
EP FUND.OF GENERAL,ORG...-MOD.MASTERING
8th Edition
ISBN: 9780134326061
Author: McMurry
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 9, Problem 9.31UKC
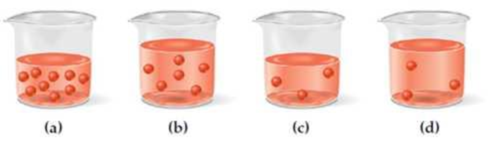
A beaker containing 150.0 mL of 0.1 M glucose is represented by (a). Which of the drawings (b)–(d) represents the solution that results when 50.0 mL is withdrawn from (a) and then diluted by a factor of 4?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Given the values for the changes in enthalpy (AH) and entropy (AS), which processes can take place at 298 K without
violating the Second Law of Thermodynamics?
(a) AH =
(b) AH = -84 kJ mol¹ (−20 kcal mol¹), AS = -125 J mol-¹ K-¹ (-30 cal mol-¹ K-¹)
(c) AH = +84 kJ mol¹ (+20 kcal mol¯¹), AS = +125 J mol¯¹ K−¹ (+30 cal mol-¹ K-¹)
(d) AH = +84 kJ mol-1 (+20 kcal mol-¹), AS = -125 J mol¯¹ K-¹ (-30 cal mol-¹ K-¹)
-84 kJ mol¹ (-20 kcal mol¹), AS = +125 J mol¯¹ K-¹ (+30 cal mol-¹ K-¹)
What chemical interactions are primarily responsible for the fact that oil and water do not mix easily?
ionic interactions
covalent bonds
hydrogen bonding
Ohydrophobic effect
The carbon-nitrogen peptide bond is rigid, but rotation can take place
about the bond between the nitrogen and α-carbon atoms and the bond
between the α-carbon and the carbonyl carbon atom. These bond angles
define the conformation of the peptide chain.
Consider the Ramachandran plot shown to answer both questions.
(degrees)
180
120
60
60
0
Which observation most likely describes part of an observed a helix?
p=-59 degrees; = +150 degrees; many Ser residues
cp-140 degrees; w = +130 degrees; many Val residues
op-57 degrees; w = -47 degrees; many Pro residues
op-60 degrees; y = -47 degrees; many Ala residues
p = +60 degrees; y = +40 degrees; many Gly residues
-60
-120
-180
-180
0
➜ (degrees)
180
Chapter 9 Solutions
EP FUND.OF GENERAL,ORG...-MOD.MASTERING
Ch. 9.1 - Classify the following liquid mixtures as...Ch. 9.2 - Write the formula of sodium sulfate decahydrate,...Ch. 9.2 - What mass of Glaubers salt must be used to provide...Ch. 9.2 - Which of the following pairs of substances would...Ch. 9.4 - A solution is prepared by dissolving 12.5 g of KBr...Ch. 9.5 - Prob. 9.4PCh. 9.5 - Prob. 9.5PCh. 9.6 - How does the body increase oxygen availability at...Ch. 9.6 - A metal alloy contains 15.8% nickel (m/m)%. What...Ch. 9.6 - Prob. 9.7P
Ch. 9.6 - Prob. 9.8PCh. 9.6 - Prob. 9.9PCh. 9.6 - Prob. 9.10PCh. 9.6 - Prob. 9.11PCh. 9.6 - What is the molarity of a solution that contains...Ch. 9.6 - How many moles of solute are present in the...Ch. 9.6 - The concentration of cholesterol (C27H46O) in...Ch. 9.6 - Prob. 9.15PCh. 9.7 - Prob. 9.16PCh. 9.7 - Prob. 9.17PCh. 9.8 - Prob. 9.5CIAPCh. 9.8 - Prob. 9.6CIAPCh. 9.8 - Prob. 9.7CIAPCh. 9.8 - Prob. 9.18PCh. 9.8 - Prob. 9.19PCh. 9.9 - A solution is prepared by dissolving 0.67 mol of...Ch. 9.9 - Prob. 9.21PCh. 9.9 - Prob. 9.22KCPCh. 9.9 - What is the freezing point of a solution of 1.0...Ch. 9.9 - Prob. 9.24PCh. 9.10 - What is the osmolarity of the following solutions?...Ch. 9.10 - Prob. 9.26PCh. 9.11 - Prob. 9.8CIAPCh. 9.11 - Prob. 9.9CIAPCh. 9 - Assume that two liquids are separated by a...Ch. 9 - Prob. 9.28UKCCh. 9 - Prob. 9.29UKCCh. 9 - Assume that you have two full beakers, one...Ch. 9 - A beaker containing 150.0 mL of 0.1 M glucose is...Ch. 9 - Prob. 9.32APCh. 9 - Prob. 9.33APCh. 9 - Prob. 9.34APCh. 9 - Prob. 9.35APCh. 9 - Which of the following are solutions? (a)Italian...Ch. 9 - Prob. 9.37APCh. 9 - The solubility of NH3 gas in water at an NH3...Ch. 9 - Prob. 9.39APCh. 9 - Is a solution highly concentrated if it is...Ch. 9 - How is mass/volume percent concentration defined...Ch. 9 - Prob. 9.42APCh. 9 - Prob. 9.43APCh. 9 - A 750.0 mL bottle of Listerine is of a 21% (v/v)...Ch. 9 - Prob. 9.45APCh. 9 - Prob. 9.46APCh. 9 - Prob. 9.47APCh. 9 - Prob. 9.48APCh. 9 - Prob. 9.49APCh. 9 - How many moles of each substance are needed to...Ch. 9 - Prob. 9.51APCh. 9 - If you had only 23 g of KOH remaining in a bottle,...Ch. 9 - Prob. 9.53APCh. 9 - The lethal dosage of potassium cyanide (KCN) in...Ch. 9 - What is the molarity of the following solutions?...Ch. 9 - How many grams of solute are in the following...Ch. 9 - How many milliliters of a 0.75 M HCl solution do...Ch. 9 - Prob. 9.58APCh. 9 - Prob. 9.59APCh. 9 - Sodium thiosulfate (Na2S2O3), the major component...Ch. 9 - Prob. 9.61APCh. 9 - What is the final volume of NaOH solution prepared...Ch. 9 - Prob. 9.63APCh. 9 - What is the concentration of a NaCl solution, in...Ch. 9 - Concentrated (12.0 M) hydrochloric acid is sold...Ch. 9 - Dilute solutions of NaHCO3 are sometimes used in...Ch. 9 - Prob. 9.67APCh. 9 - Prob. 9.68APCh. 9 - Prob. 9.69APCh. 9 - What is the total anion concentration (in mEq/L)...Ch. 9 - Kaochlor, a 10% (m/v) KCl solution, is an oral...Ch. 9 - Prob. 9.72APCh. 9 - The concentration of Cl ion in blood is...Ch. 9 - Prob. 9.74APCh. 9 - Which lowers the freezing point of 2.0 kg of water...Ch. 9 - Which solution has the higher boiling point, 0.500...Ch. 9 - Methanol, CH3OH, is sometimes used as an...Ch. 9 - Hard candy is prepared by dissolving pure sugar...Ch. 9 - Prob. 9.79APCh. 9 - Prob. 9.80APCh. 9 - Which of the following solutions has the higher...Ch. 9 - Which of the following solutions will give rise to...Ch. 9 - A pickling solution for preserving food is...Ch. 9 - Prob. 9.84APCh. 9 - Prob. 9.85CPCh. 9 - Emergency treatment of cardiac arrest victims...Ch. 9 - Nitric acid, HNO3, is available commercially at a...Ch. 9 - Prob. 9.88CPCh. 9 - A typical dosage of statin drugs for the treatment...Ch. 9 - Prob. 9.90CPCh. 9 - Prob. 9.91CPCh. 9 - Prob. 9.92CPCh. 9 - Prob. 9.93CPCh. 9 - Prob. 9.94CPCh. 9 - Prob. 9.95CPCh. 9 - Prob. 9.96GPCh. 9 - Look up the maximum concentrations set by the U.S....Ch. 9 - Look up the composition of Ringers solution used...Ch. 9 - To prevent accumulation of ice on roads and...Ch. 9 - Many carbonate minerals are insoluble in water and...Ch. 9 - Prob. 9.101GP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- The α and ẞ subunits of hemoglobin bear a remarkable structural similarity to myoglobin. However, certain residues that are hydrophilic in myoglobin are hydrophobic in the subunits of hemoglobin. Why might this be the case? Hemoglobin forms long, extended structures that feature repeated sequences, whereas myoglobin forms globular structures. Hydrophobic residues on the surface of hemoglobin subunits interact with similar regions on the other subunits through van der Waals interactions. Hydrophilic residues on the surface of myoglobin form ionic interactions with similar regions on other myoglobin molecules. Myoglobin is a water-soluble protein, whereas hemoglobin is found in the hydrophobic environment of membranes.arrow_forwardProtein disulfide isomerase (PDI) is a protein folding enzyme that catalyzes disulfide-sulfhydryl exchange reactions. Ribonuclease is an enzyme with two ẞ-sheets, three α-helices, and four disulfide bridges critical to the stability of its structure. PDI rapidly converts inactive, scrambled ribonuclease to enzymatically active ribonuclease. This process is driven by the decrease in free energy as the scrambled conformations are converted into the stable, active conformation of the enzyme. In contrast, PDI rapidly inactivates insulin. What does PDI's effect on insulin imply about the structure of insulin? The three-dimensional shape of insulin does not contain any disulfide bonds. The active conformation of insulin has more ẞ-sheets than α-helices. The active conformation of insulin is not the most thermodynamically preferred form. The amino acid sequence of insulin is similar to the amino acid sequence of ribonuclease.arrow_forwardIn the 1950s, Christian Anfinsen demonstrated the renaturation of the protein ribonuclease (RNase) in vitro. After reduction and the addition of urea, the protein was in an unfolded state. After removing the urea and then the reducing agent, the protein oxidized and refolded, with greater than 90% activity. If reducing agent removal occurs before removing the urea, the protein showed less than 5% activity. Why does RNase refold incorrectly if the reducing agent is removed before urea removal? Contaminants in the RNase preparation would form covalent bonds with the protein, preventing reactivation. Urea would participate in weak bonding interactions with RNase, preventing oxidation of cysteine. Disulfide bonds are not positioned correctly unless weak bonding interactions are present. The protein would not fully denature.arrow_forward
- A hospital blood processing unit uses apheresis to concentrate red blood cells (RBCs) fromwhole blood donations. Whole blood is drawn from a donor at a rate of 100 g/min and passedthrough a centrifuge-based apheresis system. The whole blood contains 45% RBC and 1%WBCs and platelets by mass, with the remainder consisting of plasma. The separated plasma andnon-RBC components are returned to the donor at a rate of 90 g/min.The goal of the system is to collect a concentrated RBC fraction containing 80% red blood cells.Assume that red blood cells do not break down during processing and that no fluid loss occurs inthe system.(a) How long will it take to collect 100g of the concentrated RBC fraction?arrow_forwardPlease give an intuitive explanation of the blood-brain barrier. Specifically, what is it, what is its structure and function, what types of cells compose it, etc.,..?arrow_forwardObtain the sequence for the 5-HT receptor HTR1A and generate a hydropathy plot usingthe ExPASY tool ProtScale, the appropriate window, and the Kyte-Doolittle weightingalgorithm. How many transmembrane domains are present in this receptor?arrow_forward
- From the reaction data below, determine whether the reaction is first order or second order and calculate the rate constant. Time (s) 0 Reactant (mM) 5.4 1 4.6 2 3.9 3 3.2 4 2.7 5 2.3 Only a plot of In[reactant] versus t gives a straight line, so the reaction is first order . The negative of the slope, k, is 0.171arrow_forwardHair grows at a rate of about 20 cm/yr. All this growth is concentrated at the base of the hair fiber, where a-keratin filaments are synthesized inside living epidermal cells and assembled into ropelike structures. Two-chan 14 Protofilament 20-30 A Two-chain Intermediate flament -Protob Protofilament Cross section of a hair The fundamental structural element of a keratin is the a helix, which has 3.6 amino acid residues per turn and a rise of 5.4 A perlum. 54A (36) Amino terminus Carbon Hydrogen Oxygen Nitrogen group Carboxyl terminus Assuming that the biosynthesis of a helical keratin chains is the rate-limiting factor in the growth of hair, calculate the rate at which peptide bonds of a-keratin chains must be synthesized (peptide bonds per second) to account for the observed yearly growth of hair. 0422 rate of peptide bond formation: Income bonds/sarrow_forwardSpecific rotation is a measure of a solution's capacity to rotate circularly polarized light. The unfolding of the a helix of a polypeptide to a random conformation is accompanied by a large decrease in specific rotation. Polyglutamate, a polypeptide made up of only 1-Glu residues, has the a helix conformation at pH 3. When researchers raise the pH to 7, there is a large decrease in the specific rotation of the solution. Similarly, polylysine (1.-Lys residues) is an a helix at pH 10, but when researchers lower the pH to 7 the specific rotation also decreases, as shown in the graph. a Helix Specific rotation Poly(Glu) a Helix Random conformation Poly(Lys) Random conformation T + ° 2 4 6 В 10 12 14 PH Complete the statements about the molecular mechanism for these changes in specific rotation. Increasing the pH of a polyglutamate solution from 6 to 7 causes the carboxyl group of each glutamate residue Comed Artwer lose a proton. The negatively charged groups in each glutamate residue…arrow_forward
- From the reaction data below, determine whether the reaction is first order or second order and calculate the rate constant. Time (s) 0 Reactant (mM) 6.2 1 3.1 2 2.1 3 1.6 4 1.3 5 1.1 Only a plot of 1/[reactant] versus t gives a straight line, so the reaction is 0.150 mM-1 s-1 . S second order . The slope, k, isarrow_forwardFrom the reaction data below, determine whether the reaction is first order or second order and calculate the rate constant. Time (s) 0 Reactant (mM) 5.4 1 4.6 2 3.9 3 3.2 4 2.7 5 2.3 Only a plot of In[reactant] versus t gives a straight line, so the reaction is s-1. . The negative of the slope, k, isarrow_forwardA protein has a molecular mass of 400 kDa when measured by size-exclusion chromatography. When subjected to gel electrophoresis in the presence of sodium dodecyl sulfate (SDS), the protein gives three bands with molecular masses of 180, 160, and 60 kDa. When electrophoresis is carried out in the presence of SDS and dithiothreitol (DTT), three bands again form, this time with molecular masses of 160, 90, and 60 kDa. How many subunits does the protein have, and what is the molecular mass of each? four subunits: 180, 160, 90, and 60 kDa three subunits: 180, 160, and 60 kDa three subunits: 160, 90, and 60 kDa four subunits: 160, 90, 90, and 60 kDa Correct Answerarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Essentials of Pharmacology for Health ProfessionsNursingISBN:9781305441620Author:WOODROWPublisher:Cengage
 Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning



Essentials of Pharmacology for Health Professions
Nursing
ISBN:9781305441620
Author:WOODROW
Publisher:Cengage



Human Physiology: From Cells to Systems (MindTap ...
Biology
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Cengage Learning
GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license