
Chemistry In Context
9th Edition
ISBN: 9781259638145
Author: Fahlman, Bradley D., Purvis-roberts, Kathleen, Kirk, John S., Bentley, Anne K., Daubenmire, Patrick L., ELLIS, Jamie P., Mury, Michael T., American Chemical Society
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3.4, Problem 3.11YT
You Decide Exposure
Examine the curve shown in Figure 3.13. Can you think of another example of something that is required for good health, but dangerous to health in high quantities?
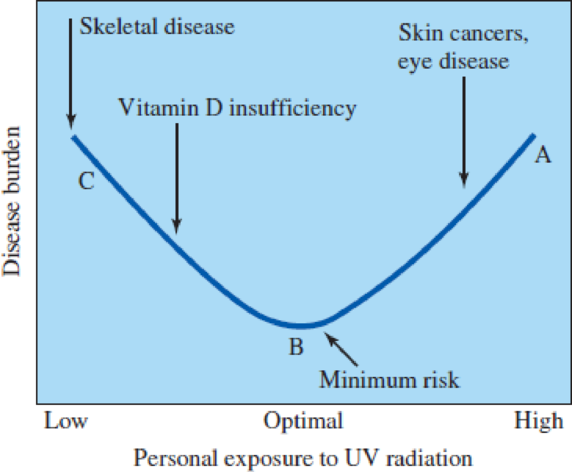
Figure 3.13
The delicate balance of “optimal” exposure to UV radiation and health.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Calculating standard reaction free energy from standard reduction...
Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free energy AG° for the following redox reaction.
Be sure your answer has the correct number of significant digits.
NO (g) +H₂O (1) + Cu²+ (aq) → HNO₂ (aq) +H* (aq)+Cu* (aq)
kJ
-
☐ x10
x10
olo
18
Ar
Calculating the pH of a weak base titrated with a strong acid
b
An analytical chemist is titrating 116.9 mL of a 0.7700M solution of aniline (C6H5NH2) with a 0.5300M solution of HNO3. The pK of aniline is 9.37.
Calculate the pH of the base solution after the chemist has added 184.2 mL of the HNO 3 solution to it.
Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HNO3 solution added.
Round your answer to 2 decimal places.
pH = ☐
☑
5
QUESTION: Find the standard deviation for the 4 different groups
5.298
3.977
223.4
148.7
5.38
4.24
353.7
278.2
5.033
4.044
334.6
268.7
4.706
3.621
305.6
234.4
4.816
3.728
340.0
262.7
4.828
4.496
304.3
283.2
4.993
3.865
244.7
143.6
STDEV =
STDEV =
STDEV =
STDEV =
Chapter 3 Solutions
Chemistry In Context
Ch. 3.1 - Prob. 3.2YTCh. 3.1 - Using Equation 3.1, answer the following...Ch. 3.1 - Prob. 3.4YTCh. 3.1 - Prob. 3.5YTCh. 3.1 - Scientific Practices Energy from the Sun Examine...Ch. 3.2 - Skill Building Wavelength, Frequency, and Energy...Ch. 3.3 - Skill Building The ABCs of Solar UV a. Arrange...Ch. 3.3 - Prob. 3.9YTCh. 3.4 - Prob. 3.10YTCh. 3.4 - You Decide Exposure Examine the curve shown in...
Ch. 3.4 - Prob. 3.12YTCh. 3.4 - Prob. 3.13YTCh. 3.5 - Prob. 3.14YTCh. 3.5 - Prob. 3.15YTCh. 3.6 - Prob. 3.16YTCh. 3.7 - Draw the Lewis structure for each molecule. a. HBr...Ch. 3.7 - Prob. 3.18YTCh. 3.7 - Prob. 3.19YTCh. 3.7 - You Decide The Ozone Layer Based on the reactions...Ch. 3.8 - Prob. 3.21YTCh. 3.8 - Prob. 3.22YTCh. 3.8 - Prob. 3.23YTCh. 3.8 - Prob. 3.24YTCh. 3.9 - Prob. 3.25YTCh. 3.10 - Prob. 3.26YTCh. 3.10 - Prob. 3.27YTCh. 3.10 - Prob. 3.28YTCh. 3.10 - Prob. 3.29YTCh. 3.11 - Skill Building Particulate Classification An...Ch. 3.11 - Prob. 3.31YTCh. 3.11 - Prob. 3.32YTCh. 3.11 - Prob. 3.33YTCh. 3.11 - Scientific Practices UV Interactions with Matter...Ch. 3 - How does ozone differ from oxygen in its chemical...Ch. 3 - Prob. 2QCh. 3 - Prob. 3QCh. 3 - Prob. 4QCh. 3 - Prob. 5QCh. 3 - a. What is a Dobson unit? b. Does a reading of 320...Ch. 3 - Using the periodic table as a guide, specify the...Ch. 3 - Consider this representation of a periodic table....Ch. 3 - Give the name and symbol for the element with this...Ch. 3 - Prob. 10QCh. 3 - Assuming that the octet rule applies, draw the...Ch. 3 - Prob. 12QCh. 3 - Consider these two waves representing different...Ch. 3 - Use Figure 3.4 to specify the region of the...Ch. 3 - What determines the color of light? Describe the...Ch. 3 - Prob. 16QCh. 3 - Does all light travel at the same speed in a...Ch. 3 - Arrange these types of radiation in order of...Ch. 3 - The microwaves in home microwave ovens have a...Ch. 3 - Ultraviolet radiation is categorized as UVA, UVB,...Ch. 3 - Calculate the wavelength, in nanometers, of the...Ch. 3 - The distance from Earth to the Sun is about 1.50 ...Ch. 3 - Draw Lewis structures for any two different CFCs.Ch. 3 - Prob. 24QCh. 3 - Prob. 25QCh. 3 - Prob. 26QCh. 3 - The following free radicals all play a role in...Ch. 3 - a. How were the original measurements of increases...Ch. 3 - Prob. 29QCh. 3 - The EPA has used the slogan Ozone: Good Up High,...Ch. 3 - Nobel Laureate F. Sherwood Rowland referred to the...Ch. 3 - Prob. 32QCh. 3 - Prob. 33QCh. 3 - Prob. 34QCh. 3 - Prob. 35QCh. 3 - The average length of an OO single bond is 132 pm....Ch. 3 - Prob. 37QCh. 3 - Describe why ozone is more reactive than oxygen...Ch. 3 - Prob. 39QCh. 3 - Prob. 40QCh. 3 - Prob. 41QCh. 3 - All the reports of the damage caused by UV...Ch. 3 - Prob. 43QCh. 3 - Prob. 44QCh. 3 - Prob. 45QCh. 3 - Prob. 46QCh. 3 - Prob. 47QCh. 3 - Development of the stratospheric ozone hole has...Ch. 3 - Prob. 49QCh. 3 - Prob. 50QCh. 3 - Resonance structures can be used to explain the...Ch. 3 - Prob. 52QCh. 3 - Prob. 53QCh. 3 - Prob. 54QCh. 3 - Prob. 55QCh. 3 - Many different types of ozone generators...Ch. 3 - The effect a chemical substance has on the ozone...Ch. 3 - Cooking with an electric stove can have a negative...Ch. 3 - One mechanism that helps break down ozone in the...Ch. 3 - Polar stratospheric clouds (PSCs) play an...Ch. 3 - Prob. 61Q
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- QUESTION: Fill in the answers in the empty green boxes regarding 'Question 5: Calculating standard error of regression' *The images of the data showing 'coefficients for the standard curve' have been providedarrow_forwardUsing the Nernst equation to calculate nonstandard cell voltage Try Again Your answer is wrong. In addition to checking your math, check that you used the right data and DID NOT round any intermediate calculations. A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction: 2+ 2+ Sn²+ Ba(s) (aq) + Ba (s) Sn (s) + Ba²+ (aq) →>> Suppose the cell is prepared with 6.10 M Sn 2+ 2+ in one half-cell and 6.62 M Ba in the other. Calculate the cell voltage under these conditions. Round your answer to 3 significant digits. 1.71 V ☐ x10 ☑ 5 0/5 ? 00. 18 Ararrow_forwardQuestion: Find both the b (gradient) and a (y-intercept) value from the list of data below: (x1 -x̄) 370.5 (y1 - ȳ) 5.240 (x2 - x̄) 142.5 (y2 - ȳ) 2.004 (x3 - x̄) 28.5 (y3 - ȳ) 0.390 (x4 - x̄) -85.5 (y4 - ȳ) -1.231 (x5 - x̄) -199.5 (y5 - ȳ) -2.829 (x6 - x̄) -256.5 (y6 - ȳ) -3.575arrow_forward
- Calculating standard reaction free energy from standard reduction... Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free energy AG° for the following redox reaction. Be sure your answer has the correct number of significant digits. 3Cu+ (aq) + Cro²¯ (aq) +4H₂O (1) → 3Cu²+ (aq) +Cr(OH)3 (s)+5OH˜¯ (aq) 0 kJ ☐ x10 00. 18 Ararrow_forwardCalculating the pH of a weak base titrated with a strong acid An analytical chemist is titrating 241.7 mL of a 0.4900M solution of methylamine (CH3NH2) with a 0.7800M solution of HNO3. The pK of methylamine is 3.36. Calculate the pH of the base solution after the chemist has added 17.7 mL of the HNO3 solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HNO3 solution added. Round your answer to 2 decimal places. pH = ☑ ? 18 Ararrow_forwardThe following is two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 223.4 148.7 353.7 278.2 334.6 268.7 305.6 234.4 340.0 262.7 304.3 283.2 244.7 143.6 QUESTION: For both groups of data calculate the answers attached in the image.arrow_forward
- The following is a two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 340.0mmol/L 262.7mmol/L QUESTION: For both groups (Regular & Salt Reduced tomato sauce) of data provide answers to the following calculations below: 1. Standard Deviation (Sx) 2. T Values (t0.05,4) 3. 95% Confidence Interval (mmol/L) 4. [Na+] (mg/100 mL) 5. 95% Confidence Interval (mg/100 mL)arrow_forwardIf we have leucine (2-amino-4-methylpentanoic acid), alanine (2-aminopropanoic acid) and phenylalanine (2-amino-3-phenylpropanoic acid), indicate the tripeptides that can be formed (use the abbreviated symbols Leu., Ala and Phe).arrow_forwardBriefly state why trifluoroacetic acid is more acidic than acetic acid.arrow_forward
- Explain why acid chlorides are more reactive than amides in reactions with nucleophiles.arrow_forwardCalculating the pH of a weak base titrated with a strong acid An analytical chemist is titrating 101.7 mL of a 0.3500M solution of piperidine (C5H10NH) with a 0.05700M solution of HClO4. The pK of piperidine is 2.89. Calculate the pH of the base solution after the chemist has added 682.9 mL of the HClO solution to it. 4 Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HClO solution added. 4 Round your answer to 2 decimal places. pH = .11 00. 18 Ararrow_forwardThe following is a two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 340.0 262.7 QUESTION: For both groups of data provide answers to the calculations attached in the imagearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY