
Chemistry in Context
8th Edition
ISBN: 9780073522975
Author: American Chemical Society
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 8, Problem 6Q
Consider the galvanic cell pictured. A coating of impure silver metal begins to appear on the surface of the silver electrode as the cell discharges.
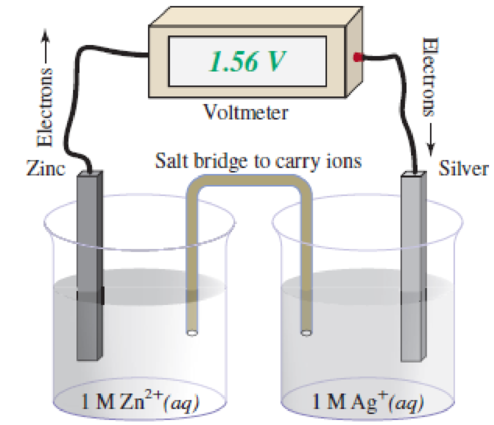
- a. Identify the anode and write the oxidation half-reaction.
- b. Identify the cathode and write the reduction half-reaction.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Try: Convert the given 3D perspective structure to Newman projection about C2 - C3 bond (C2 carbon in the
front). Also, show Newman projection of other possible staggered conformers and circle the most stable
conformation. Use the template shown.
F
H3C
Br
H
None
16. Consider the probability distribution p(x) = ax", 0 ≤ x ≤ 1 for a positive integer n.
A. Derive an expression for the constant a, to normalize p(x).
B. Compute the average (x) as a function of n.
C. Compute σ2 = (x²) - (x)², the variance of x, as a function of n.
Chapter 8 Solutions
Chemistry in Context
Ch. 8.1 - Prob. 8.3YTCh. 8.1 - Prob. 8.4YTCh. 8.3 - Prob. 8.9CTCh. 8.4 - Prob. 8.10CTCh. 8.4 - Prob. 8.11SCCh. 8.5 - Prob. 8.13SCCh. 8.5 - Prob. 8.14CTCh. 8.6 - Prob. 8.16CTCh. 8.7 - Prob. 8.17CTCh. 8.7 - Prob. 8.18YT
Ch. 8.7 - Prob. 8.21CTCh. 8.8 - Prob. 8.23CTCh. 8 - Prob. 1QCh. 8 - Prob. 2QCh. 8 - Which chemical species gets oxidized and which...Ch. 8 - What is the difference between a galvanic cell and...Ch. 8 - Two common units associated with electricity are...Ch. 8 - Consider the galvanic cell pictured. A coating of...Ch. 8 - Prob. 7QCh. 8 - a. How does the voltage from a tiny AAA alkaline...Ch. 8 - Prob. 9QCh. 8 - The mercury battery has been used extensively in...Ch. 8 - Prob. 12QCh. 8 - During the conversion of O2(g) to H2O(l) in a fuel...Ch. 8 - How does the reaction between hydrogen and oxygen...Ch. 8 - This diagram represents the hydrogen fuel cell...Ch. 8 - What is a PEM fuel cell? How does it differ from...Ch. 8 - In addition to hydrogen, methane also has been...Ch. 8 - Prob. 18QCh. 8 - Potassium and lithium both are reactive Group 1...Ch. 8 - What challenges keep hydrogen fuel cells from...Ch. 8 - Every year, 5.6 1021 kJ of energy comes to Earth...Ch. 8 - Prob. 22QCh. 8 - Prob. 23QCh. 8 - Prob. 24QCh. 8 - Prob. 25QCh. 8 - Consider these three sources of light: a candle, a...Ch. 8 - Prob. 27QCh. 8 - What is the difference between an electrolytic...Ch. 8 - Provide some differences between a leadacid...Ch. 8 - Prob. 30QCh. 8 - The company ZPower is promoting its silverzinc...Ch. 8 - Prob. 32QCh. 8 - Prob. 33QCh. 8 - You never need to plug in Toyotas gasolinebattery...Ch. 8 - Prob. 35QCh. 8 - What is the tragedy of the commons? How does this...Ch. 8 - Hydrogen is considered an environmentally friendly...Ch. 8 - Fuel cells were invented in 1839 but never...Ch. 8 - Hydrogen and methane both can react with oxygen in...Ch. 8 - Engineers have developed a prototype fuel cell...Ch. 8 - How can the principles of green chemistry be...Ch. 8 - Prob. 42QCh. 8 - Prob. 43QCh. 8 - Small quantities of hydrogen gas can be prepared...Ch. 8 - Prob. 45QCh. 8 - Prob. 46QCh. 8 - Prob. 47QCh. 8 - Prob. 48QCh. 8 - Although Alessandro Volta is credited with the...Ch. 8 - Prob. 50QCh. 8 - If all of todays technology presently based on...Ch. 8 - Prob. 52QCh. 8 - Prob. 53QCh. 8 - Prob. 54QCh. 8 - Prob. 55Q
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 451. Use the diffusion model from lecture that showed the likelihood of mixing occurring in a lattice model with eight lattice sites: Case Left Right A B C Permeable Barrier → and show that with 2V lattice sites on each side of the permeable barrier and a total of 2V white particles and 2V black particles, that perfect de-mixing (all one color on each side of the barrier) becomes increasingly unlikely as V increases.arrow_forward46. Consider an ideal gas that occupies 2.50 dm³ at a pressure of 3.00 bar. If the gas is compressed isothermally at a constant external pressure so that the final volume is 0.500 dm³, calculate the smallest value Rest can have. Calculate the work involved using this value of Rext.arrow_forwardNonearrow_forward
- 2010. Suppose that a 10 kg mass of iron at 20 C is dropped from a heigh of 100 meters. What is the kinetics energy of the mass just before it hits the ground, assuming no air resistance? What is its speed? What would be the final temperature of the mass if all the kinetic energy at impact is transformed into internal energy? The molar heat capacity of iron is Cpp = 25.1J mol-¹ K-1 and the gravitational acceleration constant is 9.8 m s¯² |arrow_forwardell last during 7. Write the isotopes and their % abundance of isotopes of i) Cl ii) Br 8. Circle all the molecules that show Molecular ion peak as an odd number? c) NH2CH2CH2NH2 d) C6H5NH2 a) CH³CN b) CH3OHarrow_forwardCalsulate specific heat Dissolution of NaOH ก ง ง Mass of water in cup Final temp. of water + NaOH Initial temp. of water AT Water AH Dissolution NaOH - "CaicuraORT. AH (NaOH)=-AH( 30g (water) 29.0°C 210°C 8°C (82) 100 3.. =1003.20 Conjosarrow_forward
- Please provide throrough analysis to apply into further problems.arrow_forwardMolecular ion peak: the peak corresponding to the intact morecure (with a positive charge) 4. What would the base peak and Molecular ion peaks when isobutane is subjected to Mass spectrometry? Draw the structures and write the molecular weights of the fragments. 5. Circle most stable cation a) tert-butyl cation b) Isopropyl cation c) Ethyl cation. d)Methyl cationarrow_forwardHow many arrangements are there of 15 indistinguishable lattice gas particles distributed on: a.V = 15 sites b.V = 16 sites c.V = 20 sitesarrow_forward
- For which element is the 3d subshell higher in energy than that 4s subshell? Group of answer choices Zr Ca V Niarrow_forwardii) Molecular ion peak :the peak corresponding to the intact molecule (with a positive charge) What would the base peak and Molecular ion peaks when isobutane is subjected to Mass spectrometry? Draw the structures and write the molecular weights of the fragments. Circle most stable cation a) tert-butyl cation b) Isopropyl cation c) Ethyl cation. d) Methyl cation 6. What does a loss of 15 represent in Mass spectrum? a fragment of the molecule with a mass of 15 atomic mass units has been lost during the ionization Process 7. Write the isotopes and their % abundance of isotopes of i) Clarrow_forwardChoose a number and match the atomic number to your element on the periodic table. For your element, write each of these features on a side of your figure. 1. Element Name and symbol 2. Family and group 3. What is it used for? 4. Sketch the Valence electron orbital 5. What ions formed. What is it's block on the periodic table. 6. Common compounds 7. Atomic number 8. Mass number 9. Number of neutrons- (show calculations) 10. Sketch the spectral display of the element 11.Properties 12. Electron configuration 13. Submit a video of a 3-meter toss in slow-moarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning  ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781285199030
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Introduction to Electrochemistry; Author: Tyler DeWitt;https://www.youtube.com/watch?v=teTkvUtW4SA;License: Standard YouTube License, CC-BY