
(a)
Interpretation: The formation of the products has to be found via an azo coupling reaction by treating with NaNO2 and HCl.
Concept Introduction:
Aryldiazonium salts on coupling with an activated
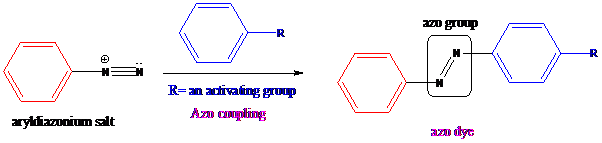
To find: Get the product via an azo coupling reaction by treating the compound (a) with NaNO2 and HCl
Find the nature of the given
(b)
Interpretation: The formation of the products has to be found via an azo coupling reaction by treating with NaNO2 and HCl.
Concept Introduction:
Aryldiazonium salts on coupling with an activated aromatic ring give an azo dye which produces a deep color in the final product. This color is due to the extended conjugation in it. This reaction occurs by electrophilic substitution reaction. This method is called an azo coupling. The obtained final compound is known as azo dyes.

To find: Get the product via an azo coupling reaction by treating the compound (b) with NaNO2 and HCl
Find the nature of the given amine (b)
(c)
Interpretation: The formation of the products has to be found via an azo coupling reaction by treating with NaNO2 and HCl.
Concept Introduction:
Aryldiazonium salts on coupling with an activated aromatic ring give an azo dye which produces a deep color in the final product. This color is due to the extended conjugation in it. This reaction occurs by electrophilic substitution reaction. This method is called an azo coupling. The obtained final compound is known as azo dyes.

To find: Get the product via an azo coupling reaction by treating the compound (c) with NaNO2 and HCl
Find the nature of the given amine (c)
(d)
Interpretation: The formation of the products has to be found via an azo coupling reaction by treating with NaNO2 and HCl.
Concept Introduction:
Aryldiazonium salts on coupling with an activated aromatic ring give an azo dye which produces a deep color in the final product. This color is due to the extended conjugation in it. This reaction occurs by electrophilic substitution reaction. This method is called an azo coupling. The obtained final compound is known as azo dyes.

To find: Get the product via an azo coupling reaction by treating the compound (d) with NaNO2 and HCl
Find the nature of the given amine (d)
Want to see the full answer?
Check out a sample textbook solution
Chapter 22 Solutions
ORGANIC CHEMISTRY(EBOOK)-W/WILEYPLUS
- How does the square root mean square velocity of gas molecules vary with temperature? Illustrate this relationship by plotting the square root mean square velocity of N2 molecules as a function of temperature from T=100 K to T=300 K.arrow_forwardDraw product B, indicating what type of reaction occurs. F3C CF3 NH2 Me O .N. + B OMearrow_forwardBenzimidazole E. State its formula. sState the differences in the formula with other benzimidazoles.arrow_forward
- Draw product A, indicating what type of reaction occurs. F3C CN CF3 K2CO3, DMSO, H₂O2 Aarrow_forward19) Which metal is most commonly used in galvanization to protect steel structures from oxidation? Lead a. b. Tin C. Nickel d. Zinc 20) The following molecule is an example of a: R₁ R2- -N-R3 a. Secondary amine b. Secondary amide c. Tertiary amine d. Tertiary amidearrow_forwardpls helparrow_forward
- pls helparrow_forward35) Complete the following equation by drawing the line the structure of the products that are formed. Please note that in some cases more than one product is possible. You must draw all possible products to recive full marks! a. ethanol + 2-propanol + H2SO4 → b. OH conc. H2SO4 CH2 H3C CH + K2Cr2O7 C. d. H3C A pressure CH3 + H2 CH Pt catalystarrow_forward21) The rate of reaction depends upon: a. the concentration and nature of reactants b. the temperature of the reaction C. whether or not a catalyst was used d. all of the above 22) A Maxwell-Boltzmann curve shows the distribution of molecular energies in a reaction system. When the temperature in this system is increased, the peak is a. higher and further to the right. b. higher and further to the left. c. lower and further to the right. d. lower and further to the left. 23) Which of the following correctly describes the reaction represented by the reaction below? CaCO3 (s) + energy → CaO (s) + CO2 (g) a. It is exothermic and the potential energy is greater in the reactants than the products. b. c. It is exothermic and the potential energy is greater in the products than the reactants. It is endothermic and the potential energy is greater in the products than the reactants. d. It is endothermic and the potential energy is equal for the products and reactants.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





