
Concept explainers
(a)
Interpretation: The name of the following compound needs to be determined.
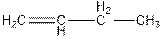
Concept Introduction : The name of an organic compound has three parts, prefix, parent chain and suffix. The format of naming is prefix+parent chain+suffix. The parent chain is the longest chain of carbon atoms in the compound. Suffix denote the name of
(a)
Answer to Problem 13E
The name of the compound is but-1-ene.
Explanation of Solution
The structure of the compound is,
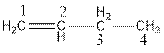
The parent chain consists of four carbon atoms. So, the name of parent chain is ‘but’. The compound contains one double bond so, suffix is ‘ene’. So, the name of the compound is but-1-ene.
(b)
Interpretation: The name of the following compound needs to be determined.
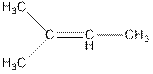
Concept Introduction : The name of an organic compound has three parts, prefix, parent chain and suffix. The format of naming is prefix+parent chain+suffix. The parent chain is the longest chain of carbon atoms in the compound. Suffix denote the name of functional group and prefix denote the substituent present in the compound.
(b)
Answer to Problem 13E
The name of the compound is 2-methylbut-2-ene.
Explanation of Solution
The compound is as follows:
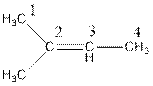
In the above compound, four carbon atoms present in the parent chain. So, the name of the parent chain is ‘but’. The compound consists of double bond so, the suffix is ‘ene’. There is one methyl substituent present at C2 so, the name of the compound is 2-methylbut-2-ene.
(c)
Interpretation: The name of the following compound needs to be determined.
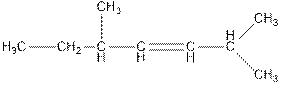
Concept Introduction : The name of an organic compound has three parts, prefix, parent chain and suffix. The format of naming is prefix+parent chain+suffix. The parent chain is the longest chain of carbon atoms in the compound. Suffix denote the name of functional group and prefix denote the substituent present in the compound.
(c)
Answer to Problem 13E
The name of the compound is 2, 5-dimethylhept-3-ene.
Explanation of Solution
The compound is as follows:
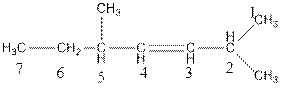
In the above compound, parent chain consists of seven carbon atoms. So, the name of parent chain is ‘hept’ and there is one double bond at C3 so, suffix is ‘ene’. There are two methyl groups present at C2 and C5 so, the name of the compound is 2,5-dimethylhept-3-ene.
(d)
Interpretation: The name of the following compound needs to be determined.
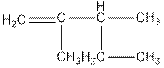
Concept Introduction: The name of an organic compound has three parts, prefix, parent chain and suffix. The format of naming is prefix+parent chain+suffix. The parent chain is the longest chain of carbon atoms in the compound. Suffix denote the name of functional group and prefix denote the substituent present in the compound.
(d)
Answer to Problem 13E
The name of the compound is 2,3-dimethylpent-1-ene.
Explanation of Solution
The compound is as follows:
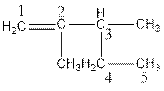
In the compound, five carbon atoms present in the parent chain. So, the name of parent chain is ‘pent’ and one double bond present in C1 so, suffix ‘ene’ should be used. Two methyl groups present as substituent in C2 and C3. So, the name of the compound is 2,3-dimethylpent-1-ene.
(e)
Interpretation: The name of the following compound needs to be determined.
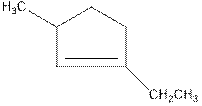
Concept Introduction: In naming of cyclic compound, naming is done in the format,
Prefix+Cyclo+parent chain+suffix
Prefix denotes the name of the substituent. If more than one substituent are present, they should be arranged in alphabetical order.
(e)
Answer to Problem 13E
The name of the compound is 1-ethyl-3-methylcyclopent-1-ene.
Explanation of Solution
The compound is as follows:
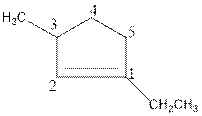
Above compound is a cyclic compound. The cyclic ring consists of five carbon atoms. So, the name of parent chain is ‘cyclopent’. One double bond is present in C1 so, suffix ‘ene’ should be used. One ethyl substituent is present in C1 and one methyl substituent is present in C3. So, the name of the compound is 1-ethyl-3-methylcyclopent-1-ene.
(f)
Interpretation: The name of the following compound needs to be determined.
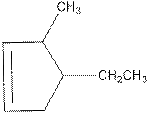
Concept Introduction: In naming of cyclic compound, naming is done in the format,
Prefix+Cyclo+parent chain+suffix
Prefix denotes the name of the substituent. If more than one substituent are present, they should be arranged in alphabetical order.
(f)
Answer to Problem 13E
The name of the compound is 4-ethyl-3-methylcyclopent-1-ene.
Explanation of Solution
The compound is as follows:
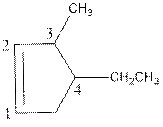
Above compound is a cyclic compound. The cyclic ring consists of five carbon atoms. So, the name of parent chain is ‘cyclopent’. One double bond is present in C1and C2 so, suffix ‘ene’ should be used. One ethyl substituent is present in C4 and one methyl substituent is present in C3. So, the name of the compound is 4-ethyl-3-methylcyclopent-1-ene.
(g)
Interpretation: The name of the following compound needs to be determined.
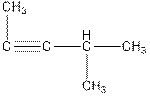
Concept Introduction: The name of an organic compound has three parts, prefix, parent chain and suffix. The format of naming is prefix+parent chain+suffix. The parent chain is the longest chain of carbon atoms in the compound. Suffix denotes the name of functional group and prefix denote the substituent present in the compound.
(g)
Answer to Problem 13E
The name of the compound is 4-methylpent-2-yne.
Explanation of Solution
The compound is as follows:
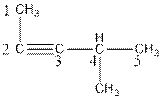
The parent chain of the compound consist of five carbon atoms, so, the name of parent chain is ‘pent’ and one triple bond is present in C2-C3, so, the suffix will be ‘yne’. One methyl substituent is present in C4. So, the name of the compound is 4-methylpent-2-yne.
Want to see more full solutions like this?
Chapter 21 Solutions
EBK WEBASSIGN FOR ZUMDAHL'S CHEMICAL PR
- Use the Henderson-Hasselbalch equation to calculate pH of a buffer containing 0.050M benzoic acidand 0.150M sodium benzoate. The Ka of benzoic acid is 6.5 x 10-5arrow_forwardA. Draw the structure of each of the following alcohols. Then draw and name the product you would expect to produce by the oxidation of each. a. 4-Methyl-2-heptanol b. 3,4-Dimethyl-1-pentanol c. 4-Ethyl-2-heptanol d. 5,7-Dichloro-3-heptanolarrow_forwardWhat is the pH of a 1.0 L buffer made with 0.300 mol of HF (Ka = 6.8 × 10⁻⁴) and 0.200 mol of NaF to which 0.160 mol of NaOH were added?arrow_forward
- Can I please get help with this.arrow_forwardDetermine if the following salt is neutral, acidic or basic. If acidic or basic, write the appropriate equilibrium equation for the acid or base that exists when the salt is dissolved in aqueous solution. If neutral, simply write only NR. Be sure to include the proper phases for all species within the reaction. N₂H₅ClO₄arrow_forwardPlease help me with identifying these.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





