
Concept explainers
(a)
Interpretation:
The moles of
Concept introduction:
The SI unit for amount of substance is
The amount of an element can be calculated from the given amount of compound and the moles of that element present in one mole of compound. The molecular formula of the compound tells the number of atoms of each element. The road map to calculate the moles of an element in a compound is as follows:

The expression to calculate the amount of an element in a compound is as follows:
(b)
Interpretation:
The number of
Concept introduction:
The SI unit for amount of substance is
The number of an atom of an element can be calculated from the given amount of compound, the moles of that element present in one mole of the compound and the Avogadro’ number.
The road map to calculate the number of atoms of an element in a compound is as follows:
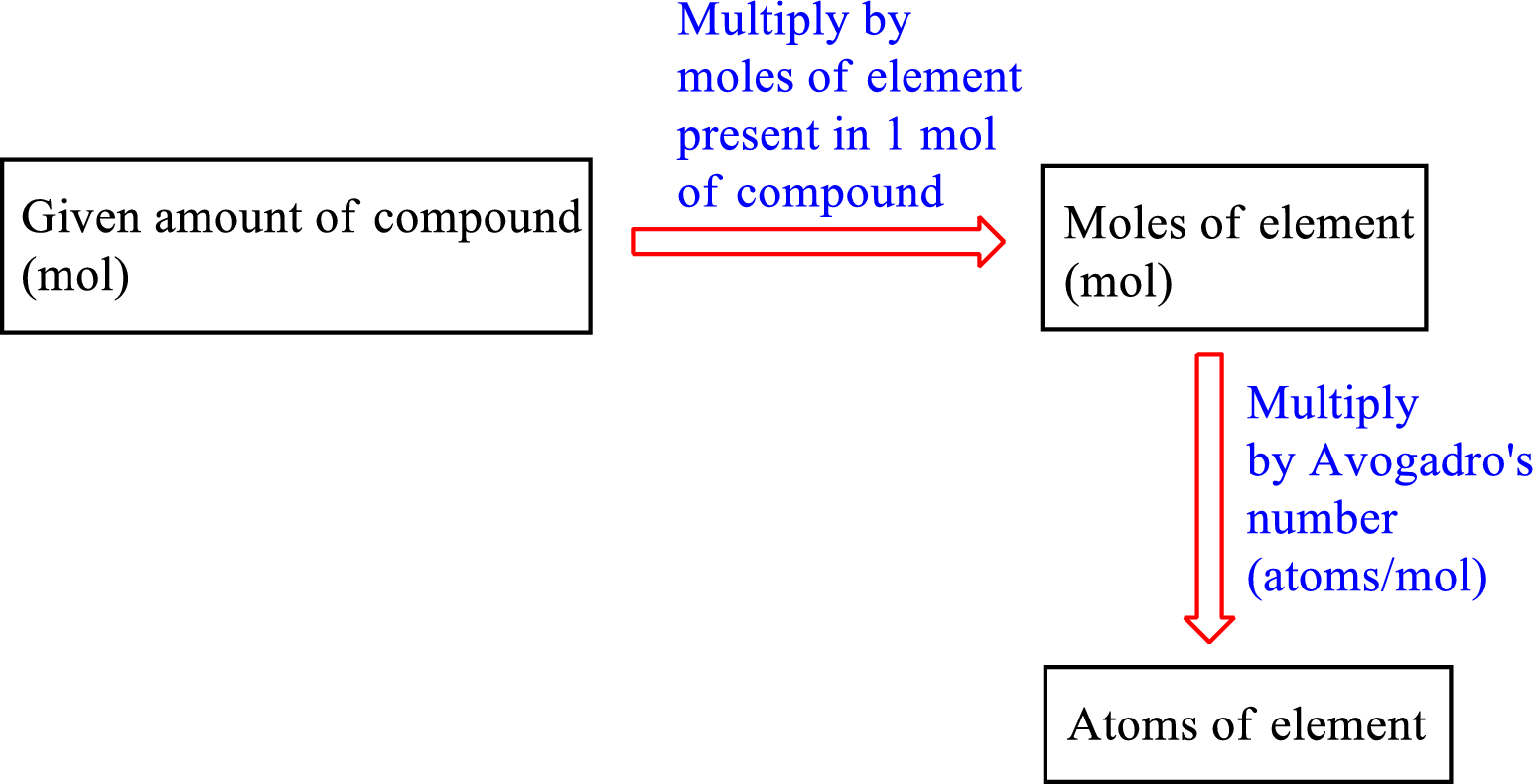
The expression to calculate the number of atoms of an element in a compound is as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Chemistry: The Molecular Nature of Matter and Change - Standalone book
- in the following reaction, the OH- acts as which of these?NO2- (aq) + H2O (l) ⇌ OH- (aq) + HNO2 (aq)a) not a weak acidb) basec) acidarrow_forwardfind the pH of a buffer made from 0.20 M HNO2 and 0.10 M NaNO2. Ka= 4.0 x 10-4a) 4.00b) 3.40c) 3.70d) 3.10arrow_forwardthe Ka for sodium dihydrogen phosphate is 6.32 x 10-8. Find the pH of a buffer made from 0.15 M H2PO4- and 0.15 M HPO42-.a) 6.98b) 7.42c) 7.00d) 7.20arrow_forward
- Find the equilibrium concentration of H3O+ starting with 0.072 M solution of acetic acid. Ka = 1.8 x 10-5. Acetic acid is HC2H3O2 (aq).HC2H3O2 (aq) + H2O (l) ⇌ H3O (aq) + C2H3O2- (aq) a) 1.3 x 10-6 b) 1.1 x 10-3 c) 1.5 x 10-2 d) 3.6 x 10-5arrow_forwardin VSEPR Theory, AX2 isarrow_forwardcalculate the pH of 0.066 M Ca(OH)2. Remember stoichiometry.arrow_forward
- Find the equilibrium concentration of H3O+ starting with 0.072 M solution of acetic acid. Ka = 1.8 x 10-5. Acetic acid is HC2H3O2 (aq).HC2H3O2 (aq) + H2O (l) ⇌ H3O (aq) + C2H3O2- (aq)arrow_forwardin VSEPR Theory AX2 isa) tetrahedralb) octahedralc) lineard) trigonal bipyramidarrow_forwardCheck How many signals would you expect to find in the H NMR spectrum of each given compound? Part 1 of 2 Part 2 of 2 Br Br 2. Cl X 2 © 2025 McGraw Hill LLC. All Rights Resarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





