Please make it in detail and draw it out too in what step what happens. Thank you for helping me!arrow_forward1) Chemoselectivity (why this functional group and not another?) 2) Regioselectivity (why here and not there?) 3) Stereoselectivity (why this stereoisomer?) 4) Changes in oxidation state. Everything in detail and draw out and write it.arrow_forwardCalculating the pH at equivalence of a titration 3/5 Izabella A chemist titrates 120.0 mL of a 0.7191M dimethylamine ((CH3)2NH) solution with 0.5501 M HBr solution at 25 °C. Calculate the pH at equivalence. The pk of dimethylamine is 3.27. Round your answer to 2 decimal places. Note for advanced students: you may assume the total volume of the solution equals the initial volume plus the volume of HBr solution added. pH = ☐ ✓ 18 Ar Boarrow_forward
- Alcohols can be synthesized using an acid-catalyzed hydration of an alkene. An alkene is combined with aqueous acid (e.. sulfuric acid in water). The reaction mechanism typically involves a carbocation intermediate. > 3rd attempt 3343 10 8 Draw arrows to show the reaction between the alkene and hydronium ion. that 2nd attempt Feedback 1st attempt تعمال Ju See Periodic Table See Hint F D Ju See Periodic Table See Hintarrow_forwardDraw the simplified curved arrow mechanism for the reaction of acetone and CHgLi to give the major product. 4th attempt Π Draw the simplified curved arrow mechanism T 3rd attempt Feedback Ju See Periodic Table See Hint H -H H -I H F See Periodic Table See Hintarrow_forwardSelect the correct reagent to accomplish the first step of this reaction. Then draw a mechanism on the Grignard reagent using curved arrow notation to show how it is converted to the final product. 4th attempt Part 1 (0.5 point) Select the correct reagent to accomplish the first step of this reaction. Choose one: OA Mg in ethanol (EtOH) OB. 2 Li in THF O C. Li in THF D. Mg in THF O E Mg in H2O Part 2 (0.5 point) Br Part 1 Bri Mg CH B CH, 1 Draw intermediate here, but no arrows. © TE See Periodic Table See Hint See Hint ין Harrow_forward
- Select the product for the following reaction. HO HO PCC OH ○ OH O HO ○ HO HO HOarrow_forward5:45 Х Select the final product for the following reaction sequence. O O 1. Mg. ether 2.D.Oarrow_forwardBased on the chart Two similarities between the molecule with alpha glycosidic linkages. Two similarities between the molecules with beta glycosidtic linkages. Two differences between the alpha and beta glycosidic linkages.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
Intermolecular Forces and Boiling Points; Author: Professor Dave Explains;https://www.youtube.com/watch?v=08kGgrqaZXA;License: Standard YouTube License, CC-BY