
CHEMISTRY-TEXT
8th Edition
ISBN: 9780134856230
Author: Robinson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 9.117SP
Isooctane,
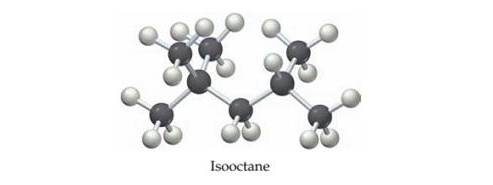
(a) Write a balanced equation for the combustion of isooctane (l) with O2 to yield CO2(g) and
(b) The standard molar heat of combustion for isooctane (l)is -5461 kJ/mol. Calculate
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
QUESTION: Fill in the answers in the empty green boxes regarding 'Question 5: Calculating standard error of regression'
*The images of the data showing 'coefficients for the standard curve' have been provided
Using the Nernst equation to calculate nonstandard cell voltage
Try Again
Your answer is wrong. In addition to checking your math, check that you used the right data and DID NOT round any intermediate calculations.
A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction:
2+
2+
Sn²+ Ba(s)
(aq) + Ba (s) Sn (s) + Ba²+ (aq)
→>>
Suppose the cell is prepared with 6.10 M Sn
2+
2+
in one half-cell and 6.62 M Ba
in the other.
Calculate the cell voltage under these conditions. Round your answer to 3 significant digits.
1.71 V
☐ x10
☑
5
0/5
?
00.
18
Ar
Question: Find both the b (gradient) and a (y-intercept) value from the list of data below:
(x1 -x̄)
370.5
(y1 - ȳ)
5.240
(x2 - x̄)
142.5
(y2 - ȳ)
2.004
(x3 - x̄)
28.5
(y3 - ȳ)
0.390
(x4 - x̄)
-85.5
(y4 - ȳ)
-1.231
(x5 - x̄)
-199.5
(y5 - ȳ)
-2.829
(x6 - x̄)
-256.5
(y6 - ȳ)
-3.575
Chapter 9 Solutions
CHEMISTRY-TEXT
Ch. 9 - Prob. 9.1PCh. 9 - Conceptual APPLY 9.2 How much work is done in...Ch. 9 - PRACTICE 9.3 The reaction between hydrogen and...Ch. 9 - Conceptual APPLY 9.4 The following reaction has E...Ch. 9 - Use the following thermo chemical equation to...Ch. 9 - APPLY 9.6 Approximately, 1.8106 kJ of energy is...Ch. 9 - Classify the following reaction as end o- or ex...Ch. 9 - APPLY 9.8 Instant hot packs and cold packs contain...Ch. 9 - PRACTICE 9.9 What is the specific heat of lead in...Ch. 9 - APPLY 9.10 Calculate the heat capacity (C) of a...
Ch. 9 - PRACTICE 9.11 When 25.0 mL of 1.0 M H2SO4 is added...Ch. 9 - Prob. 9.12ACh. 9 - When 1.00 g of toluene, C7H8, is burned in a bomb...Ch. 9 - Prob. 9.14PCh. 9 - Water gas is the name for the mixture of CO and H2...Ch. 9 - Prob. 9.16ACh. 9 - Prob. 9.17PCh. 9 - Prob. 9.18ACh. 9 - Use the data in Table 9.3 to calculate an...Ch. 9 - Benzene ( C6H6 ) has two resonance structures,...Ch. 9 - Prob. 9.21PCh. 9 - Prob. 9.22ACh. 9 - Prob. 9.23PCh. 9 - Prob. 9.24ACh. 9 - Prob. 9.25PCh. 9 - Prob. 9.26ACh. 9 - Prob. 9.27PCh. 9 - Prob. 9.28PCh. 9 - Prob. 9.29PCh. 9 - Prob. 9.30PCh. 9 - Prob. 9.31PCh. 9 - Prob. 9.32PCh. 9 - PROBLEM 9.33 A 12.0 gallon hold 39.9 kg of...Ch. 9 - A piece of dry ice (solid CO2) is placed inside a...Ch. 9 - Imagine a reaction that results in a change in...Ch. 9 - 9.32 Redraw the following diagram to represent the...Ch. 9 - 9.33 A reaction is carried out in a cylinder...Ch. 9 - Prob. 9.38CPCh. 9 - Prob. 9.39CPCh. 9 - Prob. 9.40CPCh. 9 - Prob. 9.41CPCh. 9 - Prob. 9.42CPCh. 9 - Prob. 9.43CPCh. 9 - What is the difference between heat and...Ch. 9 - What is internal energy?Ch. 9 - Prob. 9.46SPCh. 9 - Prob. 9.47SPCh. 9 - Which of the following are state functions, and...Ch. 9 - Prob. 9.49SPCh. 9 - Calculate the work done in joules by a chemical...Ch. 9 - The addition of H2 to C=C double bonds is an...Ch. 9 - Prob. 9.52SPCh. 9 - Prob. 9.53SPCh. 9 - 9.50 A reaction inside a cylindrical container...Ch. 9 - At a constant pressure of 0.905 atm, a chemical...Ch. 9 - RANWhen a sample of a hydrocarbon fuel is ignited...Ch. 9 - Used in welding metals, the reaction of acetylene...Ch. 9 - What is the difference between the internal energy...Ch. 9 - Prob. 9.59SPCh. 9 - The explosion of 2.00 mol of solid trinitrotoluene...Ch. 9 - The reaction between hydrogen and oxygen to yield...Ch. 9 - The enthalpy change for the reaction of 50.0 mL of...Ch. 9 - Assume that a particular reaction evolves 244 kJ...Ch. 9 - What is the enthalpy change ( H ) for a reaction...Ch. 9 - Prob. 9.65SPCh. 9 - Indicate the direction of heat transfer between...Ch. 9 - Indicate the direction of heat transfer between...Ch. 9 - The familiar "ether" used as an anesthetic agent...Ch. 9 - How much energy in kilojoules is required to...Ch. 9 - Aluminum metal reacts with chlorine with a...Ch. 9 - How much heat in kilojoules is evolved or absorbed...Ch. 9 - How much heat in kilojoules is evolved or absorbed...Ch. 9 - Nitromethane sometimes used as a fuel in drag...Ch. 9 - Prob. 9.74SPCh. 9 - How much heat in kilojoules is evolved or absorbed...Ch. 9 - Prob. 9.76SPCh. 9 - Prob. 9.77SPCh. 9 - Sodium metal is sometimes used as a cooling agent...Ch. 9 - Prob. 9.79SPCh. 9 - Assuming that Coca-Cola has the same specific heat...Ch. 9 - Calculate the amount of heat required to raise the...Ch. 9 - Instant cold packs used to treat athletic injuries...Ch. 9 - Instant hot packs contain a solid and a pouch of...Ch. 9 - When 1.045 g of CaO is added to 50.0 mL of water...Ch. 9 - When a solution containing 8.00 g of NaOH in 50.0...Ch. 9 - When 0.187 g of benzene, C6H6 , is burned in a...Ch. 9 - When 0.500 g of ethanol, C2H6O, is burned in a...Ch. 9 - When 1.50 g of magnesium metal is allowed to react...Ch. 9 - A 110.0 g piece of molybdenum metal is heated to...Ch. 9 - Citric acid has three dissociable hydrogens. When...Ch. 9 - Prob. 9.91SPCh. 9 - Prob. 9.92SPCh. 9 - Prob. 9.93SPCh. 9 - The industrial degreasing solvent methylene...Ch. 9 - Hess's law can be used to calculate reaction...Ch. 9 - Find H in kilojoules for the reaction of nitric...Ch. 9 - Set up a Hess's law cycle, and use the following...Ch. 9 - Prob. 9.98SPCh. 9 - Prob. 9.99SPCh. 9 - What phase of matter is associated with the...Ch. 9 - What is the phase of the standard states of the...Ch. 9 - Write balanced equations for the formation of the...Ch. 9 - Prob. 9.103SPCh. 9 - Prob. 9.104SPCh. 9 - Prob. 9.105SPCh. 9 - The standard enthalpy change for the reaction of...Ch. 9 - Prob. 9.107SPCh. 9 - Styrene ( C8H8 ), the precursor of polystyrene...Ch. 9 - Prob. 9.109SPCh. 9 - Prob. 9.110SPCh. 9 - Prob. 9.111SPCh. 9 - Prob. 9.112SPCh. 9 - Prob. 9.113SPCh. 9 - Prob. 9.114SPCh. 9 - Prob. 9.115SPCh. 9 - Prob. 9.116SPCh. 9 - Isooctane, C8H18, is the component of gasoline...Ch. 9 - Calculate an approximate heat of combustion for...Ch. 9 - Use the data in Table 9.3 to calculate an...Ch. 9 - Use the average bond dissociation energies in...Ch. 9 - Use the bond dissociation energies in Table 9.3 to...Ch. 9 - Prob. 9.122SPCh. 9 - Prob. 9.123SPCh. 9 - Prob. 9.124SPCh. 9 - Prob. 9.125SPCh. 9 - Tell whether the entropy changes for the following...Ch. 9 - Prob. 9.127SPCh. 9 - Prob. 9.128SPCh. 9 - Prob. 9.129SPCh. 9 - Prob. 9.130SPCh. 9 - Prob. 9.131SPCh. 9 - Tell whether reactions with the following values...Ch. 9 - Tell whether reactions with the following values...Ch. 9 - Suppose that a reaction has H=33kJ and S=58J/K. At...Ch. 9 - Suppose that a reaction has H=+41kJ and S=27J/K....Ch. 9 - Which of the reactions (a)-(d) in Problem 9.132...Ch. 9 - Vinyl chloride (H2C=CHCI), the starting material...Ch. 9 - Ethyl alcohol has Hfusion=5.02kJ/mol and melts at...Ch. 9 - Chloroform has Hvaporization=29.2kJ/mol and boils...Ch. 9 - The boiling point of a substance is defined as the...Ch. 9 - What is the melting point of benzene in kelvin if...Ch. 9 - Prob. 9.142SPCh. 9 - Methanol (CH3OH) is made industrially in two steps...Ch. 9 - Ethyl chloride ( C2H5CI ), a substance used as a...Ch. 9 - Prob. 9.145MPCh. 9 - For a process to be spontaneous, the total entropy...Ch. 9 - Prob. 9.147MPCh. 9 - Prob. 9.148MPCh. 9 - Prob. 9.149MPCh. 9 - Prob. 9.150MPCh. 9 - Phosgene, COCI2(g), is a toxic gas used as an...Ch. 9 - Prob. 9.152MPCh. 9 - Write a balanced equation for the reaction of...Ch. 9 - Hydrazine, a component of rocket fuel, undergoes...Ch. 9 - Prob. 9.155MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Calculating standard reaction free energy from standard reduction... Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free energy AG° for the following redox reaction. Be sure your answer has the correct number of significant digits. 3Cu+ (aq) + Cro²¯ (aq) +4H₂O (1) → 3Cu²+ (aq) +Cr(OH)3 (s)+5OH˜¯ (aq) 0 kJ ☐ x10 00. 18 Ararrow_forwardCalculating the pH of a weak base titrated with a strong acid An analytical chemist is titrating 241.7 mL of a 0.4900M solution of methylamine (CH3NH2) with a 0.7800M solution of HNO3. The pK of methylamine is 3.36. Calculate the pH of the base solution after the chemist has added 17.7 mL of the HNO3 solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HNO3 solution added. Round your answer to 2 decimal places. pH = ☑ ? 18 Ararrow_forwardThe following is two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 223.4 148.7 353.7 278.2 334.6 268.7 305.6 234.4 340.0 262.7 304.3 283.2 244.7 143.6 QUESTION: For both groups of data calculate the answers attached in the image.arrow_forward
- The following is a two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 340.0mmol/L 262.7mmol/L QUESTION: For both groups (Regular & Salt Reduced tomato sauce) of data provide answers to the following calculations below: 1. Standard Deviation (Sx) 2. T Values (t0.05,4) 3. 95% Confidence Interval (mmol/L) 4. [Na+] (mg/100 mL) 5. 95% Confidence Interval (mg/100 mL)arrow_forwardIf we have leucine (2-amino-4-methylpentanoic acid), alanine (2-aminopropanoic acid) and phenylalanine (2-amino-3-phenylpropanoic acid), indicate the tripeptides that can be formed (use the abbreviated symbols Leu., Ala and Phe).arrow_forwardBriefly state why trifluoroacetic acid is more acidic than acetic acid.arrow_forward
- Explain why acid chlorides are more reactive than amides in reactions with nucleophiles.arrow_forwardCalculating the pH of a weak base titrated with a strong acid An analytical chemist is titrating 101.7 mL of a 0.3500M solution of piperidine (C5H10NH) with a 0.05700M solution of HClO4. The pK of piperidine is 2.89. Calculate the pH of the base solution after the chemist has added 682.9 mL of the HClO solution to it. 4 Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HClO solution added. 4 Round your answer to 2 decimal places. pH = .11 00. 18 Ararrow_forwardThe following is a two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 340.0 262.7 QUESTION: For both groups of data provide answers to the calculations attached in the imagearrow_forward
- 7. Concentration and uncertainty in the estimate of concentration (class data) Class mean for sample (Regular) |[Cl-] (mmol/L) class mean Sn za/2 95% Confidence Interval (mmol/L) [Na+] (mg/100 mL) 95% Confidence Interval (mg/100 mL)arrow_forwardThe following is a two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 223.4 148.7 353.7 278.2 334.6 268.7 305.6 234.4 340.0 262.7 304.3 283.2 244.7 143.6 QUESTION: For both groups of data calculate the answers attached in the image.arrow_forwardGive reason(s) for six from the followings [using equations if possible] a. Addition of sodium carbonate to sulfanilic acid in the Methyl Orange preparation. b. What happened if the diazotization reaction gets warmed up by mistake. c. Addition of sodium nitrite in acidified solution in MO preparation through the diazotization d. Using sodium dithionite dihydrate in the second step for Luminol preparation. e. In nitroaniline preparation, addition of the acid mixture (nitric acid and sulfuric acid) to the product of step I. f. What is the main reason of the acylation step in nitroaniline preparation g. Heating under reflux. h. Fusion of an organic compound with sodium. HAND WRITTEN PLEASEarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Calorimetry Concept, Examples and Thermochemistry | How to Pass Chemistry; Author: Melissa Maribel;https://www.youtube.com/watch?v=nSh29lUGj00;License: Standard YouTube License, CC-BY