
Concept explainers
Stearidonic acid (C18H28O2) is an unsaturated fatty acid obtained from oils isolated from hemp and blackcurrant
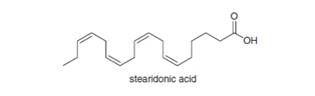
a. What fatty acid is formed when stearidonic acid is hydrogenated with excess
b. What fatty acids are formed when stearidonic acid is hydrogenated with one equivalent of
c. Draw the structure of a possible product formed when stearidonic acid is hydrogenated with one equivalent of
d. How do the melting points of the following fatty acids compare: stearidonic acid; one of the products formed in part (b); the product drawn in part (c)?
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
Additional Science Textbook Solutions
Applications and Investigations in Earth Science (9th Edition)
Chemistry: Structure and Properties (2nd Edition)
Organic Chemistry (8th Edition)
Human Anatomy & Physiology (2nd Edition)
Fundamentals Of Thermodynamics
- Q1: Curved Arrows, Bronsted Acids & Bases, Lewis Acids & Bases Considering the following reactions: a) Predict the products to complete the reactions. b) Use curved electron-pushing arrows to show the mechanism for the reaction in the forward direction. Redraw some of the compounds to explicitly illustrate all bonds that are broken and all bonds that are formed. c) Label Bronsted acids and bases in the left side of the reactions. Label conjugate acids and bases in the right side of the reactions. d) Label Lewis acids and bases, nucleophiles and electrophiles in the left side of the reactions. A. + OH CH30: OH B. + HBr C. H₂SO4 D. CF 3. CH 3 + HCI N H fluoxetine antidepressant 1↓ JDownloadarrow_forwardDon't used Ai solutionarrow_forwardPart 3: AHm,system Mass of 1.00 M HCI Vol. of 1.00 M HCI Mass of NaOH(s) Total Mass in Calorimeter Mole product if HCI limiting reactant Trial 1 62.4009 1.511g Mole product if NaOH limiting reactant Limiting reactant Initial Temperature Final Temperature 23.8°C 37.6°C Change in Temperature AHm,system (calculated) Average AHm,system (calculated) (calculated) (calculated) Trial 2 64.006g 1.9599 (calculated) (calculated) (calculated) (calculated) (calculated) (calculated) 24.7°C 41.9°C (calculated) (calculated) (2 pts. each)arrow_forward
- 1.) Using the graph below (including the line equation of y = -1.823x - 0.0162) What is the numerical value for the slope shown? 2.) What are the Unit(s) associated with the slope of the line shown? for we all remember that numerical data always has units. 3.) What would be a good title for this graph and explain your choice. 0.00 0.0 02 0.4 10.6 08 10 12 -0.20 -0.40 -0.60 -0.80 Temp, freezing, in degrees Celcius 5-1.00 -1.20 -1.40 -1:60 y=-1.823x-0.0162 -180 -2.00 Concentration of Sucrose (m)arrow_forwardDon't used Ai solutionarrow_forwardIdentify the Functional Groups (FG) in the following molecules. Classify C atoms as tertiary, 30, or quaternary 40. Identify secondary 20 and tertiary, 30 hydrogen atoms. Please provide steps to undertand each labeling. Please label in the image, so it fits explanation. I am still very unsure I undertand this.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





