
(a)
Interpretation:
When each of the compounds shown in Fig. P28.43 is heated in the presence of maleic anhydride, an inermediate is trapped as Diels-Alder adduct. The intermediate formed in each reaction, and its formation from the starting material are to be stated.
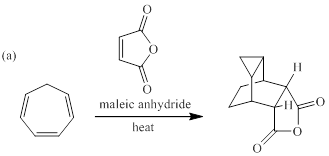
Concept introduction:
cycloaddition reaction can be referred as a reaction of two different π-electron systems to form a ring product. Notably, in this reaction two new sigma bonds are formed in product with the disappearance of two fewer π-bonds. In other words, cyclo addition reaction can be best described as the reaction of number of electrons involved. In [4+2] cycloaddition one component is having four electrons and other component is having two electrons.
(b)
Interpretation:
When each of the compounds shown in Fig. P28.43 is heated in the presence of maleic anhydride, an inermediate is trapped as Diels-Alder adduct. The intermediate formed in each reaction, and its formation from the starting material are to be stated.

Concept introduction:
Sigmatropic reaction can be described as the migration of allylic sigma bond at one end of the π-electron system to the other end of the π-electron system as an uncatalyzed intramolecular reaction. Though, the position of π-bond is changed in Sigmatropic reaction, the total number of π-bonds remain unchanged. The sigma bond can be cleaved at the middle or at the end of the π-system. The formation of sigma bond at 3, 3-position of a 1, 5-diene is called as cope rearrangement. Notably, [3, 3] sigmatropic reaction of allyl vinyl ether is termed as Claisen rearrangement.
(c)
Interpretation:
When each of the compounds shown in Fig. P28.43 is heated in the presence of maleic anhydride, an inermediate is trapped as Diels-Alder adduct. The intermediate formed in each reaction, and its formation from the starting material are to be stated.
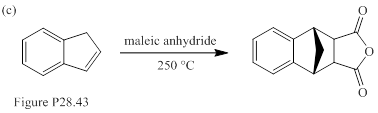
Concept introduction:
Sigmatropic reaction can be described as the migration of allylic sigma bond at one end of the π-electron system to the other end of the π-electron system as an uncatalyzed intramolecular reaction. Though, the position of π-bond is changed in Sigmatropic reaction, the total number of π-bonds remain unchanged. The sigma bond can be cleaved at the middle or at the end of the π-system. The formation of sigma bond at 3, 3-position of a 1, 5-diene is called as cope rearrangement. Notably, [3, 3] sigmatropic reaction of allyl vinyl ether is termed as Claisen rearrangement.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
ORGANIC CHEMISTRY SAPLING ACCESS + ETEX
- Q5: Label each chiral carbon in the following molecules as R or S. Make sure the stereocenter to which each of your R/S assignments belong is perfectly clear to the grader. (8pts) R OCH 3 CI H S 2pts for each R/S HO R H !!! I OH CI HN CI R Harrow_forwardCalculate the proton and carbon chemical shifts for this structurearrow_forwardA. B. b. Now consider the two bicyclic molecules A. and B. Note that A. is a dianion and B. is a neutral molecule. One of these molecules is a highly reactive compound first characterized in frozen noble gas matrices, that self-reacts rapidly at temperatures above liquid nitrogen temperature. The other compound was isolated at room temperature in the early 1960s, and is a stable ligand used in organometallic chemistry. Which molecule is the more stable molecule, and why?arrow_forward
- A mixture of C7H12O2, C9H9OCl, biphenyl and acetone was put together in a gas chromatography tube. Please decide from the GC resutls which correspond to the peak for C7,C9 and biphenyl and explain the reasoning based on GC results. Eliminate unnecessary peaks from Gas Chromatography results.arrow_forwardIs the molecule chiral, meso, or achiral? CI .CH3 H₂C CIarrow_forwardPLEASE HELP ! URGENT!arrow_forward
- Identify priority of the substituents: CH3arrow_forwardHow many chiral carbons are in the molecule? OH F CI Brarrow_forwardA mixture of three compounds Phen-A, Acet-B and Rin-C was analyzed using TLC with 1:9 ethanol: hexane as the mobile phase. The TLC plate showed three spots of R, 0.1 and 0.2 and 0.3. Which of the three compounds (Phen-A; Acet-B or Rin-C) would have the highest (Blank 1), middle (Blank 2) and lowest (Blank 3) spot respectively? 0 CH: 0 CH, 0 H.C OH H.CN OH Acet-B Rin-C phen-A A A <arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

