
Concept explainers
(a)
Interpretation:
The hybridization of each nonhydrogen atom in norethynodrel is to be determined.
Concept introduction:
Answer to Problem 3.38P
| Atom | Hybridization |
| sp |
Explanation of Solution
The structure of Norethynodrel is as below:
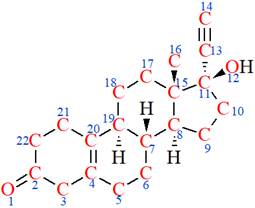
The nonhydrogen atoms are highlighted with red color and numbered.
According to VSEPR theory,
According to VSEPR theory,
According to VSEPR theory, the
According to VSEPR theory,
According to VSEPR theory, geometry of an atom has been determined, and from the geometry of the atom, its hybridization has been determined.
(b)
Interpretation:
The total number of
Concept introduction:
Answer to Problem 3.38P
There are total fifty-one
Explanation of Solution
The structure of Norethynodrel is as below:
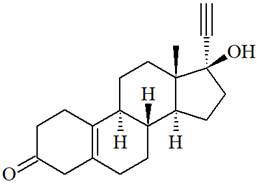
There is one C=C triple bond in norethynodrel molecule. Each triple bond is composed of two
From the
Want to see more full solutions like this?
Chapter 3 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- 3. Reactions! Fill in the information missing below. Make sure to pay attention to REGIOCHEMISTRY and STEREOCHEMISTRY. Br2 CH3OH + 4. Mechanism! Show the complete arrow pushing mechanism, including all steps and intermediates for the following reactions. To get credit for this, you MUST show how ALL bonds are broken and formed, using arrows to show the movement of electrons. H3O+ HOarrow_forwardPlease provide a synthesis for the Ester using proponoic acid, thank you!arrow_forwardPlease help with the curved arrow mechanism of this reaction, thank youarrow_forward
- Concentration (mg/l) Peak Area 0 158 10 10241 20 18425 30 26457 40 37125 50 44256 60 56124 Question: Determine the regression equation (a and b coefficients) from first principlesarrow_forwardConcentration (mg/l) Peak Area 0 158 10 10241 20 18425 30 26457 40 37125 50 44256 60 56124 You have been asked to determine the concentration of citral in a highly valued magnolia essential oil. QUESTION: Calculate the concentration of citral in your highly valued magnolia essential oil which returns a peak area of 41658arrow_forwardNeed help with these problems...if you can please help me understand problems E & F.arrow_forward
- Please help me solve these problems. Thank you in advance.arrow_forwardPredict the products of this organic reaction: O N IN A N + H2O + HCI ? Specifically, in the drawing area below draw the skeletal ("line") structure of the product, or products, of this reaction. If there's more than one product, draw them in any arrangement you like, so long as they aren't touching. If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. 田 C + Explanation Check Click and drag to start drawing a structure. C © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Centerarrow_forward6. For each of the following, fill in the synthesis arrows with reagents and show the intermediates. You DO NOT need to use the same number of arrows that are shown (you may use more or less), but the product must be formed from the reactant. Then write the mechanism of one step in the synthesis (you can choose which step to write the mechanism for), including all reagents required, clearly labeling the nucleophile and electrophile for each step, and using curved arrows to show the steps in the mechanism. a. b. OHarrow_forward
- Draw the productsarrow_forwardDraw the correct productsarrow_forwardE Organic Chemistry Maxwell Draw the correct products, in either order, for the ozonolysis reaction: 1) O3, CH2Cl2, -78 °C Product 1 + Product 2 2) Zn, HOAc Draw product 1. Select Draw Templates More C H O presented by M Draw product 2. Erase Select Draw Templates M / # # carrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





