
Concept explainers
The time at which the packet fell off the back ramp.
Explanation of Solution
Given: The speed of the airplane is
The angle is
The distance is
Formula used: Draw the diagram for the representation of the package when it fell off the ramp.
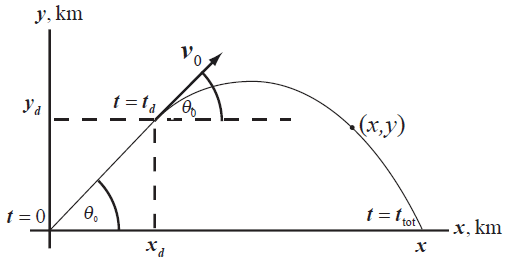
Write the expression for the distance in x direction.
Here,
Write the expression for the distance in y direction.
Here,
Write the expression for the x coordinate along the parabolic trajectory of the airplane.
Here,
Write the expression for the y coordinate along the parabolic trajectory of the airplane.
Here,
Substitute
Substitute
Simplify the above equation
Write the expression for the velocity component with respect to x axis.
Here,
Write the expression for the velocity component with respect to y axis.
At impact, the horizontal distance is
Write the expression for the distance to impact.
Simplify the above equation for
Substitute
Substitute
Solve the above equation using binomial expansion.
Calculation:
Substitute
Substitute
Substitute
Conclusion:
Thus, the time at which the packet fell off the back ramp is
Want to see more full solutions like this?
Chapter 3 Solutions
Physics for Scientists and Engineers
- Consider a uniformly charged ring of radius R with total charge Q, centered at the origin inthe xy-plane. Find the electric field (as a vector) at a point on the z-axis at a distance z above thecenter of the ring. Assume the charge density is constant along the ring.arrow_forward3) If the slider block C is moving at 3m/s, determine the angular velocity of BC and the crank AB at the instant shown. (Use equation Vs Vc wx fuc, then use equation Vs VA + Ve/athen write it in terms of w and the appropriate r equate the two and solve) 0.5 m B 1 m 60° A 45° vc = 3 m/sarrow_forward3) If the slider block C is moving at 3m/s, determine the angular velocity of BC and the crank AB at the instant shown. (Use equation Vs Vc wxf, then use equation V, VA + Va/Athen write it in terms of w and the appropriate r equate the two and solve) f-3marrow_forward
- Pls help ASAParrow_forwardPls help ASAParrow_forward14. A boy is out walking his dog. From his house, he walks 30 m North, then 23 m East, then 120 cm South, then 95 m West, and finally 10 m East. Draw a diagram showing the path that the boy walked, his total displacement, and then determine the magnitude and direction of his total displacement.arrow_forward
- Pls help ASAParrow_forward12. A motorboat traveling 6 m/s, West encounters a water current travelling 3.5 m/s, South. a) Draw a vector diagram showing the resultant velocity, then determine the resultant velocity of the motorboat. b) If the width of the river is 112 m wide, then how much time does it take for the boat to travel shore to shore? c) What distance downstream does the boat reach the opposite shore?arrow_forwardLake Erie contains roughly 4.00⋅10114.00⋅1011 m3 of water. Assume the density of this water is 1000. kg/m3 and the specific heat of water is 4186 J/kg˚C. It takes 2.31x10^19 J of energy to raise the temperature of that volume of water from 12.0 °C to 25.8 ˚C. An electric power plant can produce about 1110 MW. How many years would it take to supply this amount of energy by using the 1110 MW from an electric power plant?arrow_forward
 Classical Dynamics of Particles and SystemsPhysicsISBN:9780534408961Author:Stephen T. Thornton, Jerry B. MarionPublisher:Cengage Learning
Classical Dynamics of Particles and SystemsPhysicsISBN:9780534408961Author:Stephen T. Thornton, Jerry B. MarionPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill
Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill University Physics Volume 1PhysicsISBN:9781938168277Author:William Moebs, Samuel J. Ling, Jeff SannyPublisher:OpenStax - Rice University
University Physics Volume 1PhysicsISBN:9781938168277Author:William Moebs, Samuel J. Ling, Jeff SannyPublisher:OpenStax - Rice University College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning





