
University Physics Volume 2
18th Edition
ISBN: 9781938168161
Author: OpenStax
Publisher: OpenStax
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 69P
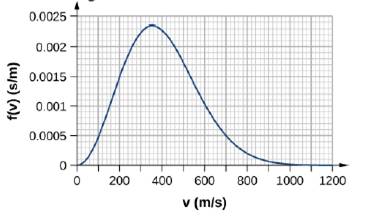
By counting squares in the following figure, estimate the fraction of argon atoms at T = 300 K that have speeds between 600 m/s and 800 m/s. The curve is correctly normalized. The value of a square is its length as measured on the x-axis times its height as measured on the y-axis, with the units given on those axes.
 `
`
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
When violet light of wavelength 415 nm falls on a single slit, it creates a central diffraction peak that is 8.60
cm wide on a screen that is 2.80 m away.
Part A
How wide is the slit?
ΟΙ ΑΣΦ
?
D= 2.7.10-8
Submit Previous Answers Request Answer
× Incorrect; Try Again; 8 attempts remaining
m
Two complex values are z1=8 + 8i, z2=15 + 7 i. z1∗ and z2∗ are the complex conjugate values.
Any complex value can be expessed in the form of a+bi=reiθ. Find θ for (z1-z∗2)/z1+z2∗. Find r and θ for (z1−z2∗)z1z2∗ Please show all steps
Calculate the center of mass of the hollow cone
shown below. Clearly specify the origin and the
coordinate system you are using.
Z
r
Y
h
X
Chapter 2 Solutions
University Physics Volume 2
Ch. 2 - Check Your Understanding The recommended daily...Ch. 2 - Check Understanding The density of in a Classroom...Ch. 2 - Check Your Understanding Liquids and solids have...Ch. 2 - Check Your Understanding If you consider a very...Ch. 2 - Check Your Understanding Which has a longer mean...Ch. 2 - Check Your Understanding Suppose 2 moles of helium...Ch. 2 - Two H2 molecules can react with one O2 molecule to...Ch. 2 - Under what circumstances would you expect a gas to...Ch. 2 - A constant-volume gas thermometer contains a fixed...Ch. 2 - Inflate a balloon at room temperature. Leave the...
Ch. 2 - In the last chapter, free convection was explained...Ch. 2 - How is momentum related to the pressure exerted by...Ch. 2 - If one kind of molecule has double the radius of...Ch. 2 - What is the average velocity of the air molecules...Ch. 2 - Why do the atmospheres of Jupiter, Saturn, Uranus,...Ch. 2 - Statistical mechanics says that in a gas...Ch. 2 - Which is more dangerous, a closet where tanks of...Ch. 2 - Experimentally it appears that many polyatomic...Ch. 2 - One might think that the internal energy of...Ch. 2 - You mix 5 moles of H2 at 300 K with 5 moles of He...Ch. 2 - One cylinder contains helium gas and another...Ch. 2 - Repeat the previous question if one gas is still...Ch. 2 - An ideal gas is at a temperature of 300 K. To...Ch. 2 - The gauge pressure in your car tires is...Ch. 2 - Suppose a gas-filled incandescent light bulb is...Ch. 2 - People buying food in sealed bags at high...Ch. 2 - How many moles are there in (a) 0.0500 g of N2 gas...Ch. 2 - A cubic container of volume 2.00 L holds 0.500 mol...Ch. 2 - Calculate the number of moles in the 2.00-L volume...Ch. 2 - An airplane passenger has 100 cm3 of air in his...Ch. 2 - A company advertises that it delivers helium at a...Ch. 2 - According to...Ch. 2 - An expensive vacuum system can achieve a pressure...Ch. 2 - The number density N/V of gas molecules at a...Ch. 2 - A bicycle tire contains 2.00 L of gas at an...Ch. 2 - In a common demonstration, a bottle is heated and...Ch. 2 - A high-pressure gas cylinder contains 50.0 L of...Ch. 2 - Find the number of moles in 2.00 L of gas at 35.0 ...Ch. 2 - Calculate the depth to which Avogadro's number of...Ch. 2 - (a) What is the gauge pressure in a 25.0 cc car...Ch. 2 - A person hits a tennis ball with a mass of 0.058...Ch. 2 - A person is in a closed room (a racquetball court)...Ch. 2 - Five bicyclists are riding at the following...Ch. 2 - Some incandescent light bulbs are filled with...Ch. 2 - Typical molecular speeds (vrms) are large, even at...Ch. 2 - What is the average kinetic energy in joules of...Ch. 2 - What is the ratio of the average translational...Ch. 2 - What is the total translational kinetic energy of...Ch. 2 - The product of the pressure and volume of a sample...Ch. 2 - What is the gauge pressure inside a tank of...Ch. 2 - If the rms speed of oxygen molecules inside a...Ch. 2 - The escape velocity of any object from Earth is...Ch. 2 - The escape velocity from the Moon is much smaller...Ch. 2 - Nuclear fusion, the energy solute of Sun, hydrogen...Ch. 2 - Suppose that the typical speed (vrms) of carbon...Ch. 2 - (a) Hydrogen molecules (molar mass is equal to...Ch. 2 - There are two important isotopes of uranium, U235...Ch. 2 - The partial pressure of carbon dioxide in the...Ch. 2 - Dry air consists of approximately 78% nitrogen,...Ch. 2 - (a) Using data from the previous problem, find the...Ch. 2 - (a) Given that air is 21% oxygen, find the minimum...Ch. 2 - (a) If the partial pressure of water vapor is 8.05...Ch. 2 - To give a helium atom nonzero angular momentum...Ch. 2 - (a) How much heat must be added to raise the...Ch. 2 - A sealed, rigid container of 0.560 mol of an...Ch. 2 - A sample of neon gas (Ne, molar mass M=20.2 g/mol)...Ch. 2 - A steel container of mass 135 g contains 24.0 g of...Ch. 2 - A sealed room has a volume of 24 m3. It's filled...Ch. 2 - Heliox, a mixture of helium and oxygen, is...Ch. 2 - Professional divas sometimes use heliox,...Ch. 2 - In car racing, one advantage of mixing liquid...Ch. 2 - In a sample of hydrogen sulfide ( M=34.1 g/mol) at...Ch. 2 - Using the approximation v1v1+v f(v)dvf(v1)v for...Ch. 2 - Using the method of the preceding problem,...Ch. 2 - By counting squares in the following figure,...Ch. 2 - Using a numerical integration method such as...Ch. 2 - Find (a) the most probable speed, (b) the average...Ch. 2 - Repeat the preceding problem for nitrogen...Ch. 2 - At what temperature is the average speed of carbon...Ch. 2 - The most probable speed for molecules of a gas at...Ch. 2 - a) At what temperature do oxygen molecules have...Ch. 2 - In the deep space between galaxies, the density of...Ch. 2 - (a) Find the density in SI units of air at a...Ch. 2 - The air inside a hot-air balloon has a temperature...Ch. 2 - When an air bubble rises from the bottom to the...Ch. 2 - (a) Use the ideal gas equation to estimate the...Ch. 2 - One process for decaffeinating coffee uses carbon...Ch. 2 - On a winter day when the air temperature is 0 ,...Ch. 2 - On a warm day when the air temperature is 30 , a...Ch. 2 - (a) People often think of humid air as "heavy."...Ch. 2 - The mean flee path for helium at a certain...Ch. 2 - The mean free path for methane at a temperature of...Ch. 2 - In the chapter on fluid mechanics, Bernoulli's...Ch. 2 - Find the total number of collisions between...Ch. 2 - (a) Estimate the specific heat capacity of sodium...Ch. 2 - A sealed, perfectly insulated container contains...Ch. 2 - Find the ratio f(vp)/f(vrms) for hydrogen gas (...Ch. 2 - Unreasonable results. (a) Find the temperature of...Ch. 2 - Unreasonable results. (a) Find the sped of...Ch. 2 - An airtight dispenser for drinking water is 25 cm...Ch. 2 - Eight bumper cars, each with a mass of 322 kg. are...Ch. 2 - Verify that vp=2kBTm.`Ch. 2 - Verify the normalization equation 0f(v)dv=1 In...Ch. 2 - Verify that v=8kBTm. Make the same scaling...Ch. 2 - Verify that vrms=v2=3kBTm.`
Additional Science Textbook Solutions
Find more solutions based on key concepts
28. Consider the reaction
Express the rate of the reaction in terms of the change in concentration of e...
Chemistry: Structure and Properties (2nd Edition)
Which one of the following is not a fuel produced by microorganisms? a. algal oil b. ethanol c. hydrogen d. met...
Microbiology: An Introduction
9. Blocks with masses of 1 kg, 2 kg, and 3 kg are lined up in a row on a frictionless table. All three are push...
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
11. The conducting loop in Figure Q25.11 is moving into the region between the magnetic poles shown.
a. Is the ...
College Physics: A Strategic Approach (3rd Edition)
What name is given to the zone of greatest seismic activity?
Applications and Investigations in Earth Science (9th Edition)
An obese 55-year-old woman consults her physician about minor chest pains during exercise. Explain the physicia...
Biology: Life on Earth with Physiology (11th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- 12. If all three collisions in the figure below are totally inelastic, which will cause more damage? (think about which collision has a larger amount of kinetic energy dissipated/lost to the environment? I m II III A. I B. II C. III m m v brick wall ע ע 0.5v 2v 0.5m D. I and II E. II and III F. I and III G. I, II and III (all of them) 2marrow_forwardCan you solve this 2 question teach me step by step and draw for mearrow_forwardFrom this question and answer can you explain how get (0,0,5) and (5,0,,0) and can you teach me how to solve thisarrow_forward
- Can you solve this 2 question and teach me using ( engineer method formula)arrow_forward11. If all three collisions in the figure below are totally inelastic, which brings the car of mass (m) on the left to a halt? I m II III m m ע ע ע brick wall 0.5v 2m 2v 0.5m A. I B. II C. III D. I and II E. II and III F. I and III G. I, II and III (all of them)arrow_forwardHow can you tell which vowel is being produced here ( “ee,” “ah,” or “oo”)? Also, how would you be able to tell for the other vowels?arrow_forward
- You want to fabricate a soft microfluidic chip like the one below. How would you go about fabricating this chip knowing that you are targeting a channel with a square cross-sectional profile of 200 μm by 200 μm. What materials and steps would you use and why? Disregard the process to form the inlet and outlet. Square Cross Sectionarrow_forward1. What are the key steps involved in the fabrication of a semiconductor device. 2. You are hired by a chip manufacturing company, and you are asked to prepare a silicon wafer with the pattern below. Describe the process you would use. High Aspect Ratio Trenches Undoped Si Wafer P-doped Si 3. You would like to deposit material within a high aspect ratio trench. What approach would you use and why? 4. A person is setting up a small clean room space to carry out an outreach activity to educate high school students about patterning using photolithography. They obtained a positive photoresist, a used spin coater, a high energy light lamp for exposure and ordered a plastic transparency mask with a pattern on it to reduce cost. Upon trying this set up multiple times they find that the full resist gets developed, and they are unable to transfer the pattern onto the resist. Help them troubleshoot and find out why pattern of transfer has not been successful. 5. You are given a composite…arrow_forwardTwo complex values are z1=8 + 8i, z2=15 + 7 i. z1∗ and z2∗ are the complex conjugate values. Any complex value can be expessed in the form of a+bi=reiθ. Find r and θ for (z1-z∗2)/z1+z2∗. Find r and θ for (z1−z2∗)z1z2∗ Please show all stepsarrow_forward
- An electromagnetic wave is traveling through vacuum in the positive x direction. Its electric field vector is given by E=E0sin(kx−ωt)j^,where j^ is the unit vector in the y direction. If B0 is the amplitude of the magnetic field vector, find the complete expression for the magnetic field vector B→ of the wave. What is the Poynting vector S(x,t), that is, the power per unit area associated with the electromagnetic wave described in the problem introduction? Give your answer in terms of some or all of the variables E0, B0, k, x, ω, t, and μ0. Specify the direction of the Poynting vector using the unit vectors i^, j^, and k^ as appropriate. Please explain all stepsarrow_forwardAnother worker is performing a task with an RWL of only 9 kg and is lifting 18 kg, giving him an LI of 2.0 (high risk). Questions:What is the primary issue according to NIOSH?Name two factors of the RWL that could be improved to reduce risk.If the horizontal distance is reduced from 50 cm to 30 cm, how does the HM change and what effect would it have?arrow_forwardTwo complex values are z1=8 + 8i, z2=15 + 7 i. z1∗ and z2∗ are the complex conjugate values. Any complex value can be expessed in the form of a+bi=reiθ. Find r and θ for z1z2∗. Find r and θ for z1/z2∗? Find r and θ for (z1−z2)∗/z1+z2∗. Find r and θ for (z1−z2)∗/z1z2∗ Please explain all steps, Thank youarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 University Physics Volume 1PhysicsISBN:9781938168277Author:William Moebs, Samuel J. Ling, Jeff SannyPublisher:OpenStax - Rice University
University Physics Volume 1PhysicsISBN:9781938168277Author:William Moebs, Samuel J. Ling, Jeff SannyPublisher:OpenStax - Rice University Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning


University Physics Volume 1
Physics
ISBN:9781938168277
Author:William Moebs, Samuel J. Ling, Jeff Sanny
Publisher:OpenStax - Rice University

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

Physics for Scientists and Engineers, Technology ...
Physics
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781285737027
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning