
Organic Chemistry
4th Edition
ISBN: 9780073402772
Author: Janice G. Smith
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 22, Problem 22.18P
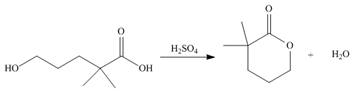
Draw a stepwise mechanism for the following reaction.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Thermodynamic analysis of electrified interfaces.
Conduct a brief thermodynamic analysis of electrified interfaces. Gibbs model.
ELECTROCAPILAR EQUATION FOR IDEALLY POLARIZED ELECTRODES.
Chapter 22 Solutions
Organic Chemistry
Ch. 22 - Prob. 22.1PCh. 22 - Draw the three possible resonance structures for...Ch. 22 - Prob. 22.3PCh. 22 - Give an IUPAC or common name for each compound.Ch. 22 - Problem 22.5 Draw the structure corresponding to...Ch. 22 - Prob. 22.6PCh. 22 - How would the compounds in each pair differ in...Ch. 22 - Problem 22.8 Deduce the structures of compounds ...Ch. 22 - Prob. 22.9PCh. 22 - Rank the compounds in each group in order of...
Ch. 22 - Prob. 22.11PCh. 22 - Prob. 22.12PCh. 22 - Prob. 22.13PCh. 22 - Prob. 22.14PCh. 22 - Problem 22.15 Draw the products of each...Ch. 22 - Problem 22.16 Draw the products of each reaction.
...Ch. 22 - Prob. 22.17PCh. 22 - Problem 22.18 Draw a stepwise mechanism for the...Ch. 22 - Prob. 22.19PCh. 22 - Problem 22.20 Fenofibrate is a...Ch. 22 - Problem 22.21 What product is formed when the...Ch. 22 - How would you synthesize olestra from sucrose?
Ch. 22 - What is the composition of the soap prepared by...Ch. 22 - Problem 22.24 Draw a stepwise mechanism for the...Ch. 22 - Prob. 22.25PCh. 22 - Problem 22.26 Some penicillins cannot be...Ch. 22 - Prob. 22.27PCh. 22 - Prob. 22.28PCh. 22 - Prob. 22.29PCh. 22 - Problem 22.30 Glucosamine is a dietry supplement...Ch. 22 - Draw the products of each reaction. a. c. b.Ch. 22 - Draw a tautomer of each compound.Ch. 22 - Draw the product of each reaction. a.b.Ch. 22 - Draw the product of each reaction. a. b.Ch. 22 - Prob. 22.35PCh. 22 - Problem 22.36 Outline two different ways that can...Ch. 22 - 22.37 Rank the following compounds in order of...Ch. 22 - Prob. 22.38PCh. 22 - Prob. 22.39PCh. 22 - Prob. 22.40PCh. 22 - Give thestructure corresponding to each name. a....Ch. 22 - Prob. 22.42PCh. 22 - Prob. 22.43PCh. 22 - 22.43 Explain why is a stronger acid and a weaker...Ch. 22 - Draw the product formed when pentanoyl chloride...Ch. 22 - Draw the product formed when pentanoic anhydride...Ch. 22 - Draw the product formed when phenylacetic acid is...Ch. 22 - Prob. 22.48PCh. 22 - Prob. 22.49PCh. 22 - Draw the product formed when phenylacetonitrile ...Ch. 22 - Prob. 22.51PCh. 22 - Prob. 22.52PCh. 22 - Prob. 22.53PCh. 22 - Prob. 22.54PCh. 22 - Prob. 22.55PCh. 22 - Prob. 22.56PCh. 22 - Draw a stepwise mechanism for each reaction. a. b.Ch. 22 - When acetic acid CH3COOH is treated with a trace...Ch. 22 - Prob. 22.59PCh. 22 - Prob. 22.60PCh. 22 - Prob. 22.61PCh. 22 - Prob. 22.62PCh. 22 - Prob. 22.63PCh. 22 - Draw a stepwise mechanism for the following...Ch. 22 - 22.63 Acid-catalyzed hydrolysis of forms compound...Ch. 22 - Prob. 22.66PCh. 22 - Devise a synthesis of each compound using...Ch. 22 - Convert 1-bromohexane (CH3CH2CH2CH2CH2CH2Br) into...Ch. 22 - Prob. 22.69PCh. 22 - Prob. 22.70PCh. 22 - Prob. 22.71PCh. 22 - Prob. 22.72PCh. 22 - Prob. 22.73PCh. 22 - Prob. 22.74PCh. 22 - Devise a synthesis of each labeled compound using...Ch. 22 - 22.70 What polyester or poly amide can be prepared...Ch. 22 - 22.71 What two monomers are needed to prepare each...Ch. 22 - Prob. 22.78PCh. 22 - How can IR spectroscopy be used to distinguish...Ch. 22 - Rank the compounds in each group in order of...Ch. 22 - 22.75 Identify the structures of each compound...Ch. 22 - 22.76 Identify the structures of A and B, isomers...Ch. 22 - Prob. 22.83PCh. 22 - 22.78 Identify the structure of compound C...Ch. 22 - 22.79 Identify the structures of D and E, isomers...Ch. 22 - 22.80 With reference to amides A and B, the...Ch. 22 - Prob. 22.87PCh. 22 - Prob. 22.88PCh. 22 - Prob. 22.89PCh. 22 - Draw a stepwise mechanism for the following...Ch. 22 - Draw a stepwise mechanism for the following...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Briefly state the electrocapillary equation for ideally polarized electrodes.arrow_forwardWhat is surface excess according to the Gibbs model?arrow_forwardUsing Benzene as starting materid show how each of the Following molecules Contel Ve syntheswed CHI 9. b -50311 с CHY 503H Ночто d. อ •NOV e 11-0-650 NO2arrow_forward
- The molecule PYRIDINE, 6th electrons and is therefore aromatre and is Assigned the Following structure contering Since aromatk moleculoy undergo electrophilic anomatic substitution, Pyridine shodd undergo The Following reaction + HNO3 12504 a. write all of the possible Mononitration Products that could Result From this reaction 18. Bared upon the reaction mechanison determime which of these producty would be the major Product of the hegetionarrow_forwarda. Explain Why electron withdrawing groups tend to be meta-Directors. Your answer Should lyclude all apropriate. Resonance contributing Structures fo. Explain why -ll is an outho -tura drccton even though chlorine has a very High Electronegativityarrow_forward9. Write Me product as well as the reaction Mechanism For each of the Following Vanctions +H₂504 4.50+ T C. +212 Fellz 237 b. Praw the potential energy Diagrams For each OF Mese Rauctions and account For any differences that appear in the two potential Puergy Diagrams which of here two reactions 19 Found to be Reversable, Rationalice your answer based upon the venation mechanisms and the potential energy diagrams.arrow_forward
- 9. Write Me product as well as the reaction Mechanism For each of the Following Veritious +H2504 4.50+ + 1/₂ Felly ◎+ 7 b. Praw he potential energy Diagrams For each OF Mese Ronctions and account for any differences that appeak in the two potential Puergy Diagramsarrow_forwardDraw the major product of this reaction. Ignore inorganic byproducts. Incorrect, 3 attempts remaining 1. excess Br2, NaOH 2. neutralizing workup Qarrow_forwardGiven the electrode Pt | Ag | Ag+ (aq), describe it.arrow_forward
- At 25°C, the reaction Zn2+ + 2e ⇄ Zn has a normal equilibrium potential versus the saturated calomel electrode of -1.0048 V. Determine the normal equilibrium potential of Zn versus the hydrogen electrode.Data: The calomel electrode potential is E° = 0.2420 V versus the normal hydrogen electrode.arrow_forwardElectrochemistry. State the difference between E and E0.arrow_forwardIn an electrolytic cell, the positive pole is always assumed to be on the right side of the battery notation. Is that correct?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY