
Concept explainers
Identify the following bases, and tell whether each is found in DNA, RNA, or both:
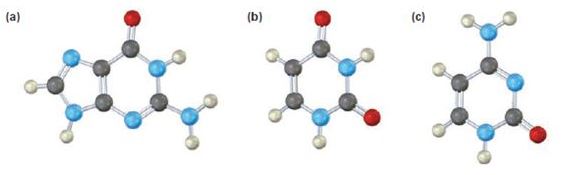
a)
Interpretation:
To identify the following bases and state whether each lie in DNA, RNA or both.
Concept introduction:
Proteins and biopolymers are made up of amino acids, nucleic acids which are joined together to form a long chain. Nucleic acids are the biopolymers of nucleotides, which is composed of a nucleoside. Each nucleoside is bonded to a phosphate group.
Each nucleoside is composed of an aldopentose sugar linked through its anomeric carbon to the nitrogen atom of a heterocyclic purine or pyrimidine base. RNA contains ribose as a sugar component and DNA contains 29-deoxyribose as a sugar component.
DNA contains four different amine bases: two substituted purines (adenine and guanine) and two substituted pyrimidines (cytosine and thymine). Adenine, guanine, and cytosine also occur in RNA, but thymine is replaced in RNA by a closely related pyrimidine base called uracil.
Answer to Problem 13VC
The base given here is: Guanine (G)
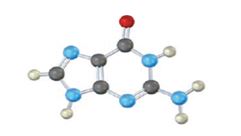
It is found in both DNA and RNA.
Explanation of Solution
In the above diagrams of bases, guanine and cytosine are found in DNA and RNA. Whereas, uracil is found in only RNA. Here the blue atoms are the symbols for nitrogen, red atom represents oxygen, black atom represents carbon and grey atom represnts hydrogen.
The base given here is: Guanine (G)

It is found in both DNA and RNA.
b)
Interpretation:
To identify the following bases and state whether each lie in DNA, RNA or both.
Concept introduction:
Proteins and biopolymers are made up of amino acids, nucleic acids which are joined together to form a long chain. Nucleic acids are the biopolymers of nucleotides, which is composed of a nucleoside. Each nucleoside is bonded to a phosphate group.
Each nucleoside is composed of an aldopentose sugar linked through its anomeric carbon to the nitrogen atom of a heterocyclic purine or pyrimidine base. RNA contains ribose as a sugar component and DNA contains 29-deoxyribose as a sugar component.
DNA contains four different amine bases: two substituted purines (adenine and guanine) and two substituted pyrimidines (cytosine and thymine). Adenine, guanine, and cytosine also occur in RNA, but thymine is replaced in RNA by a closely related pyrimidine base called uracil.
Answer to Problem 13VC
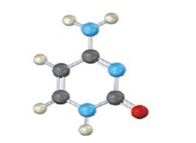
The base given here is: Uracil (U)
It is found in RNA.
Explanation of Solution
In the above diagrams of bases, guanine and cytosine are found in DNA and RNA. Whereas, uracil is found in only RNA. Here the blue atoms are the symbols for nitrogen, red atom represents oxygen, black atom represents carbon and grey atom represnts hydrogen.
The base given here is: Uracil (U)

It is found in RNA.
c)
Interpretation:
To identify the following bases and state whether each lie in DNA, RNA or both.
Concept introduction:
Proteins and biopolymers are made up of amino acids, nucleic acids which are joined together to form a long chain. Nucleic acids are the biopolymers of nucleotides, which is composed of a nucleoside. Each nucleoside is bonded to a phosphate group.
Each nucleoside is composed of an aldopentose sugar linked through its anomeric carbon to the nitrogen atom of a heterocyclic purine or pyrimidine base. RNA contains ribose as a sugar component and DNA contains 29-deoxyribose as a sugar component.
DNA contains four different amine bases: two substituted purines (adenine and guanine) and two substituted pyrimidines (cytosine and thymine). Adenine, guanine, and cytosine also occur in RNA, but thymine is replaced in RNA by a closely related pyrimidine base called uracil.
Answer to Problem 13VC
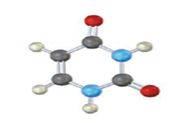
The base given here is: Cytosine (C)
It is found in both DNA and RNA.
Explanation of Solution
In the above diagrams of bases, guanine and cytosine are found in DNA and RNA. Whereas, uracil is found in only RNA. Here the blue atoms are the symbols for nitrogen, red atom represents oxygen, black atom represents carbon and grey atom represnts hydrogen.
The base given here is: Cytosine (C)

It is found in both DNA and RNA.
Want to see more full solutions like this?
Chapter 28 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Applications and Investigations in Earth Science (9th Edition)
Organic Chemistry
General, Organic, and Biological Chemistry - 4th edition
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
The Cosmic Perspective (8th Edition)
- What is the structure of the DNA backbone?arrow_forwardPLEASE PLEASE PLEASE use hand drawn structures when possarrow_forward. M 1- MATCH each of the following terms to a structure from the list below. There is only one correct structure for each term and structures may be used more than once. Place the letter of the structure in the blank to the left of the corresponding term. A. Sanger dideoxy method C. Watson-Crick B. GAUCGUAAA D. translation E. HOH2C OH OH G. transcription I. AUGGCUGAG 0 K. OPOH2C 0- OH N- H NH2 F. -OPOH2C 0- OH OH H. Maxam-Gilbert method J. replication N L. HOH2C a. b. C. d. e. f. g. B M. AGATCGCTC a pyrimidine nucleoside RNA base sequence with guanine at the 3' end. DNA base sequence with cytosine at the 3' end. a purine nucleoside DNA sequencing method for the human genome 2'-deoxyadenosine 5'-phosphate process by which mRNA directs protein synthesis OH NH2arrow_forward
- Please use hand drawn structures when neededarrow_forwardB. Classify the following amino acid. Atoms other than carbon and hydrogen are labeled. a. acidic b. basic C. neutral C. Consider the following image. Which level of protein structure is shown here? a. primary b. secondary c. tertiary d. quaternary D. Consider the following image. H RH H HR H R HR HR RH Which level of protein structure is shown in the box? a. primary b. secondary R c. tertiary d. quaternary コー Rarrow_forwardBriefly answer three from the followings: a. What are the four structures of the protein? b. Why is the side chain (R) attached to the alpha carbon in the amino acids is important for the function? c. What are the types of amino acids? And how is it depend on the (R) structure? d. Write a reaction to prepare an amino acid. prodarrow_forward
- Answe Answer A and B pleasearrow_forward3. Refer to the data below to answer the following questions: Isoelectric point Amino Acid Arginine 10.76 Glutamic Acid 3.22 Tryptophan 5.89 A. Define isoelectric point. B. The most basic amino acid is C. The most acidic amino acid is sidizo zoarrow_forward3. A gas mixture contains 50 mol% H2 and 50 mol% He. 1.00-L samples of this gas mixture are mixed with variable volumes of O2 (at 0 °C and 1 atm). A spark is introduced to allow the mixture to undergo complete combustion. The final volume is measured at 0 °C and 1 atm. Which graph best depicts the final volume as a function of the volume of added O2? (A) 2.00 1.75 Final Volume, L 1.50 1.25 1.00 0.75 0.50 0.25 0.00 0.00 0.25 0.50 2.00 (B) 1.75 1.50 Final Volume, L 1.25 1.00 0.75 0.50- 0.25 0.00 0.75 1.00 0.00 0.25 Volume O₂ added, L 2 0.50 0.75 1.00 Volume O₂ added, L 2 2.00 2.00 (C) (D) 1.75 1.75 1.50 1.50 Final Volume, L 1.25 1.00 0.75 0.50 Final Volume, L 1.25 1.00 0.75 0.50 0.25 0.25 0.00 0.00 0.00 0.25 0.50 0.75 1.00 0.00 0.25 Volume O₂ added, L 0.50 0.75 1.00 Volume O₂ added, L 2arrow_forward
- Leucine is an essential amino acid with the systematic name 2-amino-3-methylpentanoic acid. It has pai 2.36 and pKa2 = 9.60. H2N-C(R)H-COOH and R is -CH2-CH(CH3)2 A. Draw the condensed structure for leucine, and label all chirality centers with an asterisk. B. How many possible stereoisomers of leucine are there? C. Draw a Fischer projection of L-leucine and label the chirality center(s) as R or S. D. What is the p/ of leucine? E. Draw the structure of the predominant form of leucine at 10.00. F. Draw the structure of the predominant form of leucine at pH = 1.50. G. Leucine is described as an essential amino acid. What does this mean? H. Show the alkyl halide you would use to prepare leucine by the amidomalonate method. =arrow_forwarda) Write out 6 completely different reactions of acetophenone (reagent, product). b) Write out 3 preparations of 1-methylcyclohexanol, using a different starting material for each one. You may use preps where you just change the functional group, and/or preps where you construct the carbon chain. c) Write out 3 preparations of 2-ethoxybenzoic acid, a different starting material for each one. You may use preps where you just change the functional group, and/or preps where you construct the carbon chain.arrow_forward12. CH3 OH OH H&C CH3 H₂C N OH H₂C CH3 H&C CH3 H₂C' CH3 H.C CH3OH H.C CH2CH3OH CH3CEN Which one of these 17 compounds is represented by this IR and this 'H NMR spectrum? IR Spectrum 3000 4000 3000 NMR Spectrum 2000 £500 RAVENUMBER 2000 1500 9 8 6 5 10 HP-00-290 ppm m 1000 500 1000 4 °arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,





