
Concept explainers
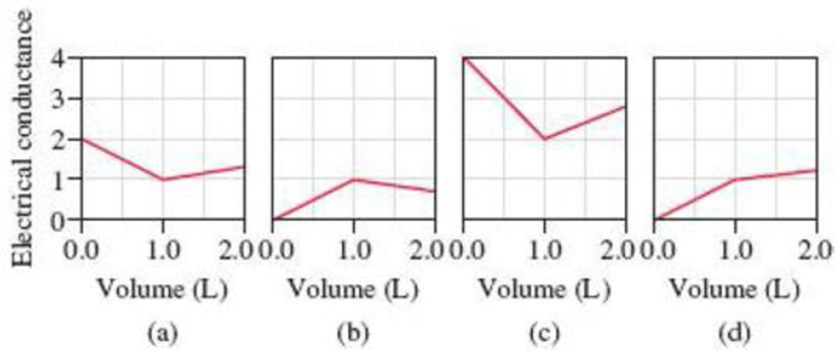
Because the acid-base and precipitation reactions discussed in this chapter all involve ionic species, their progress can be monitored by measuring the electrical conductance of the solution. Match each of the following reactions with one of the diagrams shown here. The electrical conductance is shown in arbitrary units. Explain the significance of the point at which the slope changes in each diagram.
(1) A 1.0 M KOH solution is added to 1.0 L of 1.0 M HC2H3O2.
(2) A 1.0 M NaOH solution is added to 1.0 L of 1.0 M HCl.
(3) A 1.0 M BaCl2 solution is added to 1.0 L of 1.0 M K2SO4.
(4) A 1.0 M NaCl solution is added to 1.0 L of 1.0 M AgNO3.
(5) A 1.0 M HC2H3O2 solution is added to 1.0 L of 1.0 M NH3.
(a)
Interpretation:
The given each reaction are should be matched with given each diagram and significance of slope change points in the given diagrams should be explained.
Concept introduction:
Precipitation reaction:
- If precipitate is formed, when two solutions are mixed together is called precipitation reaction.
- The amount of precipitate formed is related to the amount of reactants taken in to the reaction.
Neutralization reaction:
- The reaction between acid and base to gives a salt is the known as neutralization reaction.
Strong and weak electrolytes:
- The compound dissolved in water and completely dissociates to produces the ions is known as strong electrolytes.
- The compound dissolved in water but not completely dissociates to produces the ions is known as strong electrolytes.
Electrical conductivity of electrolytes:
- The strong electrolytes are having high electrical conductivity than weak electrolytes.
- The number of ion in solution is directly proportional to the electrical conductivity of electrolytes.
Conductivity titration:
- The measurement of electrical conductivity of titration mixture to gives a end point if the reaction.
- The sudden change in the slope is a equivalent point of the titration and it is the end point.
To find the electrical conductance, when
Answer to Problem 9.168QP
- The reactions (2) and (4) are matched with diagram (a).
- The reaction (5) is matched with diagram (b).
- The reaction (3) is matched with diagram (c).
- The reaction (1) is matched with diagram (d).
The slope change points in the given diagrams are end or equivalent points of the tractions.
Record the given data
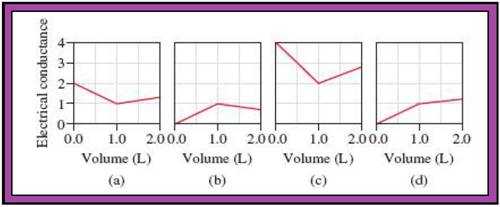
Fig.1
Explanation of Solution
If the conductance unit will be twice its concentration (molarity), when compound is completely dissociates into equal number of ions in solution.
Reaction of
Volume of
If
If
If Conductance unit of
(b)
Interpretation:
The given each reaction are should be matched with given each diagram and significance of slope change points in the given diagrams should be explained.
Concept introduction:
Precipitation reaction:
- If precipitate is formed, when two solutions are mixed together is called precipitation reaction.
- The amount of precipitate formed is related to the amount of reactants taken in to the reaction.
Neutralization reaction:
- The reaction between acid and base to gives a salt is the known as neutralization reaction.
Strong and weak electrolytes:
- The compound dissolved in water and completely dissociates to produces the ions is known as strong electrolytes.
- The compound dissolved in water but not completely dissociates to produces the ions is known as strong electrolytes.
Electrical conductivity of electrolytes:
- The strong electrolytes are having high electrical conductivity than weak electrolytes.
- The number of ion in solution is directly proportional to the electrical conductivity of electrolytes.
Conductivity titration:
- The measurement of electrical conductivity of titration mixture to gives a end point if the reaction.
- The sudden change in the slope is a equivalent point of the titration and it is the end point.
To find the electrical conductance when,
Answer to Problem 9.168QP
- The reactions (2) and (4) are matched with diagram (a).
- The reaction (5) is matched with diagram (b).
- The reaction (3) is matched with diagram (c).
- The reaction (1) is matched with diagram (d).
The slope change points in the given diagrams are end or equivalent points of the tractions.
Record the given data

Fig.1
Explanation of Solution
If the conductance unit will be twice its concentration (molarity), when compound is completely dissociates into equal number of ions in solution.
Reaction of
Volume of
If
If
(c)
Interpretation:
The given each reaction are should be matched with given each diagram and significance of slope change points in the given diagrams should be explained.
Concept introduction:
Precipitation reaction:
- If precipitate is formed, when two solutions are mixed together is called precipitation reaction.
- The amount of precipitate formed is related to the amount of reactants taken in to the reaction.
Neutralization reaction:
- The reaction between acid and base to gives a salt is the known as neutralization reaction.
Strong and weak electrolytes:
- The compound dissolved in water and completely dissociates to produces the ions is known as strong electrolytes.
- The compound dissolved in water but not completely dissociates to produces the ions is known as strong electrolytes.
Electrical conductivity of electrolytes:
- The strong electrolytes are having high electrical conductivity than weak electrolytes.
- The number of ion in solution is directly proportional to the electrical conductivity of electrolytes.
Conductivity titration:
- The measurement of electrical conductivity of titration mixture to gives a end point if the reaction.
- The sudden change in the slope is a equivalent point of the titration and it is the end point.
To find the electrical conductance when,
Answer to Problem 9.168QP
- The reactions (2) and (4) are matched with diagram (a).
- The reaction (5) is matched with diagram (b).
- The reaction (3) is matched with diagram (c).
- The reaction (1) is matched with diagram (d).
The slope change points in the given diagrams are end or equivalent points of the tractions.
Record the given data

Fig.1
Explanation of Solution
If the conductance unit will be twice its concentration (molarity), when compound is completely dissociates into equal number of ions in solution.
Reaction of
Volume of
If
If
(d)
Interpretation:
The given each reaction are should be matched with given each diagram and significance of slope change points in the given diagrams should be explained.
Concept introduction:
Precipitation reaction:
- If precipitate is formed, when two solutions are mixed together is called precipitation reaction.
- The amount of precipitate formed is related to the amount of reactants taken in to the reaction.
Neutralization reaction:
- The reaction between acid and base to gives a salt is the known as neutralization reaction.
Strong and weak electrolytes:
- The compound dissolved in water and completely dissociates to produces the ions is known as strong electrolytes.
- The compound dissolved in water but not completely dissociates to produces the ions is known as strong electrolytes.
Electrical conductivity of electrolytes:
- The strong electrolytes are having high electrical conductivity than weak electrolytes.
- The number of ion in solution is directly proportional to the electrical conductivity of electrolytes.
Conductivity titration:
- The measurement of electrical conductivity of titration mixture to gives a end point if the reaction.
- The sudden change in the slope is a equivalent point of the titration and it is the end point.
To find the electrical conductance, when
Answer to Problem 9.168QP
- The reactions (2) and (4) are matched with diagram (a).
- The reaction (5) is matched with diagram (b).
- The reaction (3) is matched with diagram (c).
- The reaction (1) is matched with diagram (d).
The slope change points in the given diagrams are end or equivalent points of the tractions.
Record the given data

Fig.1
Explanation of Solution
If the conductance unit will be twice its concentration (molarity), when compound is completely dissociates into equal number of ions in solution.
Reaction of
Volume of
If
If
(e)
Interpretation:
The given each reaction are should be matched with given each diagram and significance of slope change points in the given diagrams should be explained.
Concept introduction:
Precipitation reaction:
- If precipitate is formed, when two solutions are mixed together is called precipitation reaction.
- The amount of precipitate formed is related to the amount of reactants taken in to the reaction.
Neutralization reaction:
- The reaction between acid and base to gives a salt is the known as neutralization reaction.
Strong and weak electrolytes:
- The compound dissolved in water and completely dissociates to produces the ions is known as strong electrolytes.
- The compound dissolved in water but not completely dissociates to produces the ions is known as strong electrolytes.
Electrical conductivity of electrolytes:
- The strong electrolytes are having high electrical conductivity than weak electrolytes.
- The number of ion in solution is directly proportional to the electrical conductivity of electrolytes.
Conductivity titration:
- The measurement of electrical conductivity of titration mixture to gives a end point if the reaction.
- The sudden change in the slope is a equivalent point of the titration and it is the end point.
To find the electrical conductance, when
Answer to Problem 9.168QP
- The reactions (2) and (4) are matched with diagram (a).
- The reaction (5) is matched with diagram (b).
- The reaction (3) is matched with diagram (c).
- The reaction (1) is matched with diagram (d).
The slope change points in the given diagrams are end or equivalent points of the tractions.
Record the given data

Fig.1
Explanation of Solution
If the conductance unit will be twice its concentration (molarity), when compound is completely dissociates into equal number of ions in solution.
Reaction of
Volume of
If
If
Match the calculated conductance unit of each reaction in given diagrams in Fig.1.
- The reactions (2) and (4) are matched with diagram (a).
- The reaction (5) is matched with diagram (b).
- The reaction (3) is matched with diagram (c).
- The reaction (1) is matched with diagram (d).
The slope change points in the given diagrams are end or equivalent points of the tractions.
Want to see more full solutions like this?
Chapter 9 Solutions
Chemistry: Atoms First
- CH3 CH H3C CH3 H OH H3C- -OCH2CH3 H3C H -OCH3 For each of the above compounds, do the following: 1. List the wave numbers of all the IR bands in the 1350-4000 cm-1 region. For each one, state what bond or group it represents. 2. Label equivalent sets of protons with lower-case letters. Then, for each 1H NMR signal, give the 8 value, the type of splitting (singlet, doublet etc.), and the number protons it represents. of letter δ value splitting # of protons 3. Redraw the compound and label equivalent sets of carbons with lower-case letters. Then for each set of carbons give the 5 value and # of carbons it represents. letter δ value # of carbonsarrow_forwardDraw the correct ionic form(s) of arginine at the pKa and PI in your titration curve. Use your titration curve to help you determine which form(s) to draw out.arrow_forwardPlease correct answer and don't use hand ratingarrow_forward
- Nonearrow_forwardNonearrow_forwardCarbohydrates- Draw out the Hawthorne structure for a sugar from the list given in class. Make sure to write out all atoms except for carbons within the ring. Make sure that groups off the carbons in the ring are in the correct orientation above or below the plane. Make sure that bonds are in the correct orientation. Include the full name of the sugar. You can draw out your curve within the text box or upload a drawing below.arrow_forward
- How many milliliters of 97.5(±0.5) wt% H2SO4 with a density of 1.84(±0.01) g/mL will you need to prepare 2.000 L of 0.110 M H2SO4? If the uncertainty in delivering H2SO4 is ±0.01 mL, calculate the absolute uncertainty in the molarity (0.110 M). Assume there is negligible uncertainty in the formula mass of H2SO4 and in the final volume (2.000 L) and assume random error.arrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forwardWhat should be use to complete the reaction? CN CNarrow_forward
- Don't used hand raiting and don't used Ai solutionarrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forward* How many milliliters of 97.5(±0.5) wt% H2SO4 with a density of 1.84(±0.01) g/mL will you need to prepare 2.000 L of 0.110 M H2SO4? * If the uncertainty in delivering H2SO4 is ±0.01 mL, calculate the absolute uncertainty in the molarity (0.110 M). Assume there is negligible uncertainty in the formula mass of NaOH and in the final volume (2.000 L) and assume random error.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





