
Name each compound depicted in the ball-and-stick models.
a. 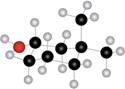 b.
b. 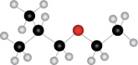 c.
c. 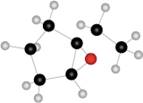
(a)
Interpretation: The name of given compound depicted in the ball-and-stick model is to be stated.
Concept introduction: One should follow the given steps to give the IUPAC name of cyclic alcohol. The first step is naming of ring that contains the carbon bonded to the hydroxyl group. While naming, the -e ending of parent cycloalkane must changed to the suffix -ol. The second step is numbering of carbon chain by providing lowest number to the
Answer to Problem 36P
The name of the given compound depicted in the ball-and-stick model is
Explanation of Solution
The given structure of alcohol is in the form of ball-and-stick model. It is converted into skeletal structure by replacing black ball with
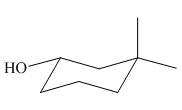
Figure 1
One should follow the given steps to give the IUPAC name of cyclic alcohol. The first step is naming of ring that contains the carbon bonded to the hydroxyl group. While naming, the -e ending of parent cycloalkane must changed to the suffix -ol. The second step is numbering of carbon chain by providing lowest number to the
The above structure of cyclic alcohol shows that it consists of
Thus, the name of the given compound depicted in the ball-and-stick model is
The name of the given compound depicted in the ball-and-stick model is
(b)
Interpretation: The name of given compound depicted in the ball-and-stick model is to be stated.
Concept introduction: One should follow the given steps to give the IUPAC name of ether. The first step is naming of longest chain as an alkane and the shorter chain as an alkoxy group. The second step is applying the other nomenclature rules.
Answer to Problem 36P
The name of the given compound depicted in the ball-and-stick model is
Explanation of Solution
The given structure of ether is in the form of ball-and-stick model. It is converted into skeletal structure by replacing black ball with
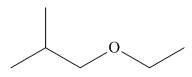
Figure 2
One should follow the given steps to give the IUPAC name of ether. The first step is naming of longest chain as an alkane and the shorter chain as an alkoxy group. The second step is applying the other nomenclature rules.
The above structure of ether shows that it consists of
Thus, the name of the given compound depicted in the ball-and-stick model is
The name of the given compound depicted in the ball-and-stick model is
(c)
Interpretation: The name of given compound depicted in the ball-and-stick model is to be stated.
Concept introduction: One should follow the given steps to give the IUPAC name of an epoxyalkane. The first step is naming of alkane chain or ring to which oxygen atom is bonded, and using the epoxy (prefix) to name epoxide. The second step is designating the location of the atoms (using two numbers) to which the oxygen atom is bonded.
Answer to Problem 36P
The name of the given compound depicted in the ball-and-stick model is
Explanation of Solution
The given structure of epoxide is in the form of ball-and-stick model. It is converted into skeletal structure by replacing black ball with
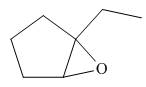
Figure 3
One should follow the given steps to give the IUPAC name of an epoxyalkane. The first step is naming of alkane chain or ring to which oxygen atom is bonded, and using the epoxy (prefix) to name epoxide. The second step is designating the location of the atoms (using two numbers) to which the oxygen atom is bonded.
The above structure of epoxide shows that the cyclopentane ring is bonded to the epoxide ring on
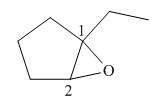
Figure 4
Thus, the name of the given compound depicted in the ball-and-stick model is
The name of the given compound depicted in the ball-and-stick model is
Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry (6th Edition)
Additional Science Textbook Solutions
Microbiology: An Introduction
Physical Science
Biochemistry: Concepts and Connections (2nd Edition)
Human Biology: Concepts and Current Issues (8th Edition)
Fundamentals Of Thermodynamics
- Why is analysing salt content (using Mohr titration) in both regular & salt reduced tomato sauce important?arrow_forwardIn the image below, correctly name the glassware # _P ( Blank 1) and T ( Blank 2). 景 A W Blank # 1 Blank #2 1000 +19 E E D 0 0-0 G H A A K Π 12 R M N S 0-0-arrow_forwardFeedback: Your answer is incorrect. Predict the major products of the following organic reaction: CN Δ + A ? NC Some important notes: • Draw the major product, or products, of the reaction in the drawing area below. • If there aren't any products, because no reaction will take place, check the box below the drawing area instead. • Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers. esc Check 80 MH F1 F2 F3 F4 F5 50 @ # C % 95 € Save For Later Sub 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy C A DII F6 F7 F8 7 * 8 Λ & 6 F9 F10 9 0 4arrow_forward
- Incorrect Feedback: Your answer is incorrect. Predict the major products of the following organic reaction: ཤིགས་བྱ རྩ་ཅད་ཀྱིས་༢༩ + Some important notes: A ^ ? • Draw the major product, or products, of the reaction in the drawing area below. • If there aren't any products, because no reaction will take place, check the box below the drawing area instead. • Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers. E Check 0 لا Save For La ©2025 McGraw Hill LLC. All Rights Reserved. Terms of All F9 Aarrow_forwardPredict the major products of the following organic reaction: + Δ A ? Some important notes: • Draw the major product, or products, of the reaction in the drawing area below. • If there aren't any products, because no reaction will take place, check the box below the drawing area instead. • Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers. Explanation Check Click and drag to start drawing a structure. 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use Privaarrow_forwardesc 2 Incorrect Feedback: Your answer is incorrect. Can the molecule on the right-hand side of this organic reaction be made in good yield from no more than two reactants, in one step, by moderately heating the reactants? ? A O • If your answer is yes, then draw the reactant or reactants in the drawing area below. You can draw the reactants in any arrangement you like. . If your answer is no, check the box under the drawing area instead. Check F1 ! @ X C Save For Later Submit Assignment 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility 80 et A ད 1 4 F2 F3 F4 F5 F6 F7 F8 F9 F10 F11 F12 # $ 45 % A 6 87 & * 8 9 ) 0 + ||arrow_forward
- Can the molecule on the right-hand side of this organic reaction be made in good yield from no more than two reactants, in one step, by moderately heating the reactants? ?A Δ O • If your answer is yes, then draw the reactant or reactants in the drawing area below. You can draw the reactants in any arrangement you like. • If your answer is no, check the box under the drawing area instead. Explanation Check Click and drag to start drawing a structure. 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibilit ku F11arrow_forward१ eq ine teaching and × + rn/takeAssignment/takeCovalentActivity.do?locator-assignment-take [Review Topics] [References] Write an acceptable IUPAC name for the compound below. (Only systematic names, not common names are accepted by this question.) Keep the information page open for feedback reference. The IUPAC name is In progress mit Answer Retry Entire Group 5 more group attempts remaining Cengage Learning | Cengage Technical Support Save and Exitarrow_forwardDraw the molecules.arrow_forward
- Draw the mechanism for the acid-catalyzed dehydration of 2-methyl-hexan-2-ol with arrows please.arrow_forward. Draw the products for addition reactions (label as major or minor) of the reaction between 2-methyl-2-butene and with following reactants : Steps to follow : A. These are addition reactions you need to break a double bond and make two products if possible. B. As of Markovnikov rule the hydrogen should go to that double bond carbon which has more hydrogen to make stable products or major product. Here is the link for additional help : https://study.com/academy/answer/predict-the-major-and-minor-products-of-2-methyl- 2-butene-with-hbr-as-an-electrophilic-addition-reaction-include-the-intermediate- reactions.html H₂C CH3 H H3C CH3 2-methyl-2-butene CH3 Same structure CH3 IENCESarrow_forwardDraw everything on a piece of paper including every single step and each name provided using carbons less than 3 please.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





