
Chemistry
9th Edition
ISBN: 9781133611097
Author: Steven S. Zumdahl
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 33E
You have liquid in each graduated cylinder shown:
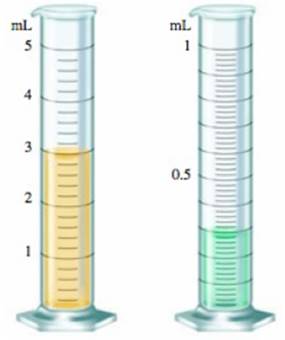
You then add both samples to a beaker. How would you write the number describing the total volume? What limits the precision of this number?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
(EXM 2, PRBLM 3) Here is this problem, can you explain it to me and show how its done. Thank you I need to see the work for like prbl solving.
can someone draw out the reaction mechanism for this reaction showing all bonds, intermediates and side products
Comment on the general features of the 1H-NMR spectrum of isoamyl ester provided below
What would be the best choices for the missing reagents 1 and 3 in this synthesis?
1. PPh3
3
2. n-BuLi
• Draw the missing reagents in the drawing area below. You can draw them in any arrangement you like.
• Do not draw the missing reagent 2. If you draw 1 correctly, we'll know what it is.
• Note: if one of your reagents needs to contain a halogen, use bromine.
Click and drag to start drawing a structure.
Chapter 1 Solutions
Chemistry
Ch. 1 - Define and explain the differences between the...Ch. 1 - Is the scientific method suitable for solving...Ch. 1 - Which of the following statements could be tested...Ch. 1 - For each of the following pieces of glassware,...Ch. 1 - A student performed an analysis of a sample for...Ch. 1 - Compare and contrast the multiplication/division...Ch. 1 - Explain how density can be used as a conversion...Ch. 1 - On which temperature scale (F, C. or K) docs 1...Ch. 1 - Distinguish between physical changes and chemical...Ch. 1 - Why is the separation of mixtures into pure or...
Ch. 1 - a. There are 365 days per year, 24 hours per day,...Ch. 1 - Prob. 2ALQCh. 1 - When a marble is dropped into a beaker of water,...Ch. 1 - Prob. 4ALQCh. 1 - You may have noticed that when water boils, you...Ch. 1 - If you place a glass rod over a burning candle,...Ch. 1 - Which characteristics of a solid, a liquid, and a...Ch. 1 - Sketch a magnified view (showing atoms/molecules)...Ch. 1 - Paracelsus, a sixteenth-century alchemist and...Ch. 1 - What is wrong with the following statement? "The...Ch. 1 - Why is it incorrect to say that the results of a...Ch. 1 - You have a 1.0-cm3 sample of lead and a 1.0-cm3...Ch. 1 - Consider the addition of 15.4 to 28. What would a...Ch. 1 - Consider multiplying 26.2 by 16.43. What would a...Ch. 1 - The difference between a law and a theory is the...Ch. 1 - The scientific method is a dynamic process. What...Ch. 1 - Explain the fundamental steps of the scientific...Ch. 1 - Prob. 20QCh. 1 - A measurement is a quantitative observation...Ch. 1 - To determine the volume of a cube, a student...Ch. 1 - What are significant figures? Show how to indicate...Ch. 1 - A cold front moves through and the temperature...Ch. 1 - Give four examples illustrating each of the...Ch. 1 - Which of the following are exact numbers? a. There...Ch. 1 - Indicate the number of significant figures in each...Ch. 1 - How many significant figures are there in each of...Ch. 1 - How many significant figures are in each of the...Ch. 1 - Round off each of the following numbers to the...Ch. 1 - Use exponential notation to express the number...Ch. 1 - You have liquid in each graduated cylinder shown:...Ch. 1 - The beakers shown below have different precisions....Ch. 1 - Evaluate each of the following, and write the...Ch. 1 - Perform the following mathematical operations, and...Ch. 1 - Perform the following mathematical operations, and...Ch. 1 - Perform the following mathematical operations, and...Ch. 1 - Perform each of the following conversions. a. 8.43...Ch. 1 - a. How many kilograms are in 1 teragram? b. How...Ch. 1 - Perform the following unit conversions. a....Ch. 1 - Perform the following unit conversions. a. 908 oz...Ch. 1 - Use the following exact conversion factors to...Ch. 1 - Although the preferred SI unit of area is the...Ch. 1 - Precious metals and gems are measured in troy...Ch. 1 - Apothecaries (druggists) use the following set of...Ch. 1 - For a pharmacist dispensing pills or capsules, it...Ch. 1 - A children's pain relief elixir contains 80. mg...Ch. 1 - Science fiction often uses nautical analogies to...Ch. 1 - The world record for the hundred meter dash is...Ch. 1 - Would a car traveling at a constant speed of 65...Ch. 1 - You pass a road sign saying New York 112 km. If...Ch. 1 - Prob. 53ECh. 1 - In recent years, there has been a large push for...Ch. 1 - Prob. 55ECh. 1 - Carbon monoxide (CO) detectors sound an alarm when...Ch. 1 - Convert the following Fahrenheit temperatures to...Ch. 1 - A thermometer gives a reading of 96.1F 0.2F. What...Ch. 1 - Convert the following Celsius temperatures to...Ch. 1 - Convert the following Kelvin temperatures to...Ch. 1 - At what temperature is the temperature in degrees...Ch. 1 - The average daytime temperatures on the earth and...Ch. 1 - Use the figure below to answer the following...Ch. 1 - Ethylene glycol is the main component in...Ch. 1 - A material will float on the surface of a liquid...Ch. 1 - Prob. 66ECh. 1 - A star is estimated to have a mass of 2 1036 kg....Ch. 1 - A rectangular block has dimensions 2.9 cm 3.5 cm ...Ch. 1 - Diamonds are measured in carats, and 1 carat =...Ch. 1 - Ethanol and benzene dissolve in each other. When...Ch. 1 - A sample containing 33.42 g of metal pellets is...Ch. 1 - The density of pure silver is 10.5 g/cm3 at 20C....Ch. 1 - In e-ach of the following pairs, which has the...Ch. 1 - a. Calculate the mass of ethanol in 1.50 qt of...Ch. 1 - In each of the following pairs, which has the...Ch. 1 - Using Table 1.5, calculate the volume of 25.0 g of...Ch. 1 - The density of osmium (the densest metal) is 22.57...Ch. 1 - A copper wire (density = 8.96 g/cm3) has a...Ch. 1 - Match each description below with the following...Ch. 1 - Define the following terms: solid, liquid, gas,...Ch. 1 - What is the difference between homogeneous and...Ch. 1 - Classify the following mixtures as homogeneous or...Ch. 1 - Classify each of the following as a mixture or a...Ch. 1 - Suppose a teaspoon of magnesium filings and a...Ch. 1 - If a piece of hard, white blackboard chalk is...Ch. 1 - During a very cold winter, the temperature may...Ch. 1 - Classify the following as physical or chemical...Ch. 1 - The properties of a mixture are typically averages...Ch. 1 - In Shakespeares Richard III, the First Murderer...Ch. 1 - Prob. 91AECh. 1 - In the opening scenes of the movie Raiders of the...Ch. 1 - Prob. 93AECh. 1 - This year, like many past years, you begin to feel...Ch. 1 - Which of the following are chemical changes? Which...Ch. 1 - A column of liquid is found to expand linearly on...Ch. 1 - A 25.00-g sample of a solid is placed in a...Ch. 1 - For each of the following, decide which block is...Ch. 1 - According to the Official Rules of Baseball, a...Ch. 1 - The density of an irregularly shaped object was...Ch. 1 - The chemist in Example 1.14 did some further...Ch. 1 - The longest river in the world is the Nile River...Ch. 1 - Secretariat is known as the horse with the fastest...Ch. 1 - The hottest temperature recorded in the United...Ch. 1 - Prob. 106CWPCh. 1 - Which of the following statements is(are) true? a....Ch. 1 - Which of the following describes a chemical...Ch. 1 - A rule of thumb in designing experiments is to...Ch. 1 - Draw a picture showing the markings (graduations)...Ch. 1 - Many times errors are expressed in terms of...Ch. 1 - A person weighed 15 pennies on a balance and...Ch. 1 - On October 21, 1982, the Bureau of the Mint...Ch. 1 - As part of a science project, you study traffic...Ch. 1 - Sterling silver is a solid solution of silver and...Ch. 1 - Make molecular-level (microscopic) drawings for...Ch. 1 - Confronted with the box shown in the diagram, you...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Identify the missing organic reactants in the following reaction: X + Y H+ two steps Note: This chemical equation only focuses on the important organic molecules in the reaction. Additional inorganic or small-molecule reactants or products (like H2O) are not shown. In the drawing area below, draw the skeletal ("line") structures of the missing organic reactants X and Y. You may draw the structures in any arrangement that you like, so long as they aren't touching. Click and drag to start drawing a structure. Х :arrow_forwardDraw the mechanism of friedel-crafts acylation using acetyl chloride of m-Xylenearrow_forwardI need help naming these in IUPACarrow_forward
- H R Part: 1/2 :CI: is a/an electrophile Part 2 of 2 Draw the skeletal structure of the product(s) for the Lewis acid-base reaction. Include lone pairs and formal charges (if applicable) on the structures. 4-7: H ö- H Skip Part Check X :C1: $ % L Fi Click and drag to start drawing a structure. MacBook Pro & ㅁ x G 0: P Add or increase positive formal cha Save For Later Submit ©2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Centearrow_forwardDraw the friedel-crafts acylation mechanism of m-Xylenearrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forward
- 1. Base on this experimental results, how do you know that the product which you are turning in is methyl 3-nitrobenzoate(meta substituted product ) rather than either of the other two products? 2. What observation suggests that at least a small amount of one or both of the other two isomers are in the mother liquor?arrow_forwardExplain Huckel's rule.arrow_forwardhere is my question can u help me please!arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Measurement and Significant Figures; Author: Professor Dave Explains;https://www.youtube.com/watch?v=Gn97hpEkTiM;License: Standard YouTube License, CC-BY
Trigonometry: Radians & Degrees (Section 3.2); Author: Math TV with Professor V;https://www.youtube.com/watch?v=U5a9e1J_V1Y;License: Standard YouTube License, CC-BY