
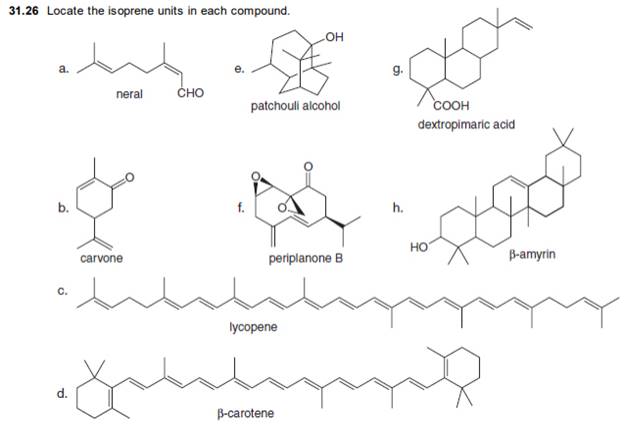
Concept explainers
Locate the isoprene units in each compound.
a. 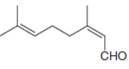 e.
e. 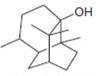 g.
g. 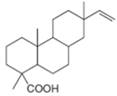
neral patchouli alcohol dextropimaric acid
b. 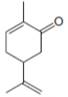 f.
f. 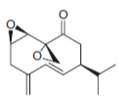 h.
h.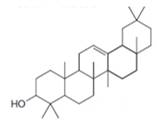
carvone periplanone B
c. 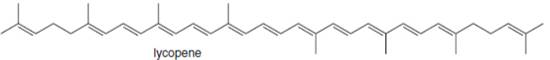
d. 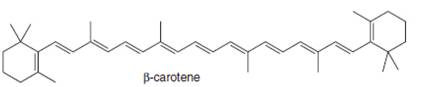
(a)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the neral are highlighted by blue color as shown below.
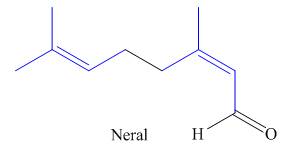
Figure 1
Explanation of Solution
The structure of isoprene is,
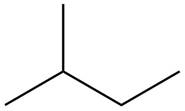
Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are highlighted by blue color as shown below.

Figure 1
The highlighted bonds represent the isoprene unit. There are two isoprene units present in the given compound.
The isoprene units in the neral are shown in Figure 1.
(b)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the carvone are highlighted by blue color as shown below.
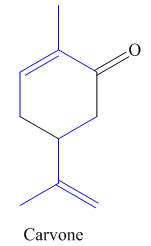
Figure 3
Explanation of Solution
The structure of isoprene is,
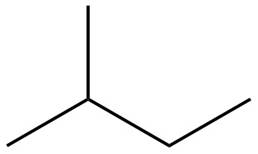
Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are highlighted by blue color as shown below.

Figure 3
The highlighted bonds represent the isoprene unit. There are two isoprene units present in the given compound.
The isoprene units in the carvone are shown in Figure 3.
(c)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the lycopene are highlighted by blue color as shown below.
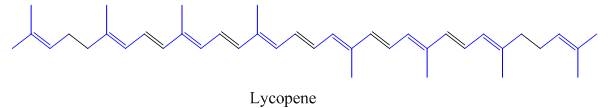
Figure 4
Explanation of Solution
The structure of isoprene is,
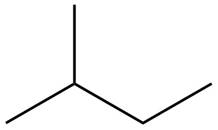
Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are are highlighted by blue color as shown below.

Figure 4
The highlighted bonds represent the isoprene unit. There are eight isoprene units present in the given compound.
The isoprene units in the lycopene are shown in Figure 4.
(d)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the
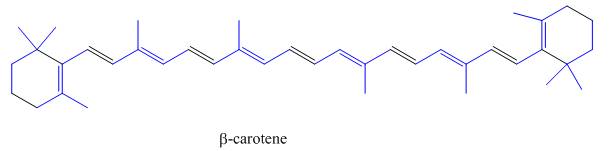
Figure 5
Explanation of Solution
The structure of isoprene is,
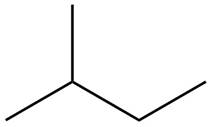
Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are highlighted by blue color as shown below.

Figure 5
The highlighted bonds represent the isoprene unit. There are eight isoprene units present in the given compound.
The isoprene units in the
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the patchouli alcohol are highlighted by blue color as shown below.
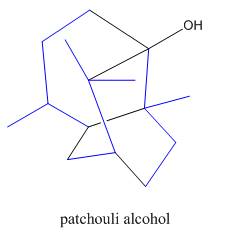
Figure 6
Explanation of Solution
The structure of isoprene is,
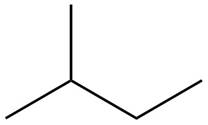
Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are are highlighted by blue color as shown below.

Figure 6
The highlighted bonds represent the isoprene unit. There are three isoprene units present in the given compound.
The isoprene units in the patchouli alcohol are shown in Figure 6.
(f)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the periplanone B are highlighted by blue color as shown below.
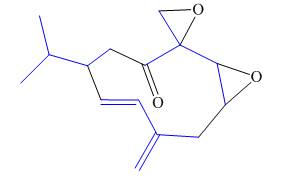
Figure 7
Explanation of Solution
The structure of isoprene is,

Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are highlighted by blue color as shown below.
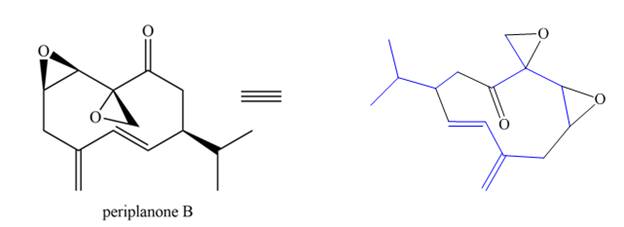
Figure 7
The highlighted bonds represent the isoprene unit. There are three isoprene units present in the given compound.
The isoprene units in the periplanone B are shown in Figure 7.
(g)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the dextropimaric acid are highlighted by blue color as shown below.
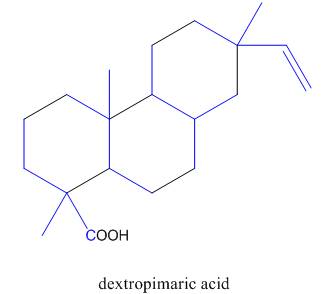
Figure 8
Explanation of Solution
The structure of isoprene is,

Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are shown below.

Figure 8
The highlighted bonds represent the isoprene unit. There are four isoprene units present in the given compound.
The isoprene units in the dextropimaric acid are shown in Figure 8.
(h)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the
Figure 9
Explanation of Solution
The structure of isoprene is,

Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are shown below.
Figure 9
The highlighted bonds represent the isoprene unit. There are four isoprene units present in the given compound.
The isoprene units in the
Want to see more full solutions like this?
Chapter 31 Solutions
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
- please help with synthesisarrow_forward10. Stereochemistry. Assign R/S stereochemistry for the chiral center indicated on the following compound. In order to recieve full credit, you MUST SHOW YOUR WORK! H₂N CI OH CI カー 11. () Stereochemistry. Draw all possible stereoisomers of the following compound. Assign R/S configurations for all stereoisomers and indicate the relationship between each as enantiomer, diastereomer, or meso. NH2 H HNH, -18arrow_forwardb) 8. Indicate whether the following carbocation rearrangements are likely to occur Please explain your rational using 10 words or less not likely to occur • The double bond is still in the Same position + Likely to oc occur WHY? -3 H3C Brave Chair Conformers. Draw the chair conformer of the following substituted cyclohexane. Peform a RING FLIP and indicate the most stable conformation and briefly explain why using 20 words or less. CI 2 -cobs ?? MUST INDICATE H -2 -2 Br EQ Cl OR AT Br H& most stable WHY? - 4arrow_forward
- CH 12 Conformational Analysis. Draw all 6 conformers (one above each letter) of the compound below looking down the indicated bond. Write the letter of the conformer with the HIGHEST and LOWEST in energies on the lines provided. NOTE: Conformer A MUST be the specific conformer of the structure as drawn below -4 NOT HOH OH 3 Conformer A: Br OH A Samo Br H 04 Br H H3 CH₂ H anti stagere Br CH clipsed H Brott H IV H MISSING 2 -2 B C D E F X 6 Conformer with HIGHEST ENERGY: 13. (1 structure LOWEST ENERGY: Nomenclature. a) Give the systematic (IUPAC) name structure. b) Draw the corresponding to this name. HINT: Do not forget to indicate stereochemistry when applicable. a) ८८ 2 "Br {t༐B,gt)-bemn€-nehpརི་ཚ༐lnoa Parent name (noname) 4 Bromo Sub = 2-methylethyl-4 Bromo nonane b) (3R,4S)-3-chloro-4-ethyl-2,7-dimethyloctane # -2 -2arrow_forwardin the scope of the SCH4U course! please show all steps as im still learning how to format my answers in the format given, thank you!arrow_forwardhelp me solve this HWarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
