
(a)
Interpretation:
The working method of ion mobility spectrometry should be describes and analogies between gas-phase electrophoresis and capillary electrophoresis should be given.
Concept introduction:
Ion mobility spectrometry:
The electrophoretic mobility is given by the constant of proportionality between the electric field strength and speeds of the ion.
Capillary electrophoresis:
The movement of solute particle respect with the applied electric field is known as electrophoresis, this electrophoresis is produced in capillary tube is known as capillary electrophoresis.
(a)
Explanation of Solution
To describe the working method of ion mobility spectrometry.
The analyte are converted into gaseous ions by irradiation of analyte plus reagent gas like acetone in air with
The produced ions are passed through a tube, which has the grid with short voltage to plus the ions.
The ions are experience the constant electrical field in the grid. The ions are detected by the detectors, which the time they are drift.
The speed of the ion respect with the applied constant short voltage of the grid and friction force of the tube are noted.
Electrophoretic mobility is the constant of proportionality between the electric field strength and speeds of the ion.
Here, f is the friction coefficient.
From the above collected data are plugged in above equation to find the electrophoretic mobility of ion in electrophoresis, the ions are separated by resulted electrophoretic mobility of ions.
To give the analogies between gas-phase electrophoresis and capillary electrophoresis.
The drift time in the capillary electrophoresis same as the capillary electrophoresis.
The applied electrical field is a driving force of ions to migrate source to detector in capillary electrophoresis and same as in gas-phase electrophoresis.
The mobility of the ion in gas-phase electrophoresis and capillary electrophoresis is governed by charge to size ration.
The smaller size higher charge ions are have greater mobility.
The retarding force of the liquids and gases in gas-phase electrophoresis and capillary electrophoresis is caused by collision with medium.
The working method of ion mobility spectrometry was describes and analogies between gas-phase electrophoresis and capillary electrophoresis was given.
(b)
Interpretation:
The shape of curve in graph of plate number (vs) voltage in ion mobility spectrometry should be explained and the disadvantage of
Concept introduction:
Ion mobility spectrometry:
The electrophoretic mobility is given by the constant of proportionality between the electric field strength and speeds of the ion.
The peak width of the Ion mobility spectrometry is given by,
Where,
From the above equation the plate number is,
Where,
(b)
Explanation of Solution
To describe the working method of ion mobility spectrometry.
To draw the graph plate number (vs) voltage in ion mobility spectrometry.
Given,
Temperature
The give data and give formula are plugged in the spread sheet and apply the spread sheet formatted formula to calculate the required values and to prepare a graph of N (vs) Voltage
To take a new spread sheet and type the heading, which is shown in figure 1 and enter the observed values in the respective columns as pre heading.
The spread sheet format formulas are entered in column A same as shown in figure 1.
The values of plate number N respect with voltage are manipulated by the formula, which is already entered in spread sheet.
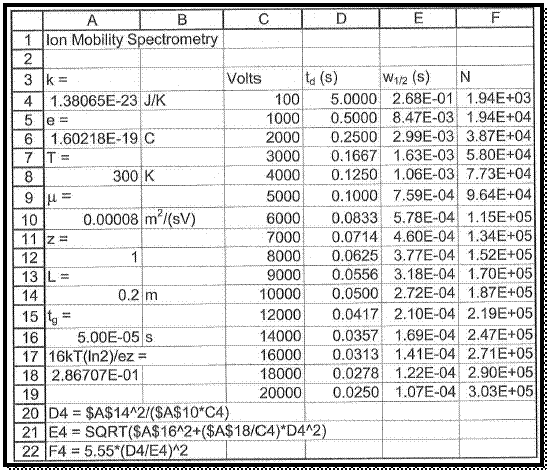
Figure 1
The graph is drawn by using values, which is in spread sheet.
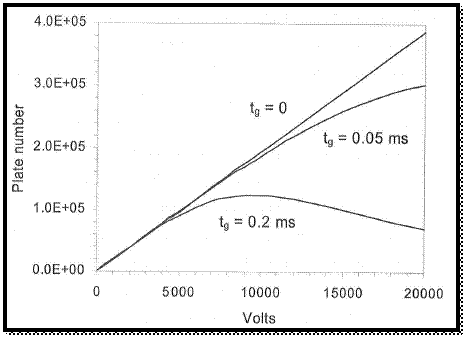
Figure 2
From the above graph, the applied voltage is increases the number of plats.
The broadening of peak is resulted by the decreasing of tome to diffusion and the initial peak width is increases with the increasing of gate open time of ion hence the plot number is decreased.
Therefore, the peak is not obtained in narrowed than the plus that admitted by the gate.
To give the disadvantage of
At the high voltage, the
The shape of curve in graph of plate number (vs) voltage in ion mobility spectrometry was explained and the disadvantage of
(c)
Interpretation:
The reason for the decreasing T increase N in ion mobility spectrometry should be given.
Concept introduction:
Ion mobility spectrometry:
The electrophoretic mobility is given by the constant of proportionality between the electric field strength and speeds of the ion.
The peak width of the Ion mobility spectrometry is given by,
Where,
From the above equation the plate number is,
Where,
(c)
Explanation of Solution
To give the reason for the decreasing T increase N in ion mobility spectrometry.
The peak width of the Ion mobility spectrometry is given by,
The equation for the plate number is,
From the above equation 1, the temperature is directly proportional peak width and in equation 2 the number of plates is indirectly proportional to the peak width. Therefore decreasing temperature is increasing number of plates because of broadening of peak is decreases with decreasing temperature.
The reason for the decreasing T increase N in ion mobility spectrometry was given.
(d)
Interpretation:
The theoretical plate number of given mobility spectrometry should be calculated.
Concept introduction:
Ion mobility spectrometry:
The electrophoretic mobility is given by the constant of proportionality between the electric field strength and speeds of the ion.
The peak width of the Ion mobility spectrometry is given by,
Where,
From the above equation the plate number is,
Where,
(d)
Answer to Problem 26.50P
The theoretical plate number is
Explanation of Solution
To calculate the theoretical plate number of given mobility spectrometry.
Given,
The equation for the plate number is,
The theoretical plate number is,
The calculated width of the peak and
The theoretical plate number is
The theoretical plate number of given ion mobility spectrometry was calculated.
(e)
Interpretation:
The resolution of the given two peaks of ion mobility spectrometry should be calculated.
Concept introduction:
Resolution:
The difference in the two peaks, which are obtained in the same experiment in same condition is given by resolution.
In the chromatographic separation, the resolution is,
Where,
(e)
Answer to Problem 26.50P
The resolution of the given two peaks of ion mobility spectrometry is
Explanation of Solution
To calculate the resolution of the given two peaks of ion mobility spectrometry.
Given,
Number of plates
Resolution of given two peaks is,
The difference in the apparent mobility of the ions, number of plates, and average apparent mobility are plugged in above equation to give the Resolution of given two peaks.
Resolution of given two peaks is
The resolution of the given two peaks of ion mobility spectrometry was calculated.
Want to see more full solutions like this?
Chapter 26 Solutions
Solution Manual for Quantitative Chemical Analysis
- dG = Vdp - SdT + μA dnA + μB dnB + ... so that under constant pressure and temperature conditions, the chemical potential of a component is the rate of change of the Gibbs energy of the system with respect to changing composition, μJ = (∂G / ∂nJ)p,T,n' Using first principles prove that under conditions of constant volume and temperature, the chemical potential is a measure of the partial molar Helmholtz energy (μJ = (∂A / ∂nJ)V,T,n')arrow_forwardThe vapor pressure of dichloromethane at 20.0 °C is 58.0 kPa and its enthalpy of vaporization is 32.7 kJ/mol. Estimate the temperature at which its vapor pressure is 66.0 kPa.arrow_forwardDraw the structure of A, the minor E1 product of the reaction. Cl Skip Part Check F1 esc CH_CH OH, D 3 2 Click and drag to start drawing a structure. 80 R3 F4 F2 F3 @ 2 # $ 4 3 Q W 95 % KO 5 F6 A F7 × G ☐ Save For Later Sub 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy C ►II A A F8 F9 F10 FL 6 7 88 & * 8 9 LLI E R T Y U A S D lock LL F G H 0 P J K L Z X C V B N M 9 Harrow_forward
- From the choices given, which two substances have the same crystal structure? (Select both) Group of answer choices ZnS (zincblende) Diamond TiO2 (rutile) ZnS (wurtzite)arrow_forwardPotassium (K) blends with germanium (Ge) to form a Zintl phase with a chemical formula of K4Ge4. Which of the following elements would you expect potassium to blend with to form an alloy? Electronegativities: As (2.0), Cl (3.0), Ge (1.8), K (0.8), S (2.5), Ti (1.5) Group of answer choices Arsenic (As) Sulfur (S) Chlorine (Cl) Titanium (Ti)arrow_forwardConsider two elements, X and Z. Both have cubic-based unit cells with the same edge lengths. X has a bcc unit cell while Z has a fcc unit cell. Which of the following statements is TRUE? Group of answer choices Z has a larger density than X X has more particles in its unit cell than Z does X has a larger density than Z Z has a larger unit cell volume than Xarrow_forward
- How many particles does a face-centered cubic (fcc) unit cell contain? Group of answer choices 2 14 8 4arrow_forwardV Highlight all of the carbon atoms that have at least one beta (B) hydrogen, using red for one ẞ hydrogen, blue for two ẞ hydrogens, and green for three ẞ hydrogens. If none of the carbon atoms have ẞ hydrogens, check the box underneath the molecule. ED X None of the carbon atoms have ẞ hydrogens. Explanation esc 2 Check * F1 F2 1 2 80 # 3 Q W tab A caps lock shift fn control F3 N S option O 694 $ F4 F5 F6 005 % E R D F LL 6 olo 18 Ar B © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility A DII F7 F8 87 & * 8 T Y U G H 4 F9 F10 ( 9 0 E F11 F12 உ J K L + || X C V B N M H H command option commandarrow_forwardConsider the reaction below and answer the following questions. Part 1 of 4 Br NaOCH2CH3 Identify the mechanisms involved. Check all that apply. SN 1 SN 2 E1 E2 None of the above Part 2 of 4 Skip Part Check esc F1 F2 lock 1 2 Q W A S #3 80 F3 F4 F5 F6 Save For © 2025 McGraw Hill LLC. All Rights Reserved. Terms ˇˇ % & 4 5 6 89 7 IK A 分 བ F7 F8 F9 F * E R T Y U 8 9 D F G H K V B N M 0 Oarrow_forward
- What kind of holes are not generated when solid-state particles adopt a close packing pattern? Group of answer choices tetrahedral cubic octahedral None of the other choices are correctarrow_forwardFor the reaction below: 1. Draw all reasonable elimination products to the right of the arrow. 2. In the box below the reaction, redraw any product you expect to be a major product. 田 Major Product: Check ☐ + I Na OH esc F1 F2 2 1 @ 2 Q W tab A caps lock S #3 80 F3 69 4 σ F4 % 95 S Click and drag to sta drawing a structure mm Save For Later 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use GO DII F5 F6 F7 F8 F9 F10 6 CO 89 & 7 LU E R T Y U 8* 9 0 D F G H J K L Z X C V B N M 36arrow_forwardProblem 7 of 10 Draw the major product of this reaction. Ignore inorganic byproducts. S' S 1. BuLi 2. ethylene oxide (C2H4O) Select to Draw a Submitarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





