
Chemistry: Atoms First
2nd Edition
ISBN: 9780073511184
Author: Julia Burdge, Jason Overby Professor
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 19, Problem 19.34QP
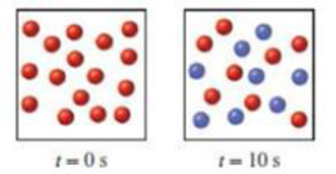
Consider the first-order reaction X → Y shown here, (a) What is the half-life of the reaction? (b) Draw pictures showing the number of X (red) and Y (blue) molecules at 20 s and at 30 s.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the epoxide formed when the following alkene is treated with mCPBA. Click the "draw
structure" button to launch the drawing utility.
draw structure ...
Rank each of the following substituted benzene molecules in order of which will react fastest (1) to slowest (4) by electrophilic
aromatic substitution.
Explanation
Check
CF3
(Choose one)
OH
(Choose one)
H
(Choose one)
(Choose one)
© 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy
Identifying electron-donating and electron-withdrawing effects
For each of the substituted benzene molecules below, determine the inductive and resonance effects the substituent will have on the
benzene ring, as well as the overall electron-density of the ring compared to unsubstituted benzene.
Molecule
Inductive Effects
Resonance Effects
Overall Electron-Density
CF3
O donating
O donating
O electron-rich
O withdrawing
withdrawing
O no inductive effects
O no resonance effects
O electron-deficient
O similar to benzene
OCH3
Explanation
Check
O donating
O donating
○ withdrawing
withdrawing
O no inductive effects
no resonance effects
electron-rich
electron-deficient
O similar to benzene
Х
© 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center
Chapter 19 Solutions
Chemistry: Atoms First
Ch. 19.3 - Prob. 19.1WECh. 19.3 - Write the rate expressions for each of the...Ch. 19.3 - Write the balanced equation corresponding to the...Ch. 19.3 - The diagrams represent a system that initially...Ch. 19.3 - Consider the reaction 4NO2(g)+O2(g)2N2O5(g) At a...Ch. 19.3 - Consider the reaction 4PH3(g)P4(g)+6H2(g) At a...Ch. 19.3 - Prob. 2PPBCh. 19.3 - Prob. 2PPCCh. 19.3 - Prob. 19.3.1SRCh. 19.3 - Prob. 19.3.2SR
Ch. 19.4 - The gas-phase reaction of nitric oxide with...Ch. 19.4 - Prob. 3PPACh. 19.4 - Prob. 3PPBCh. 19.4 - Prob. 3PPCCh. 19.4 - Prob. 19.4.1SRCh. 19.4 - Prob. 19.4.2SRCh. 19.4 - Prob. 19.4.3SRCh. 19.4 - Prob. 19.4.4SRCh. 19.4 - Prob. 19.4.5SRCh. 19.5 - Prob. 19.4WECh. 19.5 - Prob. 4PPACh. 19.5 - Prob. 4PPBCh. 19.5 - Prob. 4PPCCh. 19.5 - Prob. 19.5WECh. 19.5 - Prob. 5PPACh. 19.5 - Prob. 5PPBCh. 19.5 - Prob. 5PPCCh. 19.5 - Prob. 19.6WECh. 19.5 - Prob. 6PPACh. 19.5 - Calculate the rate constant for the first-order...Ch. 19.5 - Prob. 6PPCCh. 19.5 - Prob. 19.7WECh. 19.5 - The reaction 2A B is second order in A with a rate...Ch. 19.5 - Prob. 7PPBCh. 19.5 - Prob. 7PPCCh. 19.5 - Prob. 19.5.1SRCh. 19.5 - Prob. 19.5.2SRCh. 19.5 - Prob. 19.5.3SRCh. 19.5 - Prob. 19.5.4SRCh. 19.6 - Prob. 19.8WECh. 19.6 - Prob. 8PPACh. 19.6 - Prob. 8PPBCh. 19.6 - Prob. 8PPCCh. 19.6 - Prob. 19.9WECh. 19.6 - Prob. 9PPACh. 19.6 - Prob. 9PPBCh. 19.6 - Prob. 9PPCCh. 19.6 - Prob. 19.10WECh. 19.6 - Prob. 10PPACh. 19.6 - Prob. 10PPBCh. 19.6 - Prob. 10PPCCh. 19.6 - Prob. 19.6.1SRCh. 19.6 - Prob. 19.6.2SRCh. 19.7 - Prob. 19.11WECh. 19.7 - Prob. 11PPACh. 19.7 - Prob. 11PPBCh. 19.7 - Prob. 11PPCCh. 19.7 - Consider the gas-phase reaction of nitric oxide...Ch. 19.7 - Prob. 12PPACh. 19.7 - Prob. 12PPBCh. 19.7 - Prob. 12PPCCh. 19.7 - Prob. 19.7.1SRCh. 19.7 - Prob. 19.7.2SRCh. 19.7 - Prob. 19.7.3SRCh. 19.7 - Prob. 19.7.4SRCh. 19 - The rate of a reaction in which the reactant...Ch. 19 - The rate of a reaction in which the reactant...Ch. 19 - The rate of a reaction in which the reactant...Ch. 19 - Increasing the temperature of a reaction increases...Ch. 19 - Define activation energy. What role does...Ch. 19 - Sketch a potential energy versus reaction progress...Ch. 19 - The reaction H + H2 H2 + H has been studied for...Ch. 19 - What is meant by the rate of a chemical reaction?...Ch. 19 - Distinguish between average rate and instantaneous...Ch. 19 - What are the advantages of measuring the initial...Ch. 19 - Prob. 19.7QPCh. 19 - Consider the reaction N2(g)+3H2(g)2NH3(g) Suppose...Ch. 19 - Prob. 19.9QPCh. 19 - Prob. 19.10QPCh. 19 - Prob. 19.11QPCh. 19 - Prob. 19.12QPCh. 19 - Prob. 19.13QPCh. 19 - What are the units for the rate constants of...Ch. 19 - Consider the zeroth-order reaction: A product....Ch. 19 - Prob. 19.16QPCh. 19 - Prob. 19.17QPCh. 19 - Prob. 19.18QPCh. 19 - Prob. 19.19QPCh. 19 - Prob. 19.20QPCh. 19 - Prob. 19.21QPCh. 19 - Prob. 19.22QPCh. 19 - Prob. 19.23QPCh. 19 - Prob. 19.24QPCh. 19 - Prob. 19.25QPCh. 19 - Prob. 19.26QPCh. 19 - Prob. 19.27QPCh. 19 - Prob. 19.28QPCh. 19 - Prob. 19.29QPCh. 19 - Prob. 19.30QPCh. 19 - Prob. 19.31QPCh. 19 - Prob. 19.32QPCh. 19 - Prob. 19.33QPCh. 19 - Consider the first-order reaction X Y shown here,...Ch. 19 - Prob. 19.35QPCh. 19 - Consider the first-order reaction A B in which A...Ch. 19 - Prob. 19.37QPCh. 19 - Prob. 19.38QPCh. 19 - Prob. 19.39QPCh. 19 - Prob. 19.40QPCh. 19 - Prob. 19.41QPCh. 19 - Prob. 19.42QPCh. 19 - Prob. 19.43QPCh. 19 - Prob. 19.44QPCh. 19 - Prob. 19.45QPCh. 19 - The rate at which tree crickets chirp is 2.0 102...Ch. 19 - Prob. 19.47QPCh. 19 - The activation energy for the denaturation of a...Ch. 19 - Variation of the rate constant with temperature...Ch. 19 - Prob. 19.50QPCh. 19 - Prob. 19.51QPCh. 19 - Prob. 19.52QPCh. 19 - Prob. 19.53QPCh. 19 - What is an elementary step? What is the...Ch. 19 - Prob. 19.55QPCh. 19 - Determine the molecularity, and write the rate law...Ch. 19 - What is the rate-determining step of a reaction?...Ch. 19 - Prob. 19.58QPCh. 19 - Prob. 19.59QPCh. 19 - Classify each of the following elementary steps as...Ch. 19 - Prob. 19.61QPCh. 19 - Prob. 19.62QPCh. 19 - Prob. 19.63QPCh. 19 - Prob. 19.64QPCh. 19 - Prob. 19.65QPCh. 19 - What are the characteristics of a catalyst?Ch. 19 - Prob. 19.67QPCh. 19 - Prob. 19.68QPCh. 19 - The concentrations of enzymes in cells are usually...Ch. 19 - Prob. 19.70QPCh. 19 - Prob. 19.71QPCh. 19 - Prob. 19.72QPCh. 19 - Prob. 19.73QPCh. 19 - Prob. 19.74QPCh. 19 - Prob. 19.75QPCh. 19 - In a certain industrial process involving a...Ch. 19 - Prob. 19.77QPCh. 19 - Prob. 19.78QPCh. 19 - Explain why most metals used in catalysis arc...Ch. 19 - Prob. 19.80QPCh. 19 - Prob. 19.81QPCh. 19 - Prob. 19.82QPCh. 19 - Prob. 19.83QPCh. 19 - Prob. 19.84QPCh. 19 - The bromination of acetone is acid-catalyzed. The...Ch. 19 - The decomposition of N2O to N2 and O2 is a...Ch. 19 - Prob. 19.87QPCh. 19 - Prob. 19.88QPCh. 19 - The integrated rate law for the zeroth-order...Ch. 19 - Prob. 19.90QPCh. 19 - Prob. 19.91QPCh. 19 - Prob. 19.92QPCh. 19 - The reaction of G2 with E2 to form 2EG is...Ch. 19 - Prob. 19.94QPCh. 19 - Prob. 19.95QPCh. 19 - Prob. 19.96QPCh. 19 - Strictly speaking, the rate law derived for the...Ch. 19 - Prob. 19.98QPCh. 19 - The decomposition of dinitrogen pentoxide has been...Ch. 19 - Prob. 19.100QPCh. 19 - Prob. 19.101QPCh. 19 - Prob. 19.102QPCh. 19 - To prevent brain damage, a standard procedure is...Ch. 19 - Prob. 19.104QPCh. 19 - Prob. 19.105QPCh. 19 - Prob. 19.106QPCh. 19 - Prob. 19.107QPCh. 19 - Prob. 19.108QPCh. 19 - Prob. 19.109QPCh. 19 - Prob. 19.110QPCh. 19 - (a) What can you deduce about the activation...Ch. 19 - Prob. 19.112QPCh. 19 - Prob. 19.113QPCh. 19 - Prob. 19.114QPCh. 19 - Prob. 19.115QPCh. 19 - Prob. 19.116QPCh. 19 - Prob. 19.117QPCh. 19 - Prob. 19.118QPCh. 19 - Prob. 19.119QPCh. 19 - Prob. 19.120QPCh. 19 - Prob. 19.121QPCh. 19 - Prob. 19.122QPCh. 19 - Consider the following potential energy profile...Ch. 19 - Prob. 19.124QPCh. 19 - Prob. 19.125QPCh. 19 - Prob. 19.126QPCh. 19 - Prob. 19.127QPCh. 19 - Prob. 19.128QPCh. 19 - The following expression shows the dependence of...Ch. 19 - Prob. 19.130QPCh. 19 - The rale constant for the gaseous reaction H2(g) +...Ch. 19 - Prob. 19.132QPCh. 19 - Prob. 19.133QPCh. 19 - At a certain elevated temperature, ammonia...Ch. 19 - Prob. 19.135QPCh. 19 - The rate of a reaction was followed by the...Ch. 19 - Prob. 19.137QPCh. 19 - Prob. 19.138QPCh. 19 - Prob. 19.1KSPCh. 19 - Prob. 19.2KSPCh. 19 - Prob. 19.3KSPCh. 19 - Prob. 19.4KSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The acid-base chemistry of both EDTA and EBT are important to ensuring that the reactions proceed as desired, thus the pH is controlled using a buffer. What percent of the EBT indicator will be in the desired HIn2- state at pH = 10.5. pKa1 = 6.2 and pKa2 = 11.6 of EBTarrow_forwardCUE COLUMN NOTES (A. Determine Stereoisomers it has ⑤ Identify any meso B compounds cl Br cl -c-c-c-c-¿- 1 CI C- | 2,4-Dichloro-3-bromopentanearrow_forwardThe acid-base chemistry of both EDTA and EBT are important to ensuring that the reactions proceed as desired, thus the pH is controlled using a buffer. What percent of the EBT indicator will be in the desired HIn2- state at pH = 10.5. pKa1 = 6.2 and pKa2 = 11.6 of EBTarrow_forward
- What does the phrase 'fit for purpose' mean in relation to analytical chemistry? Please provide examples too.arrow_forwardFor each of the substituted benzene molecules below, determine the inductive and resonance effects the substituent will have on the benzene ring, as well as the overall electron-density of the ring compared to unsubstituted benzene. Molecule Inductive Effects Resonance Effects Overall Electron-Density × NO2 ○ donating O donating O withdrawing O withdrawing O electron-rich electron-deficient no inductive effects O no resonance effects O similar to benzene E [ CI O donating withdrawing O no inductive effects Explanation Check ○ donating withdrawing no resonance effects electron-rich electron-deficient O similar to benzene © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center Accesarrow_forwardUnderstanding how substituents activate Rank each of the following substituted benzene molecules in order of which will react fastest (1) to slowest (4) by electrophilic aromatic substitution. Explanation HN NH2 Check X (Choose one) (Choose one) (Choose one) (Choose one) © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center Aarrow_forward
- Identifying electron-donating and electron-withdrawing effects on benzene For each of the substituted benzene molecules below, determine the inductive and resonance effects the substituent will have on the benzene ring, as well as the overall electron-density of the ring compared to unsubstituted benzene. Inductive Effects Resonance Effects Overall Electron-Density Molecule CF3 O donating O donating O withdrawing O withdrawing O no inductive effects O no resonance effects electron-rich electron-deficient O similar to benzene CH3 O donating O withdrawing O no inductive effects O donating O withdrawing Ono resonance effects O electron-rich O electron-deficient O similar to benzene Explanation Check Х © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Centerarrow_forward* Hint: Think back to Chem 1 solubility rules. Follow Up Questions for Part B 12. What impact do the following disturbances to a system at equilibrium have on k, the rate constant for the forward reaction? Explain. (4 pts) a) Changing the concentration of a reactant or product. (2 pts) b) Changing the temperature of an exothermic reaction. (2 pts) ofarrow_forwardDraw TWO general chemical equation to prepare Symmetrical and non-Symmetrical ethers Draw 1 chemical reaction of an etherarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Physical Chemistry
Chemistry
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Wadsworth Cengage Learning,

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Kinetics: Initial Rates and Integrated Rate Laws; Author: Professor Dave Explains;https://www.youtube.com/watch?v=wYqQCojggyM;License: Standard YouTube License, CC-BY