
Organic Chemistry
4th Edition
ISBN: 9780073402772
Author: Janice G. Smith
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 26, Problem 26.51P
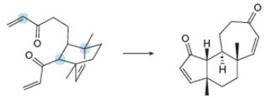 The following conversion, carried out in the presence of Grubbs catalyst and ethylene gas, involves a cascade of metathesis reactions. Draw a reaction sequence that illustrates how the reactant is converted to the product Z, and indicate where each labeled atom in the reactant ends up in Z.
The following conversion, carried out in the presence of Grubbs catalyst and ethylene gas, involves a cascade of metathesis reactions. Draw a reaction sequence that illustrates how the reactant is converted to the product Z, and indicate where each labeled atom in the reactant ends up in Z.
Z
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
what temperature does a 50% (mole
fraction) of ammonia/water liquid
mixture boil at 1 atm
1) Suppose 0.1 kg ice at 0°C (273K) is in 0.5kg water at 20°C (293K). What is the change in entropy of the ice as it melts at 0°?
To produce the original "water gas" mixture, carbon (in a combustible form known as coke) is reacted with steam: 131.4 kJ + H20(g) + C(s) → CO(g) + H2(g) From this information and the equations in the previous problem, calculate the enthalpy for the combustion or carbon to form carbon dioxide.
kindly show me how to solve both parts of the same long problem. Thanks
we were assigned to dilute 900ppm
in to 18ppm by using only 250ml vol
flask. firstly we did calc and convert
900ppm to 0.9 ppm to dilute in 1 liter.
to begin the experiment we took
0,225g of kmno4 and dissolved in to
250 vol flask. then further we took 10
ml sample sol and dissolved in to 100
ml vol flask and put it in to a
spectrometer and got value of 0.145A
.
upon further calc we got v2 as 50ml
. need to find DF, % error (expval and
accptVal), molarity, molality. please
write the whole report. thank you
The format, tables, introduction,
procedure and observation, result,
calculations, discussion and
conclusion
Chapter 26 Solutions
Organic Chemistry
Ch. 26 - Prob. 26.1PCh. 26 - Prob. 26.2PCh. 26 - Synthesize each product from the given starting...Ch. 26 - Prob. 26.4PCh. 26 - Prob. 26.5PCh. 26 - Prob. 26.6PCh. 26 - Prob. 26.7PCh. 26 - Problem 26.8
What starting materials are needed to...Ch. 26 - Prob. 26.9PCh. 26 - Prob. 26.10P
Ch. 26 - Problem 26.11
What product is formed when each...Ch. 26 - Prob. 26.12PCh. 26 - Problem 26.13
Draw the products formed when each...Ch. 26 - Problem 26.14
What products are formed when ...Ch. 26 - Prob. 26.15PCh. 26 -
What product is formed by ring-closing metathesis...Ch. 26 - Problem 26.17
What starting material is needed to...Ch. 26 - 26.18 In addition to organic halides, alkyl...Ch. 26 - 26.19 What product is formed by ring-closing...Ch. 26 - Draw the products formed in each reaction.Ch. 26 - Prob. 26.21PCh. 26 - 26.22 How can you convert ethynylcyclohexane to...Ch. 26 - 26.23 What compound is needed to convert styrene...Ch. 26 - 26.24 What steps are needed to convert to octane...Ch. 26 - Draw the products including stereoisomers formed...Ch. 26 - 26.28 Treatment of cyclohexene with and forms...Ch. 26 - Prob. 26.27PCh. 26 - 26.30 What starting material is needed to prepare...Ch. 26 - Prob. 26.29PCh. 26 - Prob. 26.30PCh. 26 - Prob. 26.31PCh. 26 - Draw the products formed in each reaction. a. f....Ch. 26 - Prob. 26.33PCh. 26 - Draw a stepwise mechanism for the following...Ch. 26 - Sulfur ylides, like the phosphorus ylides of...Ch. 26 - Although diazomethane is often not a useful...Ch. 26 - Prob. 26.37PCh. 26 - Prob. 26.38PCh. 26 - Prob. 26.39PCh. 26 - Prob. 26.40PCh. 26 - 26.43 Devise a synthesis of each compound using a...Ch. 26 - Devise a synthesis of each compound from...Ch. 26 - Devise a synthesis of each compound from benzene....Ch. 26 - Devise a synthesis of each substituted...Ch. 26 - Biaryls, compounds containing two aromatic rings...Ch. 26 - Prob. 26.46PCh. 26 - 26.49 Draw the product formed from the...Ch. 26 - Prob. 26.48PCh. 26 - Devise a synthesis of each of the following...Ch. 26 - Prob. 26.50PCh. 26 - 26.53 The following conversion, carried out in the...Ch. 26 - Prob. 26.52PCh. 26 - 26.55 Dimethyl cyclopropanes can be prepared by...Ch. 26 - Prob. 26.54P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Q5. Predict the organic product(s) for the following transformations. If no reaction will take place (or the reaction is not synthetically useful), write "N.R.". Determine what type of transition state is present for each reaction (think Hammond Postulate). I Br₂ CH3 F2, light CH3 Heat CH3 F₂ Heat Br2, light 12, light CH3 Cl2, light Noarrow_forwardNonearrow_forwardIn the phase diagram of steel (two components Fe and C), region A is the gamma austenite solid and region B contains the gamma solid and liquid. Indicate the degrees of freedom that the fields A and B have,arrow_forward
- For a condensed binary system in equilibrium at constant pressure, indicate the maximum number of phases that can exist.arrow_forwardPart V. Label ad match the carbons in compounds Jane and Diane w/ the corresponding peak no. in the Spectra (Note: use the given peak no. To label the carbons, other peak no are intentionally omitted) 7 4 2 -0.13 -0.12 -0.11 -0.10 -0.08 8 CI Jane 1 -0.09 5 210 200 190 180 170 160 150 140 130 120 110 100 -8 90 f1 (ppm) 11 8 172.4 172.0 f1 (ppr HO CI NH Diane 7 3 11 80 80 -80 -R 70 60 60 2 5 -8 50 40 8. 170 160 150 140 130 120 110 100 90 -0 80 70 20 f1 (ppm) 15 30 -20 20 -60 60 -0.07 -0.06 -0.05 -0.04 -0.03 -0.02 -0.01 -0.00 -0.01 10 -0.17 16 15 56 16 -0.16 -0.15 -0.14 -0.13 -0.12 -0.11 -0.10 -0.09 -0.08 -0.07 -0.06 -0.05 -0.04 17.8 17.6 17.4 17.2 17.0 f1 (ppm) -0.03 -0.02 550 106 40 30 20 20 -0.01 -0.00 F-0.01 10 0arrow_forwardConsider the reaction of 2-methylpropane with a halogen. With which halogen will the product be almost exclusively 2-halo-2-methylpropane? 1. F2 2. Cl2 3. Br2 4. I2arrow_forward
- Nonearrow_forwardNonearrow_forwardn Feb 3 A T + 4. (2 pts) Draw the structure of the major component of the Limonene isolated. Explain how you confirmed the structure. 5. (2 pts) Draw the fragment corresponding to the base peak in the Mass spectrum of Limonene. 6. (1 pts) Predict the 1H NMR spectral data of R-Limonene. Proton NMR: 5.3 pon multiplet (H Ringarrow_forward
- Part VI. Ca H 10 O is the molecular formula of compound Tom and gives the in the table below. Give a possible structure for compound Tom. 13C Signals summarized C1 C2 C3 C4 C5 C6 C7 13C shift (ppm) 23.5 27.0 33.0 35.8 127 162 205 DEPT-90 + DEPT-135 + +arrow_forward2. Using the following data to calculate the value of AvapH o of water at 298K. AvapH o of water at 373K is 40.7 kJ/mol; molar heat capacity of liquid water at constant pressure is 75.2J mol-1 K-1 and molar heat capacity of water vapor at constant pressure is 33.6 J mol-1 K-1.arrow_forwardPart VII. Below are the 'HNMR 13 3 C-NMR, COSY 2D- NMR, and HSQC 20-NMR (Similar with HETCOR but axes are reversed) spectra of an organic compound with molecular formula C6H13 O. Assign chemical shift values to the H and c atoms of the compound. Find the structure. Show complete solutions. Predicted 1H NMR Spectrum ли 4.7 4.6 4.5 4.4 4.3 4.2 4.1 4.0 3.9 3.8 3.7 3.6 3.5 3.4 3.3 3.2 3.1 3.0 2.9 2.8 2.7 2.6 2.5 2.4 2.3 2.2 2.1 2.0 1.9 1.8 1.7 1.6 1.5 1.4 1.3 1.2 1.1 1.0 0.9 0.8 f1 (ppm)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
GCSE Chemistry - Differences Between Compounds, Molecules & Mixtures #3; Author: Cognito;https://www.youtube.com/watch?v=jBDr0mHyc5M;License: Standard YouTube License, CC-BY