
Concept explainers
(a)
Interpretation: The product formed when the given compound in Problem 27.6 undergoes photochemical electrocyclic ring opening or ring closure is to be predicted. The given process is to be labeled as conrotatory or disrotatory. The stereochemistry around tetrahedral stereogenic centers and double bonds is to be indicated.
Concept introduction: Electrocyclic reactions involve the conversion of σ bonds into π bonds or π bonds into σ bonds. These reactions can take place in photochemical and as well as thermal conditions. The photochemical and thermal reactions take place in the presence of light and heat respectively.
Curved arrows aid in determining the movement and flow of electrons in the reaction. The electrons that take part in the
Answer to Problem 27.8P
The product formed in the given photochemical electrocyclic ring closure reaction is trans−4,5− dimethylcyclohexa −2,6− diene-1,2-dibenzene. The formation of the product takes place by conrotatory process. The stereochemistry around tetrahedral stereogenic centers is,
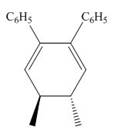
Explanation of Solution
The thermal electrocyclic ring closure reaction is shown below.
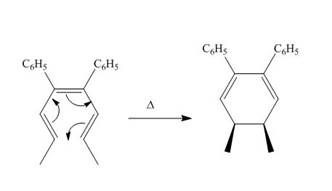
Figure 1
Electrocyclic reactions involve the conversion of σ bonds into π bonds or π bonds into σ bonds. In the product, the ring gets closed due to the rearrangement of π bonds and the formation of new σ bond. Therefore, the product formed in the given thermal electrocyclic ring closure reaction is cis−4,5− dimethylcyclohexa −2,6− diene-1,2-dibenzene.
Under photochemical condition, conrotatory process takes place in the given reaction. The orientation of substituents and stereochemistry around tetrahedral stereogenic centers and double bonds in the given reaction are shown below.
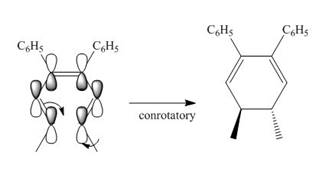
Figure 2
The conrotatory process involves the rotation of two orbitals in the same direction. The reaction in above figure implies that orbitals move in the same direction in the reactant molecule and the formation of trans product occurs. One methyl group is present above the plane and one methyl group is present below the plane in the product. Therefore, the product formed in the given photochemical electrocyclic ring closure reaction is trans−4,5− dimethylcyclohexa −2,6− diene-1,2-dibenzene.
The product formed in the given photochemical electrocyclic ring closure reaction is trans−4,5− dimethylcyclohexa −2,6− diene-1,2-dibenzene. The formation of the product takes place by conrotatory process. The stereochemistry around tetrahedral stereogenic centers is indicated in Figure 2.
(b)
Interpretation: The product formed when the given compound in Problem 27.6 undergoes photochemical electrocyclic ring opening or ring closure is to be predicted. The given process is to be labeled as conrotatory or disrotatory. The stereochemistry around tetrahedral stereogenic centers and double bonds is to be indicated.
Concept introduction: Electrocyclic reactions involve the conversion of σ bonds into π bonds or π bonds into σ bonds. These reactions can take place in photochemical and as well as thermal conditions. The photochemical and thermal reactions take place in the presence of light and heat respectively.
Curved arrows aid in determining the movement and flow of electrons in the reaction. The electrons that take part in the chemical reactions are shown by the curved arrows.
Answer to Problem 27.8P
The product formed in the given photochemical electrocyclic ring opening reaction is trans−3,4− dimethylhexa −2,4− diene. The formation of the product takes place by disrotatory process. The stereochemistry around tetrahedral stereogenic centers is,
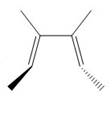
Explanation of Solution
The thermal electrocyclic ring opening reaction is shown below.
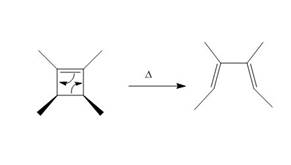
Figure 3
Electrocyclic reactions involve the conversion of σ bonds into π bonds or π bonds into σ bonds. In the product, the ring is opened due to the rearrangement of π bond and the breakdown of old σ bond. Therefore, the product formed in the given photochemical electrocyclic ring opening reaction is 3,4− dimethylhexa −2,4− diene.
Under photochemical condition, disrotatory process takes place in the given reaction. The movement of substituents and stereochemistry around tetrahedral stereogenic centers and double bonds in the given reaction are shown below.
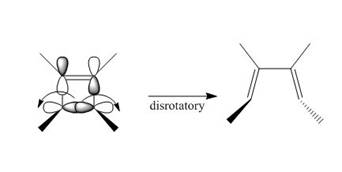
Figure 4
The disrotatory process involves the rotation of two orbitals in the opposite direction. The reaction in above figure implies that orbitals move in the opposite direction in the reactant molecule and the formation of trans product occurs. One methyl group is present above the plane and one methyl group is present below the plane in the product shows the stereochemistry. Therefore, the product formed in the given photochemical electrocyclic ring opening reaction is trans−3,4− dimethylhexa −2,4− diene. The formation of the product takes place by disrotatory process. The stereochemistry around tetrahedral stereogenic centers is indicated in Figure 4.
The product formed in the given photochemical electrocyclic ring opening reaction is trans−3,4− dimethylhexa −2,4− diene. The formation of the product takes place by disrotatory process. The stereochemistry around tetrahedral stereogenic centers is indicated in Figure 4.
Want to see more full solutions like this?
Chapter 27 Solutions
Organic Chemistry-Package(Custom)
- Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. :0: :0 H. 0:0 :0: :6: S: :0: Select to Edit Arrows ::0 Select to Edit Arrows H :0: H :CI: Rotation Select to Edit Arrows H. < :0: :0: :0: S:arrow_forward3:48 PM Fri Apr 4 K Problem 4 of 10 Submit Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. Mg. :0: Select to Add Arrows :0: :Br: Mg :0: :0: Select to Add Arrows Mg. Br: :0: 0:0- Br -190 H 0:0 Select to Add Arrows Select to Add Arrows neutralizing workup H CH3arrow_forwardIarrow_forward
- Draw the Markovnikov product of the hydrobromination of this alkene. Note for advanced students: draw only one product, and don't worry about showing any stereochemistry. Drawing dash and wedge bonds has been disabled for this problem. + Explanation Check 1 X E 4 1 1 1 1 1 HBr Click and drag to start drawing a structure. 80 LE #3 @ 2 $4 0 I அ2 % 85 F * K M ? BH 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center & 6 27 FG F10 8 9 R T Y U D F G H P J K L Z X C V B N M Q W A S H option command H command optiarrow_forwardBe sure to use wedge and dash bonds to show the stereochemistry of the products when it's important, for example to distinguish between two different major products. Predict the major products of the following reaction. Explanation Q F1 A Check F2 @ 2 # 3 + X 80 F3 W E S D $ 4 I O H. H₂ 2 R Pt % 05 LL ee F6 F5 T <6 G Click and drag to start drawing a structure. 27 & A 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center Acce Y U H DII 8 9 F10 4 J K L Z X C V B N M T H option command F11 P H commandarrow_forwardCurved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and follow the arrows to draw the intermediate and product in this reaction or mechanistic step(s). Include all lone pairs and charges as appropriate. Ignore stereochemistry. Ignore inorganic byproducts. H :0: CH3 O: OH Q CH3OH2+ Draw Intermediate protonation CH3OH CH3OH nucleophilic addition H Draw Intermediate deprotonation :0: H3C CH3OH2* protonation H 0: H CH3 H.arrow_forward
- Predicting the reactants or products of hemiacetal and acetal formation uentify the missing organic reactants in the following reaction: H+ X+Y OH H+ за Note: This chemical equation only focuses on the important organic molecules in the reaction. Additional inorganic or small-molecule reactants or products (like H2O) are not shown. In the drawing area below, draw the skeletal ("line") structures of the missing organic reactants X and Y. You may draw the structures in any arrangement that you like, so long as they aren't touching. Explanation Check Click and drag to start drawing a structure. ? olo 18 Ar © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibilityarrow_forwardcan someone please answer thisarrow_forwardPlease, please help me figure out the the moles, molarity and Ksp column. Step by step details because I've came up with about three different number and have no idea what I'm doing wrong.arrow_forward
