
Concept explainers
(a)
Interpretation:
Whether the dew condensing on a leaf can be reversed by changing the temperature or not is to be determined.
Concept introduction:
The change that takes place only in state or appearance and not in the composition is known as physical change. The atoms or the molecules of a substance do not change their identity when a substance undergoes a physical change. The substance remains the same before and after the change. Physical change brings change in physical properties only. For example, the melting of ice is a physical change.
The physical change can be represented as follows:
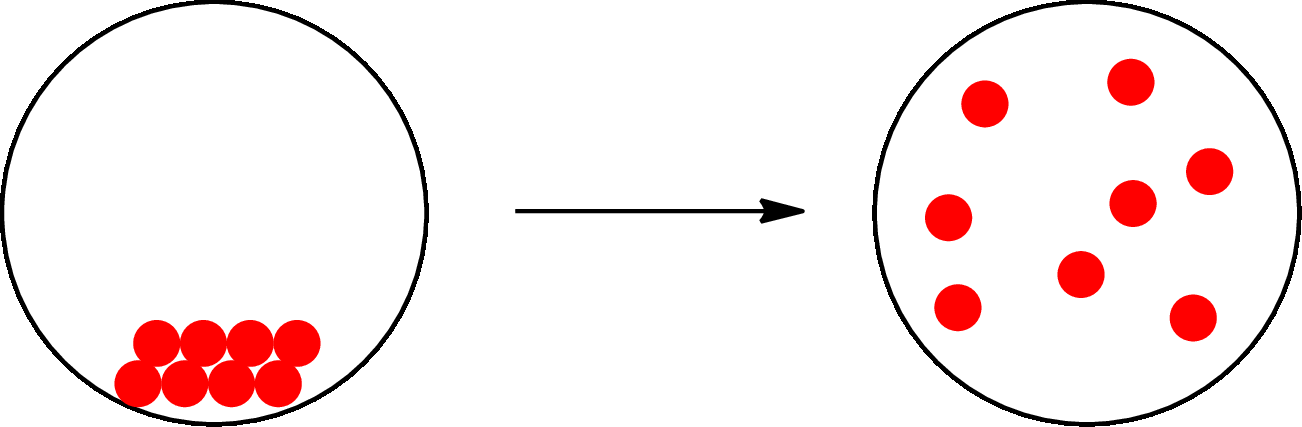
The change that takes place in the composition is known as chemical change. The atoms or the molecules of the substance rearrange and transformed into a new substance. A chemical change can change physical as well as chemical properties of a substance. For example, the burning of paper is a chemical change.
The representation of chemical change is as follows:
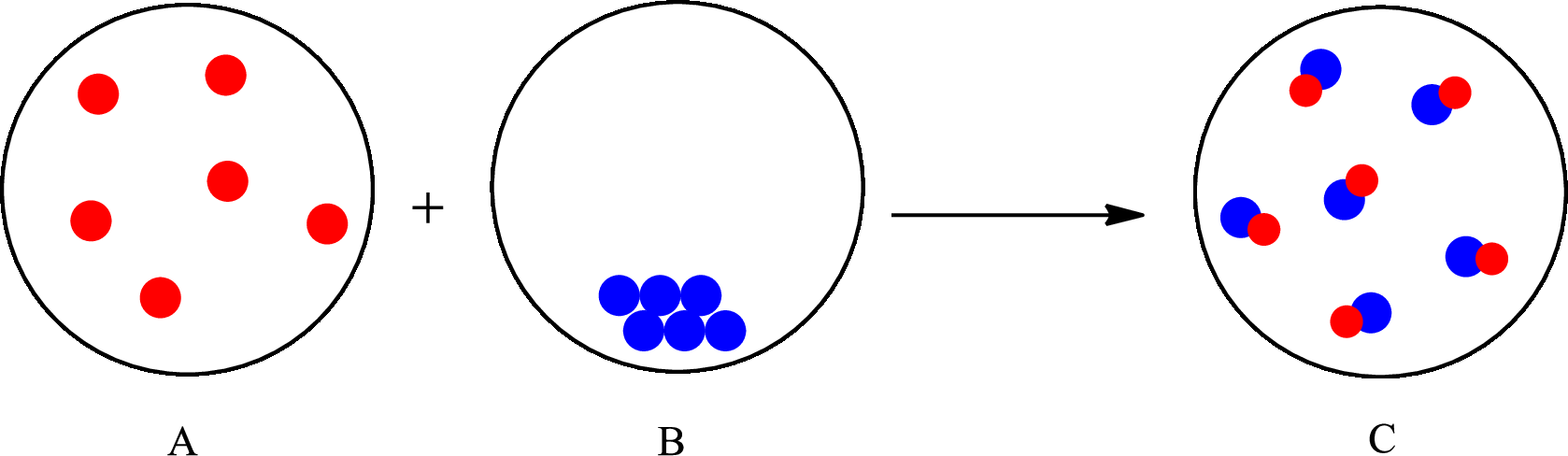
(b)
Interpretation:
Whether an egg turning hard due to boiling can be reversed by changing the temperature or not is to be determined.
Concept introduction:
The change that takes place only in state or appearance and not in the composition is known as physical change. The atoms or the molecules of a substance do not change their identity when a substance undergoes a physical change. The substance remains the same before and after the change. Physical change brings change in physical properties only. For example, the melting of ice is a physical change.
The physical change can be represented as follows:
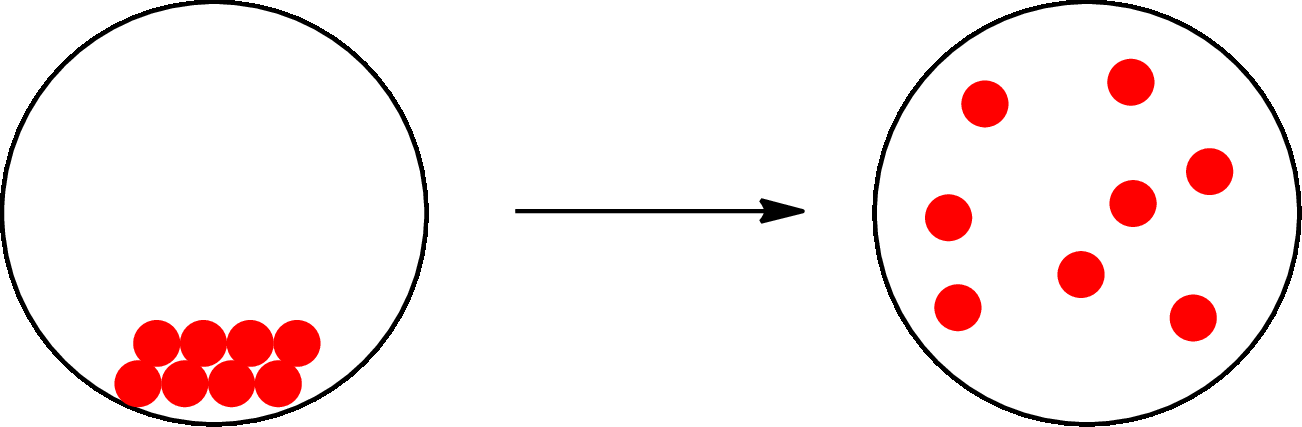
The change that takes place in the composition is known as chemical change. The atoms or the molecules of the substance rearrange and transformed into a new substance. A chemical change can change physical as well as chemical properties of a substance. For example, the burning of paper is a chemical change.
The representation of chemical change is as follows:
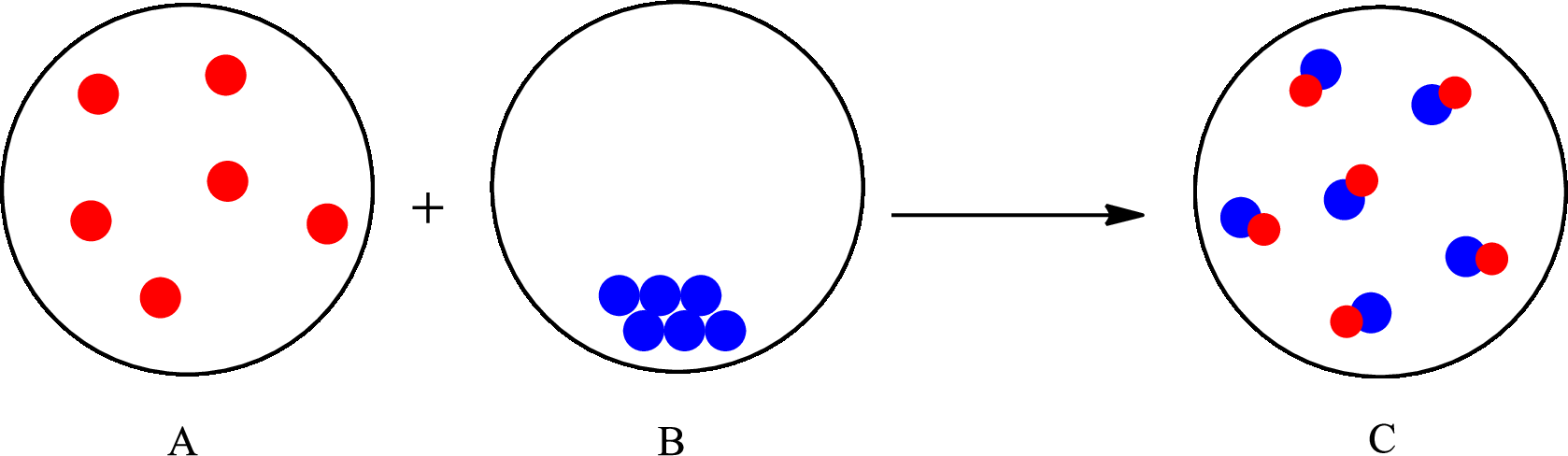
(c)
Interpretation:
Whether the melting of ice cream can be reversed by changing the temperature or not is to be determined.
Concept introduction:
The change that takes place only in state or appearance and not in the composition is known as physical change. The atoms or the molecules of a substance do not change their identity when a substance undergoes a physical change. The substance remains the same before and after the change. Physical change brings change in physical properties only. For example, the melting of ice is a physical change.
The physical change can be represented as follows:
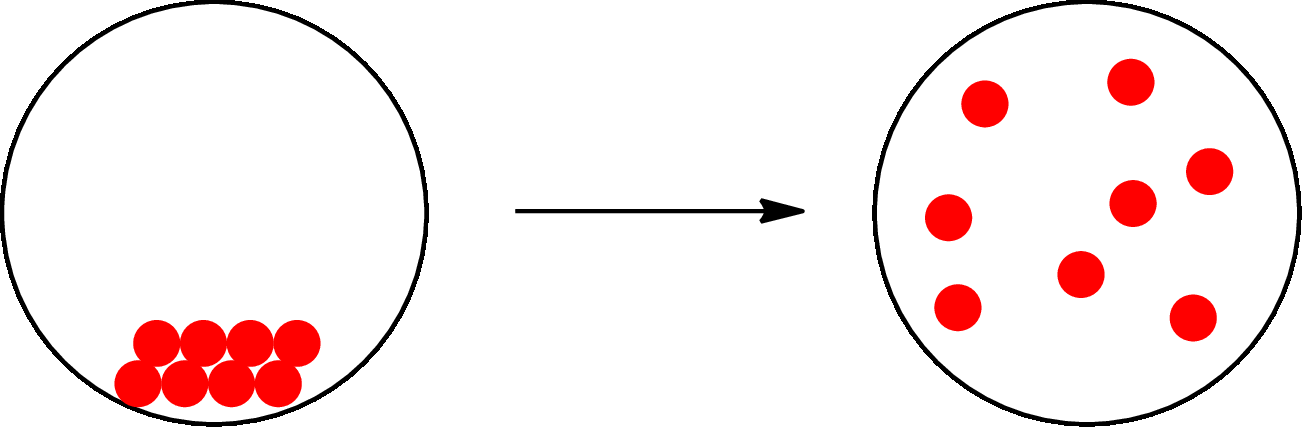
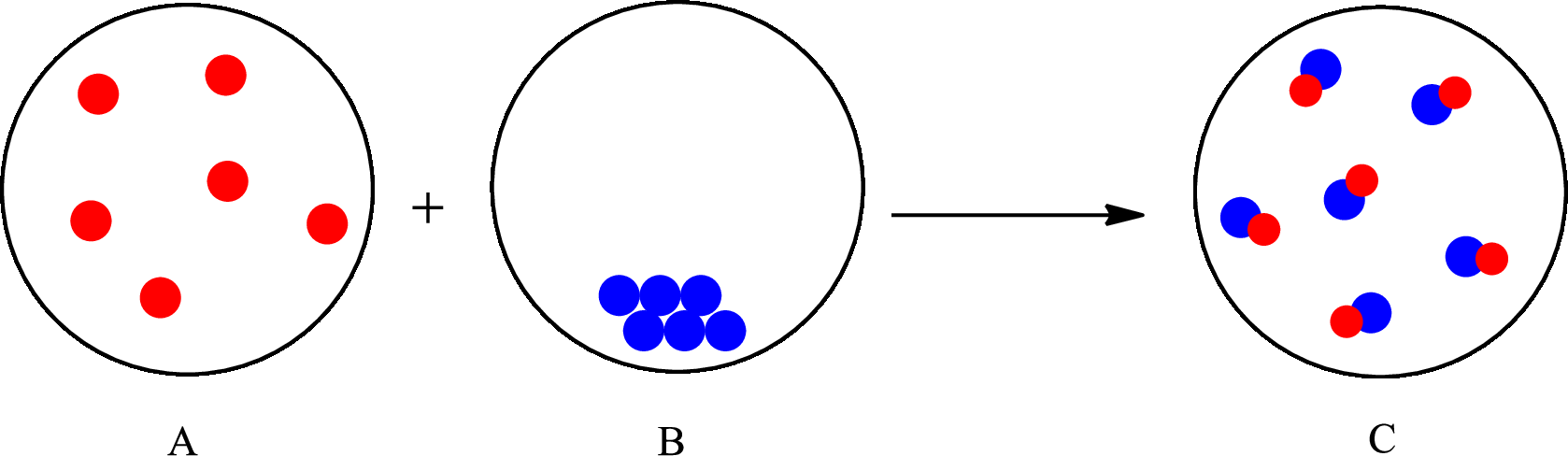
The change that takes place in the composition is known as chemical change. The atoms or the molecules of the substance rearrange and transformed into a new substance. A chemical change can change physical as well as chemical properties of a substance. For example, the burning of paper is a chemical change.
The representation of chemical change is as follows:
(d)
Interpretation:
Whether a spoonful of the batter cooking on a hot griddle can be reversed by changing the temperature or not is to be determined.
Concept introduction:
The change that takes place only in state or appearance and not in the composition is known as physical change. The atoms or the molecules of a substance do not change their identity when a substance undergoes a physical change. The substance remains the same before and after the change. Physical change brings change in physical properties only. For example, the melting of ice is a physical change.
The physical change can be represented as follows:
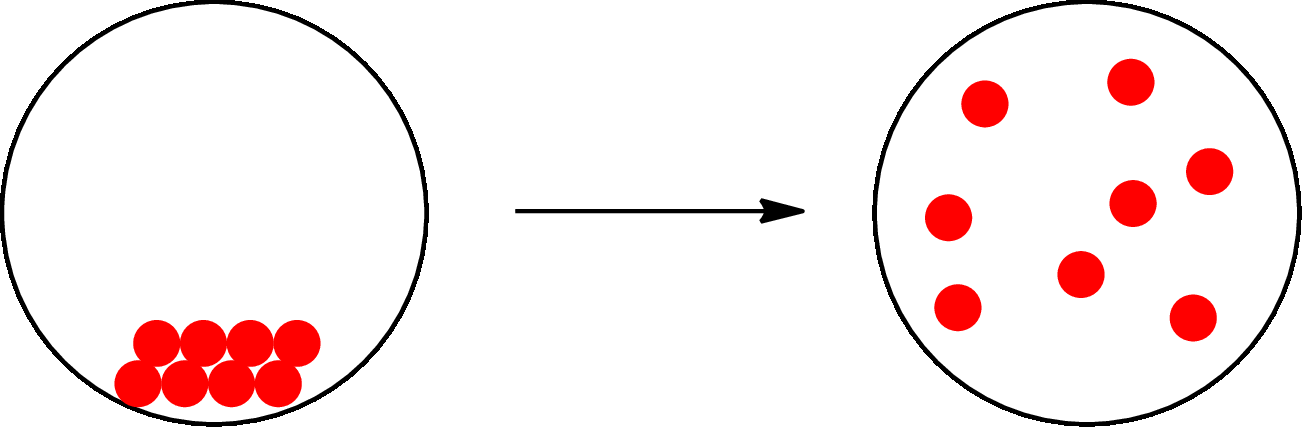
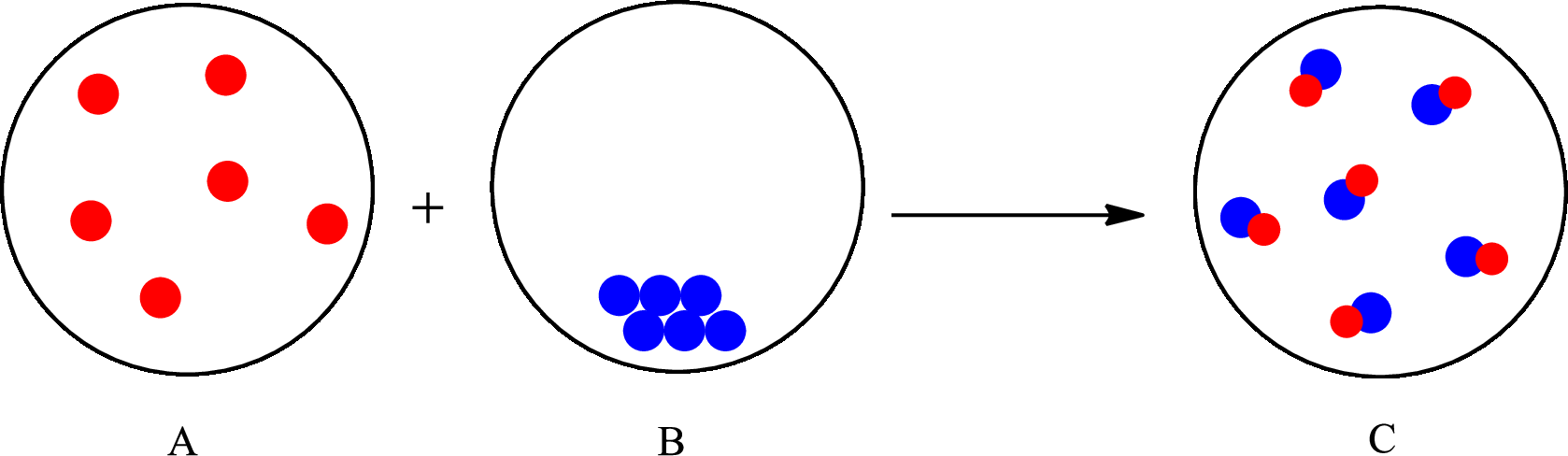
The change that takes place in the composition is known as chemical change. The atoms or the molecules of the substance rearrange and transformed into a new substance. A chemical change can change physical as well as chemical properties of a substance. For example, the burning of paper is a chemical change.
The representation of chemical change is as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Chemistry: The Molecular Nature of Matter and Change - Standalone book
- Using line angle formulas, draw thestructures of and name four alkanes that have total of 7carbons, one of which is tertiary.Please explain this in detail and can you also explain how to approach a similar problem like this as well?arrow_forwardUsing dashed line wedge projections drawthe indicated compounds and indicate whether thecompound you have drawn is R or S.(a) The two enantiomers of 2-chlorobutane. Can you please explain your steps and how you would approach a similar problem. Thank you!arrow_forward5) There are no lone pairs shown in the structure below. Please add in all lone pairs and then give the hybridization scheme for the compound. (8) 10,11 7) 1.2.3 H 4 | 14 8) COC 12 13 H 16 15 H7 9) - 5.6 C 8 H 10) H 1). 2) 3)_ 11) 12) 13) 4)_ 14) 5) 15) 16) 6)arrow_forward
- The sum of the numbers in the name of isA. 11; B. 13; C. 10; D. 12; E. none of the other answers iscorrect. I believe the awnser should be E to this problem but the solution to this problem is D 12. I'm honestly unsure how that's the solution. If you can please explain the steps to this type of problem and how to approach a problem like this it would be greatly appreciated!arrow_forwardConsider the following data for phosphorus: g atomic mass 30.974 mol electronegativity 2.19 kJ electron affinity 72. mol kJ ionization energy 1011.8 mol kJ heat of fusion 0.64 mol You may find additional useful data in the ALEKS Data tab. Does the following reaction absorb or release energy? 2+ + (1) P (g) + e → P (g) Is it possible to calculate the amount of energy absorbed or released by reaction (1) using only the data above? If you answered yes to the previous question, enter the amount of energy absorbed or released by reaction (1): Does the following reaction absorb or release energy? 00 release absorb Can't be decided with the data given. yes no ☐ kJ/mol (²) P* (8) + + + e →>> P (g) Is it possible to calculate the amount of energy absorbed or released by reaction (2) using only the data above? If you answered yes to the previous question, enter the amount of energy absorbed or released by reaction (2): ☐ release absorb Can't be decided with the data given. yes no kJ/mol аarrow_forwardThe number of hydrogens in an alkyne that has a main chain of 14carbons to which are attached a cyclobutyl ring, a benzene ring, an–OH group, and a Br is A. 34; B. 35; C. 36; D. 24; E. 43arrow_forward
- Hello! I have a 500 Hz H-NMR for 1,5-bis-(4-methoxyphenyl)-penta-1,4-dien-3-one. I need to label the signals with the corresponding H's. Then, find out if the two alkenes are cis or trans by calculating the J values. I believe that I have the H-NMR labeled correctly, but not sure if I got the J values correct to determine if the two alkenes in the compound will make the compound cis or trans.arrow_forwardWhat is the only possible H-Sb-H bond angle in SbH3?arrow_forwardpls helparrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





