
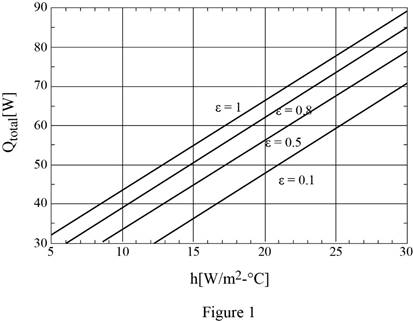
The plotting of rate of heat transfer against the convection heat transfer coefficient for the surface emissivities of 0.1, 0.5, 0.8, and 1. Also, discuss the results.
Answer to Problem 100P
The plotting of rate of heat transfer against the convection heat transfer coefficient for the surface emissivities of 0.1, 0.5, 0.8, and 1 are shown in Figure (1) and results are discussed as below.
Explanation of Solution
Calculate the rate of heat transfer by convection.
Here, change in the temperature is
Calculate the rate of heat transfer by radiation.
Here, surface temperature is
Calculate the total rate of heat transfer from the ball.
Conclusion:
Let us solve for
Substitute
Substitute
Substitute 34.35 W for
Follow the above process to calculate the rate of heat transfer against the convection heat transfer coefficient for the surface emissivities of 0.1 and 0.5 using spreadsheet including equations (I), (II), and (III) as in table (1).
| 5 | 11.4511 | 2.04127 | 13.4924 | 10.2064 | 21.6575 |
| 7.5 | 17.1767 | 2.04127 | 19.2179 | 10.2064 | 27.383 |
| 10 | 22.9022 | 2.04127 | 24.9435 | 10.2064 | 33.1086 |
| 12.5 | 28.6278 | 2.04127 | 30.669 | 10.2064 | 38.8341 |
| 15 | 34.3533 | 2.04127 | 36.3946 | 10.2064 | 44.5597 |
| 17.5 | 40.0789 | 2.04127 | 42.1201 | 10.2064 | 50.2852 |
| 20 | 45.8044 | 2.04127 | 47.8457 | 10.2064 | 56.0108 |
| 22.5 | 51.53 | 2.04127 | 53.5712 | 10.2064 | 61.7363 |
| 25 | 57.2555 | 2.04127 | 59.2968 | 10.2064 | 67.4619 |
| 27.5 | 62.9811 | 2.04127 | 65.0224 | 10.2064 | 73.1874 |
| 30 | 68.7066 | 2.04127 | 70.7479 | 10.2064 | 78.913 |
Continue table (1) for
| 16.3302 | 27.7813 | 20.4127 | 31.8638 |
| 16.3302 | 33.5068 | 20.4127 | 37.5894 |
| 16.3302 | 39.2324 | 20.4127 | 43.3149 |
| 16.3302 | 44.9579 | 20.4127 | 49.0405 |
| 16.3302 | 50.6835 | 20.4127 | 54.766 |
| 16.3302 | 56.4091 | 20.4127 | 60.4916 |
| 16.3302 | 62.1346 | 20.4127 | 66.2172 |
| 16.3302 | 67.8602 | 20.4127 | 71.9427 |
| 16.3302 | 73.5857 | 20.4127 | 77.6683 |
| 16.3302 | 79.3113 | 20.4127 | 83.3938 |
| 16.3302 | 85.0368 | 20.4127 | 89.1194 |
Show the plotting of rate of heat transfer against the convection heat transfer coefficient for the surface emissivities of 0.1, 0.5, 0.8, and 1.0 using Table (1) and (2) as in Figure (1).
Want to see more full solutions like this?
Chapter 2 Solutions
THERMODYNAMICS (LL)-W/ACCESS >CUSTOM<
- subject: combustion please include complete solution, no rounding off, with diagram/explanation etc. In a joule cycle, intake of the compressor is 40,000 cfm at 0.3 psig and 90 deg F. The compression ratio is 6.0 and the inlet temperature at the turbine portion is 1900R while at the exit, it is 15 psi. Calculate for the back work ratio in percent.arrow_forwardsubject: combustion please include complete solution, no rounding off, with diagram/explanation etc. A gasoline engine, utilizing cold air, recorded a work of 431 BTU/lb at a maximum temperature of 3,273 K and 1112 deg F temperature at the beginning of constant volume heat addition. What is the compression ratio?arrow_forwardsubject: combustion please do step by step solution and no rounding off, complete solution with diagram/explanation if needed etc. thank you! Air enters the compressor at 101,320 Pascals, 305.15K, and leaves at a pressure of 0.808MPa. The air is heated to 990.15K in the combustion chamber. For a net output of 2,125,000 Watts, find the rate of flow of air per second.arrow_forward
- The link lengths and the value of 2 and offset for some fourbar crank-slide linkages are defined in Table 1. The linkage configuration and terminology are shown in Figure 1. For the rows assigned, find (a) all possible solutions for angle & and slider position d by vector loop method. (b) the transmission angle corresponding to angle 03. (Hint: Treat the vector R4 as virtual rocker) Show your work in details: vector loop, vector equations, solution procedure. Table 1 Row Link 2 Link 3 Offset Ө a 1.4 4 1 45° b 3 8 2 -30° C 5 20 -5 225° 03 slider axis B X offset Link 2 A R3 Link 3 R4 04 R2 02 R1 d Figure 1. Xarrow_forward4. Two links made of heat treated 6061 aluminum (Sy = 276 MPa, Sys = 160 MPa) are pinned together using a steel dowel pin (Sy = 1398 MPa, Sys = 806 MPa) as shown below. The links are to support a load P with a factor of safety of at least 2.0. Determine if the link will fail first by tearout, direct shear of the pin, bearing stress on the link, or tensile stress at section AA. (Hint: find the load P for each case and choose the case that gives the smallest load.) P 8 mm P 8 mm ¡+A 3 mm →A 10 mm Parrow_forward1. For a feature other than a sphere, circularity is where: A. The axis is a straight line B. The modifier is specified with a size dimension C. All points of the surface intersected by any plane perpendicular to an axis or spine (curved line) are equidistant from that axis or spine D. All points of the surface intersected by any plane passing through a common center are equidistant from that center 2. What type of variation is limited by a circularity toler- ance zone? A. Ovality B. Tapering C. Bending D. Warping 3. How does the Rule #1 boundary affect the application of a circularity tolerance? A. The modifier must be used. B. The feature control frame must be placed next to the size dimension. C. The circularity tolerance value must be less than the limits of size tolerance. D. Circularity cannot be applied where a Rule #1 boundary exists. 4. A circularity tolerance may use a modifier. A. Ø B. F C. M D. ℗ 5. A real-world application for a circularity tolerance is: A. Assembly (i.e.,…arrow_forward
- 3. A steel bar is pinned to a vertical support column by a 10 mm diameter hardened dowel pin, Figure 1. For P = 7500 N, find: a. the shear stress in the pin, b. the direct bearing stress on the hole in the bar, c. the minimum value of d to prevent tearout failure if the steel bar has a shear strength of 175 MPa. support column pin bar thickness of bar = 8 mm h d 150 mmarrow_forwardA press that delivers 115 strokes per minute, each stroke providing a force of 7826 N throughout a distance of 18 mm. The press efficiency is 90% and is driven by a 1749-rpm motor. Determine average torque that must be provided by the motor in the units of N-m.arrow_forward·3) find the force (P) for the figures (1) and (2) 15cm 10cm 15 h=10mm h2=6mm // Call = 90 N/2 P Agate Fig (i) Ans: 1)P=112614N 2) P=1956.5 N 25cm 25 cm الفترة أو الحجم تمر بالتي عثر اكو تورشن (ک Fig (2) h₁ = 10mm 42=6mm Cmarrow_forward
 Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
