
Concept explainers
(a)
Interpretation:
The name of following molecule with appropriate stereochemical designation should be determined:
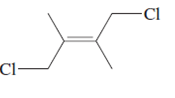
Concept introduction:
The E-configuration stands for anti-configuration, whereas, Z-configuration stands for same side configuration.
The determination of configuration is done on the basis of the
(b)
Interpretation:
The name of following molecule with appropriate stereochemical designation should be determined:
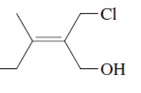
Concept introduction:
Alkenes are unsaturated hydrocarbons with double covalent bond between carbon-carbon atoms. On the basis of groups bonded with the double bonded carbon atoms, alkenes can be classified as E and Z-configuration.
The E-configuration stands for anti-configuration, whereas, Z-configuration stands for same side configuration.
The determination of configuration is done on the basis of the atomic/molecular mass of the atoms/groups attached to double bonded carbon atoms. If both higher atomic/molecular mass atom/groups are placed at the same side, then it is said to be Z-configuration and in E-configuration, these groups will be at anti-position.
(c)
Interpretation:
The name of following molecule with appropriate stereochemical designation should be determined:
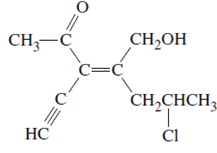
Concept introduction:
Alkenes are unsaturated hydrocarbons with double covalent bond between carbon-carbon atoms. On the basis of groups bonded with the double bonded carbon atoms, alkenes can be classified as E and Z-configuration.
The E-configuration stands for anti-configuration, whereas, Z-configuration stands for same side configuration.
The determination of configuration is done on the basis of the atomic/molecular mass of the atoms/groups attached to double bonded carbon atoms. If both higher atomic/molecular mass atom/groups are placed at the same side, then it is said to be Z-configuration and in E-configuration, these groups will be at anti-position.
(d)
Interpretation:
The name of following molecule with appropriate stereochemical designation should be determined:
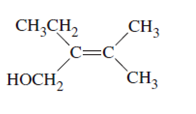
Concept introduction:
Alkenes are unsaturated hydrocarbons with double covalent bond between carbon-carbon atoms. On the basis of groups bonded with the double bonded carbon atoms, alkenes can be classified as E and Z-configuration.
The E-configuration stands for anti-configuration, whereas, Z-configuration stands for same side configuration.
The determination of configuration is done on the basis of the atomic/molecular mass of the atoms/groups attached to double bonded carbon atoms. If both higher atomic/molecular mass atom/groups are placed at the same side, then it is said to be Z-configuration and in E-configuration, these groups will be at anti-position.
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
General Chemistry: Principles And Modern Applications Plus Mastering Chemistry With Pearson Etext -- Access Card Package (11th Edition)
- Calculate the atomic packing factor of diamond knowing that the number of Si atoms per cm3 is 2.66·1022 and that the atomic radii of silicon and oxygen are, respectively, 0.038 and 0.117 nm.arrow_forwardA pdf file of your hand drawn, stepwise mechanisms for the reactions. For each reaction in the assignment, you must write each mechanism three times (there are 10 reactions, so 30 mechanisms). (A) do the work on a tablet and save as a pdf., it is expected to write each mechanism out and NOT copy and paste the mechanism after writing it just once. Everything should be drawn out stepwise and every bond that is formed and broken in the process of the reaction, and is expected to see all relevant lone pair electrons and curved arrows. Aldol: NaOH HO H Δ NaOH Δarrow_forwardNonearrow_forward
- Draw structures corresponding to the following names and give IUPAC names for the following compounds: (8 Point) a) b) c) CH3 CH2CH3 CH3CHCH2CH2CH CH3 C=C H3C H H2C=C=CHCH3 d) CI e) (3E,5Z)-2,6-Dimethyl-1,3,5,7-octatetraene f) (Z)-4-bromo-3-methyl-3-penten-1-yne g) cis-1-Bromo-2-ethylcyclopentane h) (5R)-4,4,5-trichloro-3,3-dimethyldecanearrow_forwardNonearrow_forwardReview: Design a total total synthesis synthesis of the following compound using methyloxacyclopropane and any other necessary reagents.arrow_forward
- Nonearrow_forwardDraw a Newman projection from carbon 3 to carbon 2 in the highest energy conformation for the following molecule. What is this conformation called? What kind of strain is present? Brarrow_forwardWhich of the following dienophiles is most reactive in a Diels-Alder reaction: Please explain why the correct answer to this question is option 5. Please provide a detailed explanation.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
