
Biochemistry
9th Edition
ISBN: 9781305961135
Author: Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 17RE
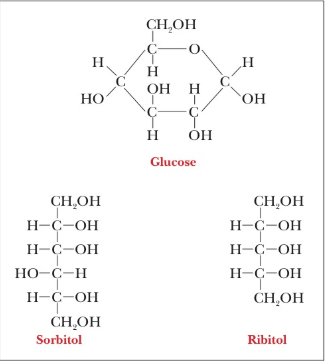
REFLECT AND APPLY How many water molecules could hydrogen-bond directly to the molecules of glucose, sorbitol, and ribitol, shown here?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
The rate data from an enzyme catalyzed reaction with and without an inhibitor present is found in the image.
Question: what is the KM and Vm and the nature of inhibition
1. Estimate the concentration of an enzyme within a living cell. Assume that:
(a): fresh tissue is 80% water and all of it is intracellular
(b): the total soluble protein represents 15% of the weight
(c): all the soluble proteins are enzymes
(d): the average molecular weight of the proteins is 150,000
(E): about 100 different enzymes are present
please help I am lost
Please help
Chapter 2 Solutions
Biochemistry
Ch. 2 - REFLECT AND APPLY Why is water necessary for life?Ch. 2 - REFLECT AND APPLY Contemplate biochemistry if...Ch. 2 - RECALL What is a van der Waals force?Ch. 2 - RECALL What is an induced dipole?Ch. 2 - RECALL What is a salt bridge?Ch. 2 - RECALL Under what circumstance is a molecule that...Ch. 2 - REFLECT AND APPLY Which would you think would be a...Ch. 2 - RECALL List the three types of van der Waals...Ch. 2 - RECALL A hydrogen bond is a special case of what...Ch. 2 - REFLECT AND APPLY Why do you think that most...
Ch. 2 - RECALL What are some macromolecules that have...Ch. 2 - BIOCHEMICAL CONNECTIONS How are hydrogen bonds...Ch. 2 - REFLECT AND APPLY Rationalize the fact that...Ch. 2 - REFLECT AND APPLY Draw three examples of types of...Ch. 2 - RECALL What are the requirements for molecules to...Ch. 2 - REFLECT AND APPLY Many properties of acetic acid...Ch. 2 - REFLECT AND APPLY How many water molecules could...Ch. 2 - REFLECT AND APPLY Both RNA and DNA have negatively...Ch. 2 - RECALL Identify the conjugate acids and bases in...Ch. 2 - RECALL Identify conjugate acids and bases in the...Ch. 2 - REFLECT AND APPLY Aspirin is an acid with a pKa of...Ch. 2 - RECALL Why does the pH change by one unit if the...Ch. 2 - MATHEMATICAL Calculate the hydrogen ion...Ch. 2 - MATHEMATICAL Calculate the hydrogen ion...Ch. 2 - MATHEMATICAL Calculate the hydroxide ion...Ch. 2 - RECALL Define the following: (a) Acid dissociation...Ch. 2 - REFLECT AND APPLY Look at Figure 2.17. If you did...Ch. 2 - BIOCHEMICAL CONNECTIONS List the criteria used to...Ch. 2 - BIOCHEMICAL CONNECTIONS What is the relationship...Ch. 2 - MATHEMATICAL What is the [CH3COO]/[CH3COOH] ratio...Ch. 2 - MATHEMATICAL What is the [CH3COO]/[CH3COOH] ratio...Ch. 2 - MATHEMATICAL What is the ratio of TRIS/TRIS-H+ in...Ch. 2 - MATHEMATICAL What is the ratio of HEPES/HEPES-H+...Ch. 2 - MATHEMATICAL How would you prepare 1 L of a 0.050...Ch. 2 - MATHEMATICAL The buffer needed for Question 35 can...Ch. 2 - MATHEMATICAL Calculate the pH of a buffer solution...Ch. 2 - MATHEMATICAL Calculate the pH of a buffer solution...Ch. 2 - MATHEMATICAL Calculate the pH of a buffer solution...Ch. 2 - MATHEMATICAL A catalog in the lab has a recipe for...Ch. 2 - MATHEMATICAL If you mix equal volumes of 0.1 M HCl...Ch. 2 - MATHEMATICAL What would be the pH of the solution...Ch. 2 - MATHEMATICAL If you have 100 mL of a 0.10 M TRIS...Ch. 2 - MATHEMATICAL What would be the pH of the solution...Ch. 2 - MATHEMATICAL Show that, for a pure weak acid in...Ch. 2 - MATHEMATICAL What is the ratio of concentrations...Ch. 2 - BIOCHEMICAL CONNECTIONS You need to carry out an...Ch. 2 - Prob. 48RECh. 2 - Prob. 49RECh. 2 - BIOCHEMICAL CONNECTIONS Which of the buffers shown...Ch. 2 - Prob. 51RECh. 2 - REFLECT AND APPLY In Section 2-4, we said that at...Ch. 2 - MATHEMATICAL Define buffering capacity. How do the...Ch. 2 - BIOCHEMICAL CONNECTIONS If you wanted to make a...Ch. 2 - BIOCHEMICAL CONNECTIONS We usually say that a...Ch. 2 - RECALL What quality of zwitterions makes them...Ch. 2 - Prob. 57RECh. 2 - Prob. 58RECh. 2 - Prob. 59RECh. 2 - BIOCHEMICAL CONNECTIONS A frequently recommended...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- The following data were recorded for the enzyme catalyzed conversion of S -> P. Question: Estimate the Vmax and Km. What would be the rate at 2.5 and 5.0 x 10-5 M [S] ?arrow_forwardPlease helparrow_forwardThe following data were recorded for the enzyme catalyzed conversion of S -> P Question: what would the rate be at 5.0 x 10-5 M [S] and the enzyme concentration was doubled? Also, the rate given in the table is from product accumulation after 10 minuets of reaction time. Verify these rates represent a true initial rate (less than 5% turnover). Please helparrow_forward
- The following data was obtained on isocitrate lyase from an algal species. Identify the reaction catalyzed by this enzyme, deduce the KM and Vmax , and determine the nature of the inhibition by oxaloacetate. Please helparrow_forwardIn the table below, there are sketches of four crystals made of positively-charged cations and negatively-charged anions. Rank these crystals in decreasing order of stability (or equivalently increasing order of energy). That is, select "1" below the most stable (lowest energy) crystal. Select "2" below the next most stable (next lowest energy) crystal, and so forth. A B 鹽 (Choose one) +2 C +2 +2 (Choose one) D 鹽雞 (Choose one) (Choose one)arrow_forward1. Draw the structures for the fats A. 16:2: w-3 and B. 18:3:49,12,15 2. Name each of the molecules below (image attached)arrow_forward
- draw the structures for the fats A. 16:2:w-3 B 18:3:9,12,15arrow_forward1. Below is a template strand of DNA. Show the mRNA and protein that would result. label the ends of the molecules ( refer to attached image)arrow_forwardAttach the followina labels to the diagram below: helicase, single stranded binding proteins, lagging strand, leading strand, DNA polymerase, primase, 5' ends (3), 3' ends (3) (image attached)arrow_forward
- 1. How much energy in terms of ATP can be obtained from tristearin (stearate is 18:0) Show steps pleasearrow_forwardMultiple choice urgent!!arrow_forward1. Write the transamination reaction for alanine. Indicate what happens next to each of the molecules in the reaction, and under what conditions it happens. 2.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY