
Concept explainers
a) Indole STRUCTURE GIVEN IN QUESTION
Interpretation:
The structure of indole is given. Its skeletal structure is to be drawn.
Concept introduction:
In skeletal structures the carbon atoms are not usually shown. Instead a carbon is assumed to be at each intersection of two lines and at the end of each line. The hydrogen atoms bonded to carbons are also not shown. The correct number of hydrogen atoms for each carbon atom is assigned keeping in mind that carbon has a valence of 4. The end of a line represents a carbon atom with three hydrogen atoms, CH3; a two-way intersection is a carbon atom with two hydrogen atoms, CH2; a three way intersection is a carbon with one hydrogen, CH; a four way intersection is a carbon with no attached hydrogen. Atoms other than carbon and hydrogen are shown. Nitrogen is trivalent and it can form one double bond and a single bond or three single bonds.
To draw:
The skeletal structure of indole.
Answer to Problem 42AP
The skeletal structure of indole is
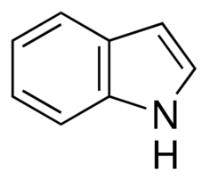
Explanation of Solution
The molecular formula of indole is C8H7N. It has bicyclic structure consisting of a six membered benzene ring fused to a five membered pyrrole ring. In skeletal structures the carbon atoms are not usually shown. Instead a carbon is assumed to be at each intersection of two lines and at the end of each line. The hydrogen atoms bonded to carbons are also not shown. The correct number of hydrogen atoms for each carbon atom is assigned keeping in mind that carbon has a valence of 4. A three way intersection is a carbon with one hydrogen, CH; a four way intersection is a carbon with no attached hydrogen. Atoms other than carbon and hydrogen are shown. Nitrogen is trivalent and it is in a three way intersection as NH.
The skeletal structure of indole is

b) 1,3- pentadiene
Interpretation:
The structure of 1,3- pentadiene is given. Its skeletal structure is to be drawn.
Concept introduction:
In skeletal structures the carbon atoms are not usually shown. Instead a carbon is assumed to be at each intersection of two lines and at the end of each line. The hydrogen atoms bonded to carbons are also not shown. The correct number of hydrogen atoms for each carbon atom is assigned keeping in mind that carbon has a valence of 4. The end of a line represents a carbon atom with three hydrogen atoms, CH3; a two-way intersection is a carbon atom with two hydrogen atoms, CH2; a three way intersection is a carbon with one hydrogen, CH; a four way intersection is a carbon with no attached hydrogen. Atoms other than carbon and hydrogen are shown.
To draw:
The skeletal structure of 1,3- pentadiene.
Answer to Problem 42AP
The skeletal structure of 1,3- pentadiene is
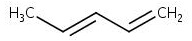
Explanation of Solution
The molecular formula of 1,3- pentadiene is C5H8. It has two double bonds, one between C1-C2 and another between C3-C4. In skeletal structures the carbon atoms are not usually shown. Instead a carbon is assumed to be at each intersection of two lines and at the end of each line. The hydrogen atoms bonded to carbons are also not shown. The correct number of hydrogen atoms for each carbon atom is assigned keeping in mind that carbon has a valence of 4. The end of a line represents a carbon atom with three hydrogen atoms, CH3; a two-way intersection is a carbon atom with two hydrogen atoms, CH2; a three way intersection is a carbon with one hydrogen, CH. Based on these concepts the structure of 1,3- pentadiene can be written.
The skeletal structure of 1,3- pentadiene is

c) 1,2- dichloro cyclopentane
Interpretation:
The structure of 1,2- dichloro cyclopentane is given. Its skeletal structure is to be drawn.
Concept introduction:
In skeletal structures the carbon atoms are not usually shown. Instead a carbon is assumed to be at each intersection of two lines and at the end of each line. The hydrogen atoms bonded to carbons are also not shown. The correct number of hydrogen atoms for each carbon atom is assigned keeping in mind that carbon has a valence of 4. The end of a line represents a carbon atom with three hydrogen atoms, CH3; a two-way intersection is a carbon atom with two hydrogen atoms, CH2; a three way intersection is a carbon with one hydrogen, CH; a four way intersection is a carbon with no attached hydrogen. Atoms other than carbon and hydrogen are shown.
To draw:
The skeletal structure of 1,2- dichloro cyclopentane.
Answer to Problem 42AP
The skeletal structure of 1,2- dichloro cyclopentane is
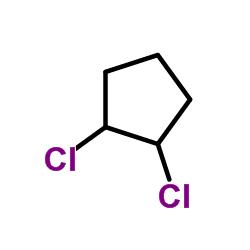
Explanation of Solution
In skeletal structures the carbon atoms are not usually shown. Instead a carbon is assumed to be at each intersection of two lines and at the end of each line. The hydrogen atoms bonded to carbons are also not shown. The correct number of hydrogen atoms for each carbon atom is assigned keeping in mind that carbon has a valence of 4. Atoms other than carbon and hydrogen are shown. The molecular formula of 1,2- dichlorocyclopentene is C5H8Cl2. A cyclopentane ring is made up of five methylene groups. In 1,2- dichlorocyclopentene the two chlorine atoms are bonded to the first and second carbons replacing one hydrogen on each. They are shown in the skeletal structure.
The skeletal structure of 1,2- dichloro cyclopentane is

d) benzoquinone
Interpretation:
The structure of benzoquinone is given. Its skeletal structure is to be drawn.
Concept introduction:
In skeletal structures the carbon atoms are not usually shown. Instead a carbon is assumed to be at each intersection of two lines and at the end of each line. The hydrogen atoms bonded to carbons are also not shown. The correct number of hydrogen atoms for each carbon atom is assigned keeping in mind that carbon has a valence of 4. Atoms other than carbon and hydrogen are shown.
To draw:
The skeletal structure of benzoquinone.
Answer to Problem 42AP
The skeletal structure of benzoquinone is
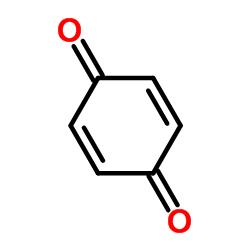
Explanation of Solution
In skeletal structures the carbon atoms are not usually shown. Instead a carbon is assumed to be at each intersection of two lines and at the end of each line. The hydrogen atoms bonded to carbons are also not shown. The correct number of hydrogen atoms for each carbon atom is assigned keeping in mind that carbon has a valence of 4. Atoms other than carbon and hydrogen are shown. The molecular formula of benzoquinone is C6H4O2. The six carbons form a six membered ring with first and fourth carbons as carbonyl groups. The other four carbons have one hydrogen each.
The skeletal structure of benzoquinone is

Want to see more full solutions like this?
Chapter 1 Solutions
Organic Chemistry - With Access (Custom)
- Indicate the product(s) B and C that are formed in the reaction: HN' OCH HC1 B + mayoritario C minoritario OCH3arrow_forwardIndicate the product(s) that are formed in the reaction: NH-NH, OCH3 -H₂O OCH3arrow_forward21.38 Arrange the molecules in each set in order of increasing acidity (from least acidic to most acidic). OH OH SH NH2 8 NH3 OH (b) OH OH OH (c) & & & CH3 NO2 21.39 Explain the trends in the acidity of phenol and the monofluoro derivatives of phenol. OH OH OH OH PK 10.0 PK 8.81 PK 9.28 PK 9.81arrow_forward
- identify which spectrum is for acetaminophen and which is for phenacetinarrow_forwardThe Concept of Aromaticity 21.15 State the number of 2p orbital electrons in each molecule or ion. (a) (b) (e) (f) (c) (d) (h) (i) DA (k) 21.16 Which of the molecules and ions given in Problem 21.15 are aromatic according to the Hückel criteria? Which, if planar, would be antiaromatic? 21.17 Which of the following structures are considered aromatic according to the Hückel criteria? ---0-0 (a) (b) (c) (d) (e) (h) H -H .8.0- 21.18 Which of the molecules and ions from Problem 21.17 have electrons donated by a heteroatom?arrow_forward1. Show the steps necessary to make 2-methyl-4-nonene using a Wittig reaction. Start with triphenylphosphine and an alkyl halide. After that you may use any other organic or inorganic reagents. 2. Write in the product of this reaction: CH3 CH₂ (C6H5)₂CuLi H₂O+arrow_forward
- 3. Name this compound properly, including stereochemistry. H₂C H3C CH3 OH 4. Show the step(s) necessary to transform the compound on the left into the acid on the right. Bri CH2 5. Write in the product of this LiAlH4 Br H₂C OHarrow_forwardWhat are the major products of the following reaction? Please provide a detailed explanation and a drawing to show how the reaction proceeds.arrow_forwardWhat are the major products of the following enolate alkylation reaction? Please include a detailed explanation as well as a drawing as to how the reaction proceeds.arrow_forward
- A block of zinc has an initial temperature of 94.2 degrees celcius and is immererd in 105 g of water at 21.90 degrees celcius. At thermal equilibrium, the final temperature is 25.20 degrees celcius. What is the mass of the zinc block? Cs(Zn) = 0.390 J/gxdegrees celcius Cs(H2O) = 4.18 J/gx degrees celcusarrow_forwardPotential Energy (kJ) 1. Consider these three reactions as the elementary steps in the mechanism for a chemical reaction. AH = -950 kJ AH = 575 kJ (i) Cl₂ (g) + Pt (s) 2C1 (g) + Pt (s) Ea = 1550 kJ (ii) Cl (g)+ CO (g) + Pt (s) → CICO (g) + Pt (s) (iii) Cl (g) + CICO (g) → Cl₂CO (g) Ea = 2240 kJ Ea = 2350 kJ AH = -825 kJ 2600 2400 2200 2000 1800 1600 1400 1200 1000 a. Draw the potential energy diagram for the reaction. Label the data points for clarity. The potential energy of the reactants is 600 kJ 800 600 400 200 0 -200- -400 -600- -800- Reaction Progressarrow_forwardCan u help me figure out the reaction mechanisms for these, idk where to even startarrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning



