
Chemistry for Engineering Students
3rd Edition
ISBN: 9781285199023
Author: Lawrence S. Brown, Tom Holme
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7, Problem 7.93PAE
7.97 Consider the structure shown below for 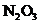 as well as any other important resonance structures.
as well as any other important resonance structures.
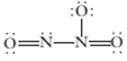
(a) What is the expected O—N—O bond angle in this structure? (b) The 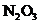 molecule contains N—O bonds of two different lengths. How many sborter N—O bonds would be present?
molecule contains N—O bonds of two different lengths. How many sborter N—O bonds would be present?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
personality of each of them in terms of nucleophile vs. electrophile (some can be considered
acids/bases but we are not looking at that here). Note you may have to use your growing intuition to
figure out the personality of one of the molecules below but I believe in you! Rationalize it out
based on what we have called strong versus weak electrophiles in past mechanisms. Consider using
the memes below to help guide your understanding!
A
OH
O
B
CH3
C
Molecule A: [Select]
Molecule B: [Select]
Molecule C: [Select]
Molecule D: [Select]
>
H
D
OH
4) Which oxygen atom in the structure below is most basic / nucleophilic? Please explain by
discussing the electron density around each oxygen atom. Show at least three resonance
structures for the compound.
оого
Can you show me this problem. Turn them into lewis dot structures for me please and then answer the question because I cant seem to comprehend it/ The diagrams on the picture look too small I guess.
Chapter 7 Solutions
Chemistry for Engineering Students
Ch. 7 - List some factors influencing the biocompatibility...Ch. 7 - • use electron configurations to explain why...Ch. 7 - • describe die energy changes in the formation of...Ch. 7 - • define electronegativity and state how...Ch. 7 - • identify or predict polar, nonpolar, and ionic...Ch. 7 - • write Lewis electron structures for molecules or...Ch. 7 - • describe chemical bonding using a model based on...Ch. 7 - • explain how hybridization reconciles observed...Ch. 7 - • predict the geometry of a molecule from its,...Ch. 7 - • use models (real or software) to help visualize...
Ch. 7 - • explain the formation of multiple bonds in terms...Ch. 7 - • identify sigma and pi bonds in a molecule and...Ch. 7 - Define the term biocompatibility.Ch. 7 - List some properties associated with biomaterials...Ch. 7 - Prob. 7.3PAECh. 7 - Prob. 7.4PAECh. 7 - Prob. 7.5PAECh. 7 - Prob. 7.6PAECh. 7 - Why is the ion not found in nature?Ch. 7 - Why do nonmetals tend to form anions rather than...Ch. 7 - Prob. 7.9PAECh. 7 - 7.10 Arrange the members of each of the following...Ch. 7 - 7.11 Arrange the following sets of anions in order...Ch. 7 - 7.12 Which pair will form a compound with the...Ch. 7 - In a lattice, a positive ion is often surrounded...Ch. 7 - Use the concept of lattice energy to rationalize...Ch. 7 - 7.13 Figure 7-2 depicts the interactions of an ion...Ch. 7 - Mat type of bond is likely to form between one...Ch. 7 - 7.14 Describe the difference between a covalent...Ch. 7 - Prob. 7.18PAECh. 7 - Sketch a graph of the potential energy of two...Ch. 7 - Prob. 7.20PAECh. 7 - 7.17 Coulombic forces are often used to explain...Ch. 7 - 7.18 In terms of the strengths of the covalent...Ch. 7 - 7.19 If the formation of chemical bonds always...Ch. 7 - 7.20 Draw the Lewis dot symbol for each of the...Ch. 7 - 7.21 Theoretical models for the structure of...Ch. 7 - 7.22 Use Lewis dot symbols to explain why chlorine...Ch. 7 - 7.23 Define the term lone pair.Ch. 7 - 7.24 How many electrons are shared between two...Ch. 7 - 7.25 How does the bond energy of a double bond...Ch. 7 - 7.26 How is electronegativity defined?Ch. 7 - 7.27 Distinguish between electron affinity and...Ch. 7 - 7.28 Certain elements in the periodic table shown...Ch. 7 - 7.29 When two atoms with different...Ch. 7 - 7.30 The bond in HF is said to be polar, with the...Ch. 7 - 7.31 Why is a bond between two atoms with...Ch. 7 - Prob. 7.36PAECh. 7 - 7.33 In each group of three bonds, which bond is...Ch. 7 - Prob. 7.38PAECh. 7 - Prob. 7.39PAECh. 7 - 7.35 Which one of the following contains botb...Ch. 7 - Prob. 7.41PAECh. 7 - Prob. 7.42PAECh. 7 - 7.37 Draw the Lewis structure for each of the...Ch. 7 - 7.38 Draw a Lewis structure for each of the...Ch. 7 - Prob. 7.45PAECh. 7 - 7.40 Why is it impossible for hydrogen to be the...Ch. 7 - Prob. 7.47PAECh. 7 - 7.42 Draw resonance structure for (a) (b) and (c)Ch. 7 - Prob. 7.49PAECh. 7 - Prob. 7.50PAECh. 7 - Prob. 7.51PAECh. 7 - 7.46 Consider the nitrogen-oxygen bond lengths in...Ch. 7 - 7.47 Which of the species listed has a Lewis...Ch. 7 - 7.48 Identify what is incorrect in the Lewis...Ch. 7 - 7.49 Identify what is incorrect in the Lewis...Ch. 7 - 7.50 Chemical species are said to be isoelectronic...Ch. 7 - 7.51 Explain the concept of wave interference in...Ch. 7 - Distinguish between constructive and destructive...Ch. 7 - How is the concept of orbital overlap related to...Ch. 7 - 7.52 How does orbital overlap explain the buildup...Ch. 7 - 7.53 How do sigma and pi bonds differ? How are...Ch. 7 - 7.54 CO , CO2 , CH3OH , and CO32 , all contain...Ch. 7 - 7.55 Draw the Lewis dot structure of the following...Ch. 7 - 7.56 Draw the Lewis dot structures of the...Ch. 7 - 7.57 What observation about molecules compels us...Ch. 7 - Prob. 7.66PAECh. 7 - 7.59 What type of hybrid orbital is generated by...Ch. 7 - Considering only s and p atomic orbitals, list all...Ch. 7 - 7.61 What hybrid orbitals would be expected for...Ch. 7 - 7.62 What type of hybridization would you expect...Ch. 7 - 7.63 What physical concept forms the premise of...Ch. 7 - 7.64 Predict the geometry of the following...Ch. 7 - Prob. 7.73PAECh. 7 - Prob. 7.74PAECh. 7 - Prob. 7.75PAECh. 7 - 7.68 Give approximate values for the indicated...Ch. 7 - 7.69 Propene has the chemical formula Describe the...Ch. 7 - Prob. 7.78PAECh. 7 - Describe what happens to the shape about the...Ch. 7 - Prob. 7.80PAECh. 7 - Prob. 7.81PAECh. 7 - 7.72 How does an MSN differ from amorphous silica...Ch. 7 - Prob. 7.83PAECh. 7 - Prob. 7.84PAECh. 7 - Prob. 7.85PAECh. 7 - Prob. 7.86PAECh. 7 - 7.91 A Lewis structure for the oxalate ion is...Ch. 7 - Prob. 7.88PAECh. 7 - 7.93 An unknown metal M forms a chloride with the...Ch. 7 - Prob. 7.90PAECh. 7 - Prob. 7.91PAECh. 7 - 7.96 Consider the hydrocarbons whose structures...Ch. 7 - 7.97 Consider the structure shown below for as...Ch. 7 - Prob. 7.94PAECh. 7 - Prob. 7.95PAECh. 7 - Prob. 7.96PAECh. 7 - 7.101 Lead selenide nanocrystals may provide a...Ch. 7 - Prob. 7.98PAECh. 7 - Prob. 7.99PAECh. 7 - 7.104 Hydrogen azide, HN3 , is a liquid that...Ch. 7 - Prob. 7.101PAECh. 7 - Prob. 7.102PAECh. 7 - 7.107 How do the Lewis symbols for C, Si, and Ge...Ch. 7 - Prob. 7.104PAECh. 7 - Prob. 7.105PAE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The fire releases 2.80 x 107 Joules of heat energy for each liter of oil burned. The water starts out at 24.5 °C, raising the water's temperature up to 100 °C, and then raises the temperature of the resulting steam up to 325 °C. How many liters of water will be needed to absorb the heat from the fire in this way, for each 1.0 liter of crude oil burned? 4186 J/(kg°C) = heat of water 2020 J/(kg°C) = heat of steam 2,256,000 (i.e. 2.256 x 106) J/kg = latent heat of vaporization for water (at the boiling point of 100 °C).arrow_forward6 Which of the following are likely to be significant resonance structures of a resonance hybrid? Draw another resonance structure for each of the compounds you select as being a resonance form. (A Br: Br: A B C D Earrow_forwardWrite the systematic (IUPAC) name for the following organic molecules. Note for advanced students: you do not need to include any E or Z prefixes in your names. Br structure Br Br Oweuarrow_forward
- Conservation of mass was discussed in the background. Describe how conservation of mass (actual, not theoretical) could be checked in the experiment performed.arrow_forwardWhat impact would adding twice as much Na2CO3 than required for stoichiometric quantities have on the quantity of product produced? Initial results attachedarrow_forwardGiven that a theoretical yield for isolating Calcium Carbonate in this experiment would be 100%. From that information and based on the results you obtained in this experiment, describe your success in the recovery of calcium carbonate and suggest two possible sources of error that would have caused you to not obtain 100% yield. Results are attached form experimentarrow_forward
- 5) Calculate the flux of oxygen between the ocean and the atmosphere(2 pts), given that: (from Box 5.1, pg. 88 of your text): Temp = 18°C Salinity = 35 ppt Density = 1025 kg/m3 Oxygen concentration measured in bulk water = 263.84 mmol/m3 Wind speed = 7.4 m/s Oxygen is observed to be about 10% initially supersaturated What is flux if the temperature is 10°C ? (2 pts) (Hint: use the same density in your calculations). Why do your calculated values make sense (or not) based on what you know about the relationship between gas solubility and temperature (1 pt)?arrow_forwardFind a molecular formula for these unknownsarrow_forward(ME EX2) Prblms 8-11 Can you please explain problems 8 -11 to me in detail, step by step? Thank you so much! If needed color code them for me.arrow_forward
- Don't used hand raitingarrow_forwardThe following 'H NMR spectrum was taken with a 750 MHz spectrometer: 1.0 0.5 0.0 10.0 9.0 8.0 7.0 6.0 5.0 4.0 3.0 ' 2.0 1.0 0.0 (ppm) What is the difference Av in the frequency of RF ac Δν ac radiation absorbed by the a and c protons? (Note: it's not equal to the difference in chemical shifts.) Round your answer to 2 significant digits, and be sure it has an appropriate unit symbol. = O O a will shift left, c will shift right. O a will shift right, c will shift left. a and c will both shift left, with more space between them. Suppose a new spectrum is taken with a 500 MHz spectrometer. What will be true about this new spectrum? O a and c will both shift left, with less space between them. O a and c will both shift right, with more space between them. O a and c will both shift right, with less space between them. Which protons have the largest energy gap between spin up and spin down states? O None of the above. ○ a Ob Explanation Check C Ar B 2025 McGraw Hill LLC. All Rights Reserved.…arrow_forwardWhat mass of Na2CO3 must you add to 125g of water to prepare 0.200 m Na2CO3? Calculate mole fraction of Na2CO3, mass percent, and molarity of the resulting solution. MM (g/mol): Na2CO3 105.99; water 18.02. Final solution density is 1.04 g/mL.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY