
Concept explainers
Interpretation:
The IUPAC names of each of the given compounds are to be given by using
Concept introduction:
The priority of groups in nomenclature is assigned according to Cahn-Ingold-Prelog (CIP Rule) convention rules:
The higher the
If priority cannot be assigned according to atomic mass, then assign the priority according to first point of difference.
If both the priority groups are on the same side of double-bonded carbon atom, then it is known as
But, if both the priority groups are diagonal to each other, then it is
Rule for R/S Nomenclature:
4B(+)3B(-)2B(+)1B(-)
4B(+) means if 4th priority group is below then from 1 to 2 to 3 if leads a clockwise rotation then its R and if anticlockwise then it is S.
3B(-) means if 3rd priority group is below then from 1 to 2 to 4 if leads a clockwise rotation then its S and if anticlockwise then it is R.
Answer to Problem 1PP
Solution:
Explanation of Solution
a)
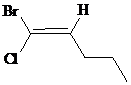
The IUPAC name with
Assign the priority to four groups, –Br gets first priority, pentyl group gets second, –Cl gets third, and –H gets fourth. Since both the priority groups are diagonal to each other, it is
Select the longest carbon chain. The parent hydrocarbon chain is pentane and hence pent- will be used and double bond is at the second position. Therefore, pent-1-ene is used.
Chloro and bromo groups are at first position. So, 1-bromo-1-chloropent-1-ene will be used.
Hence, the IUPAC name of the compound will be
b)
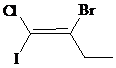
The IUPAC name with
Assign the priority to four groups, –I gets first priority, –Br gets second, –Cl gets third, and ethyl group gets fourth. Since both the priority groups are diagonal to each other, it is
Select the longest carbon chain. The parent hydrocarbon chain is butyl and hence but- will be used and double bond is at the first position. Therefore, but-1-ene is used.
Chloro and iodo groups are at first position and bromo group is at second position. So, 2-bromo-1-chloro-1-iodobut-1-ene will be used.
Hence, the IUPAC name of the compound will be
c)
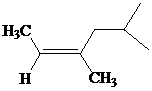
The IUPAC name with
Assign the priority to four groups, 2-methylpropyl gets first priority, both methyl groups get second, and –H gets third. Since both the priority groups are on the same side, it is
Select the longest carbon chain. The parent hydrocarbon chain is hexyl and hence hex- will be used and double bond is at the second position. Therefore, hex-2-ene is used.
There are two methyl groups at third and fifth positions. So, 3,5-dimethylhex-2-ene will be used.
Hence, the IUPAC name of the compound will be
d)
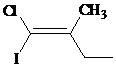
The IUPAC name with
Assign the priority to four groups, –I gets first priority, –Cl gets second, ethyl group gets third, and methyl group gets fourth. Since both the priority groups are on the same side, it is
Select the longest carbon chain. The parent hydrocarbon chain is butyl and hence but- will be used and double bond is at the first position. Therefore, but-1-ene is used.
There is one methyl group at second position, and chloro and iodo groups at first position. So, 1-chloro-1-iodo-2 methylbut-1-ene will be used.
Hence, the IUPAC name of the compound will be
e)
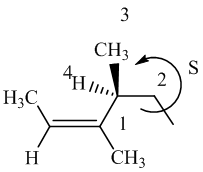
The IUPAC name with
Assign the priority to four groups, 1-methylpropyl gets first priority, both methyl groups get second, and –H gets third. Since both the priority groups are on the same side, it is
Select the longest carbon chain. The parent hydrocarbon chain is hexyl and hence hex- will be used and double bond is at the second position. Therefore, hex-2-ene is used.
There are two methyl groups at third and fourth position. So, 3,4-dimethylhex-2-ene will be used.
Carbon-4 is a chiral carbon. Assigning priority to the groups, 2-butene gets first priority, ethyl gets second, methyl gets third, and hydrogen gets fourth.
Move 1-2-3, it is in anticlockwise direction. So, it is
Hence, the IUPAC name of the compound will be
f)
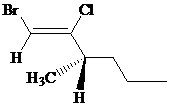
The IUPAC name can be done using the following steps:
Assign the priority to four groups, –Br gets first priority, –Cl gets second, 1-methylbutyl gets third, and –H gets fourth. Since both the priority groups are on the same side, it is
Select the longest carbon chain. The parent hydrocarbon chain is hexyl and hence hex- will be used and double bond is at the first position. Therefore, hex-1-ene is used.
There is one methyl group at third position, and chloro at second and bromo at first position. So, 1-bromo-2-chloro-3-methylhex-1-ene will be used.
Carbon-3 is a chiral carbon. Assigning priority to the groups, 1-bromo-2-chloroethene gets first priority, propyl gets second, methyl gets third, and hydrogen gets fourth.
Rotate the molecule such that –H is at a horizontal position and then move 1-2-3, it is in anticlockwise direction. So, it is
Hence, the IUPAC name of the compound will be
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry, 12e Study Guide/Student Solutions Manual
- Understanding the general acid-base properties of amino acids O Proteins Imagine each of the molecules shown below was found in an aqueous solution. Can you tell whether the solution is acidic, basic, or neutral? molecule The solution is... 010 H3N-CH-C-OH CH HO CH3 O acidic O basic neutral O (unknown) H3N HO 0 O acidic O basic neutral ○ (unknown) H3N-CH-C-O CH2 CH3-CH-CH3 O acidic O basic Oneutral ○ (unknown) O= X H2N-CH-C-O CH3 CH CH3 acidic O basic O neutral ○ (unknown) ? 000arrow_forwardImagine each of the molecules shown below was found in an aqueous solution. Can you tell whether the solution is acidic, basic, or neutral? molecule 0=0 H3N-CH-C-o HO CH2 OH The solution is... O acidic O basic O neutral O (unknown) H₂N acidic O basic O neutral ○ (unknown) + H3N O OH O acidic O basic O neutral O (unknown) H2N-CH-C-O CH3 O acidic O basic neutral ○ (unknown) X ? olo HEarrow_forwardRecognizing ampli Draw an a amino acid with a methyl (-CH3) side chain. Explanation Check Click and drag to start drawing a structure. X Carrow_forward
- Write the systematic name of each organic molecule: structure name × HO OH ☐ OH CI CI O CI OH OHarrow_forwardく Check the box under each a amino acid. If there are no a amino acids at all, check the "none of them" box under the table. Note for advanced students: don't assume every amino acid shown must be found in nature. COO H3N-C-H CH2 HO CH3 NH3 O CH3-CH CH2 OH Onone of them Explanation Check + H3N O 0. O OH + NH3 CH2 CH3-CH H2N C-COOH H O HIC + C=O H3N-C-O CH3- - CH CH2 OH Х 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center Accesarrow_forwardWrite the systematic name of each organic molecule: structure HO-C-CH2-CH3 O -OH CH3-CH2-CH2-CH2-CH2-C-OH CH3 CH3-CH-CH2-C-OH Explanation Check S namearrow_forward
- theres 2 productsarrow_forwardDraw the major product of this solvolysis reaction. Ignore any inorganic byproducts. + CH3CH2OH Drawing Q Atoms, Bonds and Rings OCH2CH3 || OEt Charges OH 00-> | Undo Reset | Br Remove Done Drag To Pan +arrow_forwardDraw the major product of this SN1 reaction. Ignore any inorganic byproducts. CH3CO2Na CH3CO2H Drawing + Br Q Atoms, Bonds and Rings OAC Charges OH ОАс Na ဂ Br Undo Reset Remove Done Drag To Pan +arrow_forward
- Organic Functional Groups entifying positions labeled with Greek letters in acids and derivatives 1/5 ssible, replace an H atom on the a carbon of the molecule in the drawing area with a ce an H atom on the ẞ carbon with a hydroxyl group substituent. ne of the substituents can't be added for any reason, just don't add it. If neither substi er the drawing area. O H OH Oneither substituent can be added. Check D 1 Accessibility ado na witharrow_forwardDifferentiate between electrophilic and nucleophilic groups. Give examples.arrow_forwardAn aldehyde/ketone plus an alcohol gives a hemiacetal, and an excess of alcohol gives an acetal. The reaction is an equilibrium; in aldehydes, it's shifted to the right and in ketones, to the left. Explain.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





