
Chemistry for Engineering Students
4th Edition
ISBN: 9781337398909
Author: Lawrence S. Brown, Tom Holme
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 2, Problem 2.29PAE
Interpretation Introduction
Interpretation:
To explain the advantages of using ion based catalyst in the
Concept introduction:
Polymerization:
Reaction that produces very large molecules consisting of many small repeating units.
Example:
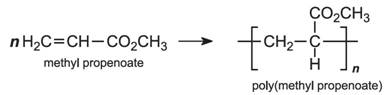
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
3. Predict the major product and give a mechanism for the following reactions:
(CH3)3COH/H₂SO4
a)
b)
NC
CH₂O
c)
LOCH,
(CH3)3COH/H2SO4
H,SO
-OH
Indicate if the aldehyde shown reacts with the provided nucleophiles in acid or base
conditions.
a NaBH4
be Li
eli
-NH2
P(Ph3)
f
KCN
g
OH excess
h CH3OH
i NaCHCCH3
Predict the major products of the following organic reaction:
+
A
?
Some important notes:
• Draw the major product, or products, of the reaction in the drawing area below.
• If there aren't any products, because no reaction will take place, check the box below the drawing area instead.
• Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are
enantiomers.
Explanation
Check
Click and drag to start drawing a structure.
C
© 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Cente
Chapter 2 Solutions
Chemistry for Engineering Students
Ch. 2 - Name at least three common polymers and give...Ch. 2 - Prob. 2COCh. 2 - Describe the nuclear model for the atom and...Ch. 2 - Prob. 4COCh. 2 - Prob. 5COCh. 2 - Prob. 6COCh. 2 - Prob. 7COCh. 2 - Prob. 8COCh. 2 - Prob. 9COCh. 2 - Prob. 10CO
Ch. 2 - Prob. 2.1PAECh. 2 - How do polymers compare to their respective...Ch. 2 - Look around you and identify several objects that...Ch. 2 - Prob. 2.4PAECh. 2 - The fact that a polymer’s physical properties...Ch. 2 - One application of conductive polymers is in...Ch. 2 - Prob. 2.7PAECh. 2 - Prob. 2.8PAECh. 2 - Why is the number of protons called the atomic...Ch. 2 - 2.10 Which isotope in each pair contains more...Ch. 2 - 2.11 Define the term isotope.Ch. 2 - 2.12 Write the complete atomic symbol for each of...Ch. 2 - 2.13 How many electrons, protons, and neutrons are...Ch. 2 - 2.14 Consider the following nuclear symbols. How...Ch. 2 - 2.15 Mercury is 16.716 times more massive than...Ch. 2 - The element gallium, used in gallium arsenide...Ch. 2 - 2.17 The atomic weight of copper is 63.55 amu....Ch. 2 - The following table presents the abundances and...Ch. 2 - 2.19 Naturally occurring uranium consists of two...Ch. 2 - Prob. 2.20PAECh. 2 - Prob. 2.21PAECh. 2 - 2.22 Provide the symbol of the following...Ch. 2 - Prob. 2.23PAECh. 2 - 2.24 Identify each of the following species as an...Ch. 2 - 2.25 Write the atomic symbol for the element whose...Ch. 2 - 2.26 In what region of the periodic table are you...Ch. 2 - Prob. 2.27PAECh. 2 - Prob. 2.28PAECh. 2 - Prob. 2.29PAECh. 2 - 2.30 Using Coulomb’s law, explain how the...Ch. 2 - Prob. 2.31PAECh. 2 - 2.32 Which of the following formulas contains the...Ch. 2 - Prob. 2.33PAECh. 2 - Prob. 2.34PAECh. 2 - Prob. 2.35PAECh. 2 - 2.36 Explain the difference between a molecular...Ch. 2 - 2.37 Why are empirical formulas preferred for...Ch. 2 - 2.38 The molecular formula for the ethylene...Ch. 2 - 239 Polybutadiene is a synthetic elastomer, or...Ch. 2 - 2.40 What distinguished the work of Mendeleev that...Ch. 2 - 2.41 How does the periodic table help to make the...Ch. 2 - 2.42 What is a period in the periodic table? From...Ch. 2 - 2.43 Name of the group to which each of the...Ch. 2 - Prob. 2.44PAECh. 2 - Prob. 2.45PAECh. 2 - 2.46 Why are nonmetals important even though they...Ch. 2 - Prob. 2.47PAECh. 2 - A materials engineer has filed for a patent for a...Ch. 2 - Prob. 2.49PAECh. 2 - 2.50 A materials engineer wants to make a new...Ch. 2 - Prob. 2.51PAECh. 2 - Prob. 2.52PAECh. 2 - 2.53 What is meant by the phrase organic...Ch. 2 - 2.54 Based on what you have learned in this...Ch. 2 - 2.55 What is a functional group? How does the...Ch. 2 - Prob. 2.56PAECh. 2 - Prob. 2.57PAECh. 2 - Prob. 2.58PAECh. 2 - 2.59 The accompanying figure shows the structure...Ch. 2 - Prob. 2.60PAECh. 2 - 2.61 Name the following covalent compounds: (a)...Ch. 2 - Prob. 2.62PAECh. 2 - Prob. 2.63PAECh. 2 - Prob. 2.64PAECh. 2 - Prob. 2.65PAECh. 2 - Prob. 2.66PAECh. 2 - Prob. 2.67PAECh. 2 - 2.68 What is a free radical? How are free radicals...Ch. 2 - Prob. 2.69PAECh. 2 - 2.70 Why do you think an inhibitor molecule is...Ch. 2 - 2.71 Use the web to determine the amount of...Ch. 2 - 2.72 How can an element have an atomic weight that...Ch. 2 - 2.73 Explain the concept of a “weighted” average...Ch. 2 - 2.74 The accompanying table provides the identity...Ch. 2 - 2.75 Chlorine has only two isotopes, one with mass...Ch. 2 - Prob. 2.76PAECh. 2 - Prob. 2.77PAECh. 2 - Prob. 2.78PAECh. 2 - Prob. 2.79PAECh. 2 - 2.80 Of the following elements, which two would...Ch. 2 - 2.81 How do binary compounds with hydrogen...Ch. 2 - Prob. 2.82PAECh. 2 - Prob. 2.83PAECh. 2 - 2.84 Early attempts to arrange the elements often...Ch. 2 - 2.85 Describe how the saying “opposites attract”...Ch. 2 - 2.86 For some uses, the relative abundance of...Ch. 2 - 2.87 What is the heaviest element to have an...Ch. 2 - 2.88 Describe how you can identify the isotope, X,...Ch. 2 - Prob. 2.89PAECh. 2 - 2.90 Naturally occurring europium has an average...Ch. 2 - 2.91 Strontium has four stable isotopes....Ch. 2 - 2.92 A candy manufacturer makes chocolate-covered...Ch. 2 - Prob. 2.93PAECh. 2 - 2.94 Use a molecular level description to...Ch. 2 - 2.95 Engineers who design bicycle frames are...Ch. 2 - 2.96 Use the web to look up the density of...Ch. 2 - 2.97 LDPE has a density in the range of...
Knowledge Booster
Similar questions
- Polar solutes are most likely to dissolve into _____, and _____ are most likely to dissolve into nonpolar solvents. A. nonpolar solutes; polar solvents B. nonpolar solvents; polar solvents C. polar solvents; nonpolar solutes D. polar solutes; nonpolar solventsarrow_forwardDeducing the Peactants Can the molecule on the right-hand side of this organic reaction be made in good yield from no more than two reactants, in one step, by moderately heating the reactants? ? Δ If your answer is yes, then draw the reactant or reactants in the drawing area below. You can draw the reactants in any arrangement you like. If your answer is no, check the box under the drawing area instead. Explanation Check Click and drag to start drawing a structure. © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center Xarrow_forwardDraw all 8 stereoisomers, circling each pair of enantiomer(s)/ mirror image compound(s)arrow_forward
- Bookmarks Profiles Tab Window Help Chemical Formula - Aktiv Che X + → C 11 a app.aktiv.com Google Chrome isn't your default browser Set as default Question 12 of 16 Q Fri Feb 2 Verify it's you New Chrome availabl- Write the balanced molecular chemical equation for the reaction in aqueous solution for mercury(I) nitrate and chromium(VI) sulfate. If no reaction occurs, simply write only NR. Be sure to include the proper phases for all species within the reaction. 3 Hg(NO3)2(aq) + Cг2(SO4)3(aq) → 3 Hg₂SO (s) + 2 Cr(NO3), (aq) ean Ui mate co ence an climate bility inc ulnerabili women, main critic CLIMATE-INI ernational + 10 O 2 W FEB 1 + 4- 3- 2- 2 2 ( 3 4 NS 28 2 ty 56 + 2+ 3+ 4+ 7 8 9 0 5 (s) (1) Ch O 8 9 (g) (aq) Hg NR CI Cr x H₂O A 80 Q A DII A F2 F3 FA F5 F6 F7 F8 F9 #3 EA $ do 50 % 6 CO & 7 E R T Y U 8 ( 9 0 F10 34 F11 川 F12 Subr + delete 0 { P }arrow_forwardDeducing the reactants of a Diels-Alder reaction n the molecule on the right-hand side of this organic reaction be made in good yield from no more than two reactants, in one step, by moderately heating the reactants? ? Δ • If your answer is yes, then draw the reactant or reactants in the drawing area below. You can draw the reactants in any arrangement you like. • If your answer is no, check the box under the drawing area instead. Explanation Check Click and drag to start drawing a structure. >arrow_forwardPredict the major products of the following organic reaction: + Some important notes: A ? • Draw the major product, or products, of the reaction in the drawing area below. • If there aren't any products, because no reaction will take place, check the box below the drawing area instead. • Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers. Explanation Check Click and drag to start drawing a structure.arrow_forward
- if the answer is no reaction than state that and please hand draw!arrow_forward"I have written solutions in text form, but I need experts to rewrite them in handwriting from A to Z, exactly as I have written, without any changes."arrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning