
Biochemistry: Concepts and Connections (2nd Edition)
2nd Edition
ISBN: 9780134641621
Author: Dean R. Appling, Spencer J. Anthony-Cahill, Christopher K. Mathews
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6, Problem 3P
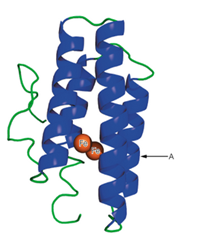
A schematic structure of the subunit of hemerythrin (an oxygen-binding protein from invertebrate animals) is shown to the right.
a. It has been found that in some of the a-helical regions of hemerythrin, about every third or fourth amino acid residue is a hydrophobic one. Suggest a structural reason for this finding.
b. What would be the effect of a mutation that placed a proline residue at point A in the structure?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
2. For the flow of fluid over a flat membrane of length 10cm,
determine the length-average mass transfer coefficient. The
relevant properties of the system are u=0.01cm²/s, D=5 x 106
cm²/s, and v = 5.0 cm/s.
Background
Freezing isn't the only challenge in cryopreservation - thawing can
be just as difficult. A microwave oven seems like a nice solution,
since it deposits energy quickly and microwaves are non-ionizing
radiation (they do not cause DNA mutation). However, water
absorbs microwaves more effectively than ice does, meaning that
the portion of an organ that has already melted will get warmer at a
higher rate than the remaining ice – the opposite of what we want!
-
The transmission of radiation through a weakly absorbing material
such as ice or water can be modeled by Beer's law, which assumes
that the rate of absorption at a depth x is proportional to the local
radiation intensity I(x) times an absorption coefficient, which is often
written as μ or a or just µ). Noting that absorption decreases the
intensity, we can write a differential equation
a
Solving the differential equation with the boundary condition on the
surface being
gives the relationship
For a standard microwave oven…
3. Dry air is inhaled at a rate of 10 liter/min through a trachea with
a diameter of 20 mm and a length of 125 mm. The inner
surface of the trachea is at a normal body temperature of 37°C
and may be assumed to be saturated with water.
a. Assuming steady, fully developed flow in the trachea,
estimate the mass transfer convection coefficient.
b. Estimate the daily water loss (liter/day) associated with
evaporation in the trachea.
Chapter 6 Solutions
Biochemistry: Concepts and Connections (2nd Edition)
Ch. 6 - Prob. 1PCh. 6 - Bovine pancreatic trypsin inhibitor (BPTI; Figure...Ch. 6 - A schematic structure of the subunit of...Ch. 6 - In the protein adenylate kinase, the C-terminal...Ch. 6 - Give two reasons to explain why a proline residue...Ch. 6 - Consider a small protein containing 101 amino acid...Ch. 6 - a. Based on a more conservative answer to Problem...Ch. 6 - The following sequence is part of a globular...Ch. 6 - a. A protein is found to be a tetramer of...Ch. 6 - Under physiological conditions, the protein...
Ch. 6 - Theoretical and experimental measurements show...Ch. 6 - The peptide hormone vasopressin is used in the...Ch. 6 - A protein gives under conditions of buffer...Ch. 6 - A protein gives a single band on SDS get...Ch. 6 - It has been postulated that the normal...Ch. 6 - Below are shown two views of the backbone...Ch. 6 - Do you expect a Pro Gly mutation in a...Ch. 6 - Rank the following in terms of predicted rates...Ch. 6 - Shown below are two cartoon views of the small...Ch. 6 - Prob. 20PCh. 6 - In most cases, mutations in the core of protein...Ch. 6 - A Leu Ala mutation at a site buried the core of...Ch. 6 - Disulfide bonds have been shown to stabilize...Ch. 6 - Cartoon renderings of the proteins Top 7 and adaH2...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- 1. Conceptual questions a. What dimensionless group describes the relative importance of convection versus diffusion. Explain the physical basis of this group. b. For mass transfer from a flowing fluid to a reactive surface, explain how convection increases the flux of solute to the surface.arrow_forwardAssessment +1501 pts /1600 Resources Solution ? Hint Sub bo Each pictured Lewis structure is invalid. Identify the error in each case. O Macmillan Learning :0▬▬0: Answer Bank wrong electron total :0- :F======F: octet-rule violation N :0:arrow_forward[s] mM V (M/s) Uninhibited 0.333 1.65 x 107 1.05 x 107 V (M/s) x 10' Inhibitor A V (M/s) x 107 Inhibitor B 0.794 x 107 0.40 1.86 x 107 1.21 x 107 0.893 x 107 0.50 2.13 x 107 1.43 x 107 1.02 x 107 0.666 2.49 x 107 1.74 x 107 1.19 x 107 1.0 2.99 x 107 2.22 x 107 1.43 x 107 2.0 3.72 x 107 3.08 x 107 1.79 x 107arrow_forward
- For a Michaelis-Menten reaction, k₁-5 x 10'/M-s, k.-2 x 10%/s, and k₂-4 x 10²/s. a) Calculate the Ks and KM for this reaction. b) Does substrate binding achieve equilibrium or steady state?arrow_forwardAssume that an enzyme-catalyzed reaction follows the scheme shown: E+S SES →E + P k₁ = 1 x 109/M-s k-1=2.5 x 10%/s k₂ = 3.4 x 107/s What is the dissociation constant for the enzyme-substrate, K,? What is the Michaelis constant, Km, for this enzyme? What is the turnover number, Keat, for this enzyme? What is the catalytic efficiency for the enzyme? If the initial Et concentration is 0.25mM, what is Vmax?arrow_forwardAn enzyme lowers the activation energy, (AG) of a reaction from 50.0 kcal/mol to 40.0 kcal/mol. Calulate the catalytic power at 310K. (R-1.987x10 kcal/mol)arrow_forward
- Draw a typical axodendritic synapse, including a specific neurotransmitter of your choice, its associated postsynaptic receptors (indicating whether they are ionotropic or metabotropic), and any associated reuptake transporters or degradation enzymes. Please include a description of what specific steps would occur as an action potential reaches the axonal terminal.arrow_forwardGive a full arrow pushing mechanism of the spontaneous redox reaction between NAD+/NADH and oxaloacetate/malate. Please include diagram drawing of the mechanism! (Thank You!)arrow_forward18. Which one of the compounds below is the major organic product obtained from the following series of reactions? 1. BH3 2. H2O2, NaOH H₂CrO4 CH2N2 oro ororos A B C D Earrow_forward
- 17. Which one of the compounds below is the major organic product obtained from the following series of reactions? CI benzyl alcohol OH PBr3 Mg 1. CO2 SOCl2 ? ether 2. H+, H₂O CI Cl HO OH CI Cl A B C D Earrow_forward14. What is the IUPAC name of this compound? A) 6-hydroxy-4-oxohexanenitrile B) 5-cyano-3-oxo-1-pentanol C) 5-cyano-1-hydroxy-3-pentanone D) 1-cyano-5-hydroxy-3-pentanone E) 5-hydroxy-3-oxopentanenitrile HO. CNarrow_forward13. What is the IUPAC name of this compound? A) 5-hydroxy-3,3-dimethylpentanoic acid B) 3,3-dimethylpentanoic acid C) 3,3-dimethyl-1-oxo-1,5-pentanediol D) 1,5-dihydroxy-3,3-dimethylpentanal E) 4-hydroxy-2,2-dimethylbutanoic acid HO OHarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning
Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning

Human Heredity: Principles and Issues (MindTap Co...
Biology
ISBN:9781305251052
Author:Michael Cummings
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Biology (MindTap Course List)
Biology
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:9781305389892
Author:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:Cengage Learning

Biology Today and Tomorrow without Physiology (Mi...
Biology
ISBN:9781305117396
Author:Cecie Starr, Christine Evers, Lisa Starr
Publisher:Cengage Learning

Principles Of Radiographic Imaging: An Art And A ...
Health & Nutrition
ISBN:9781337711067
Author:Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:Cengage Learning
Mitochondrial mutations; Author: Useful Genetics;https://www.youtube.com/watch?v=GvgXe-3RJeU;License: CC-BY