
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 7P
Draw curved arrows to show the movement of the electrons that result in formation of the given product(s).
a. 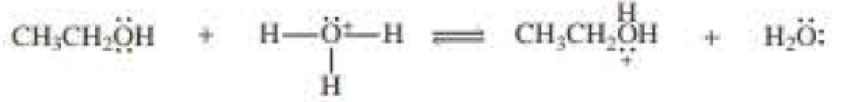
b. 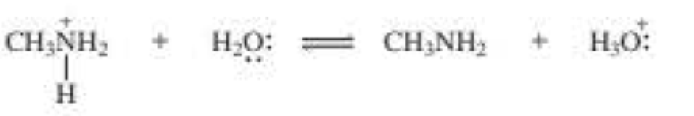
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
For the condensation reaction between Alanine and histidine write the amididation reaction mechanism using arrows then write the three letter code for the product of the reaction and the one letter code for the product of the reaction.
Write the amididation reaction mechanism of p-aminophenol and acetic acid to produce acetaminophen please use arrows.
Name the following using IUPAC.
Chapter 5 Solutions
Organic Chemistry
Ch. 5.1 - Determine the degree of unsaturation and then draw...Ch. 5.1 - Prob. 4PCh. 5.2 - Prob. 5PCh. 5.2 - Draw the structure for each of the following: a....Ch. 5.2 - What is each compounds systematic name?Ch. 5.4 - Assign relative priorities to each set of...Ch. 5.4 - Tamoxifen slows the growth of some breast tumors...Ch. 5.4 - Draw and label the E and Z isomers for each of the...Ch. 5.4 - Prob. 12PCh. 5.4 - Name each of the following:
Ch. 5.4 - Prob. 14PCh. 5.4 - Prob. 15PCh. 5.6 - Prob. 16PCh. 5.6 - Prob. 17PCh. 5.6 - Prob. 18PCh. 5.6 - Prob. 20PCh. 5.7 - a. Which of the monosubstituted cyclohexanes in...Ch. 5.7 - a. for which reaction in each set will S be more...Ch. 5.7 - a. For a reaction with H = 12 kcal/ mol and S =...Ch. 5.7 - Prob. 25PCh. 5.7 - Prob. 26PCh. 5.7 - Prob. 27PCh. 5.9 - The rate constant for a reaction can be increased...Ch. 5.9 - Prob. 30PCh. 5.9 - a. Which reaction has a greater equilibrium...Ch. 5.10 - Draw a reaction coordinate diagram for a two-step...Ch. 5.10 - a. Which step in the reaction coordinate diagram...Ch. 5.10 - Draw a reaction coordinate diagram for the...Ch. 5.11 - Prob. 35PCh. 5 - What is each compounds systematic name?Ch. 5 - Draw the structure of a hydrocarbon that has six...Ch. 5 - Draw the condensed structure for each of the...Ch. 5 - Prob. 39PCh. 5 - Prob. 40PCh. 5 - Name the following:Ch. 5 - Prob. 42PCh. 5 - 43. Draw the skeletal structure of...Ch. 5 - In a reaction in which reactant A is in...Ch. 5 - Which bond is stronger? Briefly explain why.Ch. 5 - Prob. 46PCh. 5 - Prob. 47PCh. 5 - Assign relative priorities to each set of...Ch. 5 - Prob. 49PCh. 5 - By following the curved red arrows, draw the...Ch. 5 - Prob. 51PCh. 5 - Draw structures for the following: a....Ch. 5 - Prob. 53PCh. 5 - a. Which of the following reactions has the larger...Ch. 5 - a. What is the equilibrium constant for a reaction...Ch. 5 - Prob. 56PCh. 5 - Prob. 57PCh. 5 - Given that the free energy of the twist-boat...Ch. 5 - Prob. 59PCh. 5 - Prob. 1PCh. 5 - Prob. 2PCh. 5 - Prob. 3PCh. 5 - Prob. 4PCh. 5 - Prob. 5PCh. 5 - Prob. 6PCh. 5 - Draw curved arrows to show the movement of the...Ch. 5 - Prob. 8PCh. 5 - Prob. 9PCh. 5 - Prob. 10P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- For the condensation reaction between Alamine and histamine, please help me write the amididation reaction mechanism. Then write the three letter code for the product of the reaction, then write the one letter code for the product of the reaction. arrow_forwardHow to draw the reaction mechasnism belowarrow_forwardName the following molecules with IUpacarrow_forward
- What is the molecular orbital for cyclopropenyl anion and is it aromatic, antiaromatic or nonaromatic?arrow_forwardUsing the chart describe the change from cystine to tyrosine and its impact on the protein. Using the chart describe the change from histidine to aspartic acid and its impact on the protein.arrow_forwardHow to get the predicted product of this reaction belowarrow_forward
- Please help me fill out the chart then using the chart describe the change from cystine to tyrosine and its impact on the protein. Then using the chart describe the change from histidine to aspartic acid.arrow_forwardWrite the Esterification reaction mechanism for acetic acid, and one propanol to make propanol ethanoate (molecule that gives peas its odor in flavor)arrow_forwardProvide solutionsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY