
Concept explainers
Interpretation: Ethers and alcohols needs to be explained as structural isomers using exampled. The
Concept introduction: When two or more molecules have the same formula but shows different properties, they are termed as isomers. Molecules having same molecular formula but arrangement of atoms is different in space are termed as structural isomers.
Answer to Problem 36E
Ethers and alcohols are the structural isomers of one another which can be illustrated by considering a molecular formula
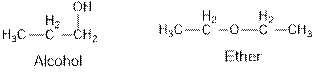
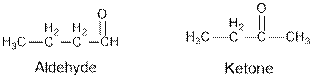
From the given table, esters are the structural isomers of carboxylic acids.
Explanation of Solution
Given information:
| Class | Functional Group | General Formula | Example |
| Halohaydrocarbon | −X (F, Cl, Br, I) | R−X | |
| Alcohols | −OH | R−OH | |
| Ethers | −O− | R−O−R | |
| Aldehydes | 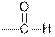 | 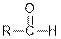 | |
| Ketones | 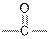 | 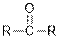 | |
| Carboxylic acids | 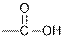 | 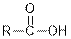 | |
| Esters | 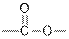 | 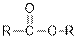 | |
| Amines |
Molecules having same molecular formula but different arrangement of atoms in space are termed as structural isomers.
Ethers and alcohols are the structural isomers of each other which can be illustrated by considering a molecular formula
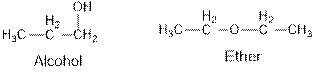
Aldehydes and ketones are the structural isomers of one another which can be illustrated by considering a molecular formula
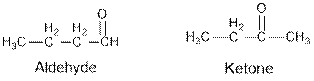
From the given table, esters are the structural isomers of carboxylic acids which can be illustrated by considering a molecular formula
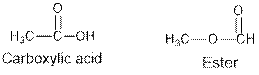
Ethers and alcohols are the structural isomers of each other which can be illustrated by considering a molecular formula
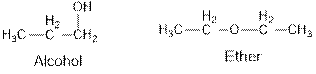
Aldehydes and ketones are the structural isomers of one another which can be illustrated by considering a molecular formula
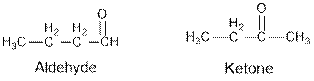
From the given table, esters are the structural isomers of carboxylic acids.
Want to see more full solutions like this?
Chapter 21 Solutions
EBK WEBASSIGN FOR ZUMDAHL'S CHEMICAL PR
- Synthesize 2-Ethyl-3-methyloxirane from dimethyl(propyl)sulfonium iodide using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize 2-Hydroxy-2-phenylacetonitrile from phenylmethanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- Synthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIf possible, please provide the formula of the compound 3,3-dimethylbut-2-enal.arrow_forwardSynthesize 1,4-dibromobenzene from acetanilide (N-phenylacetamide) using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- Indicate the products obtained by mixing (3-oxo-3-phenylpropyl)triphenylphosphonium bromide with sodium hydride.arrow_forwardWe mix N-ethyl-2-hexanamine with excess methyl iodide and followed by heating with aqueous Ag2O. Indicate the major products obtained.arrow_forwardIndicate the products obtained by mixing acetophenone with iodine and NaOH.arrow_forward
- Indicate the products obtained by mixing 2-Propanone and ethyllithium and performing a subsequent acid hydrolysis.arrow_forwardIndicate the products obtained if (E)-2-butenal and 3-oxo-butanenitrile are mixed with sodium ethoxide in ethanol.arrow_forwardQuestion 3 (4 points), Draw a full arrow-pushing mechanism for the following reaction Please draw all structures clearly. Note that this intramolecular cyclization is analogous to the mechanism for halohydrin formation. COH Br + HBr Brarrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning  Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div





