
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18.SE, Problem 32MP
Fluoxetine, a heavily prescribed antidepressant marketed under the name Prozac, can be prepared by a route that begins with reaction between a phenol and an alkyl chloride.
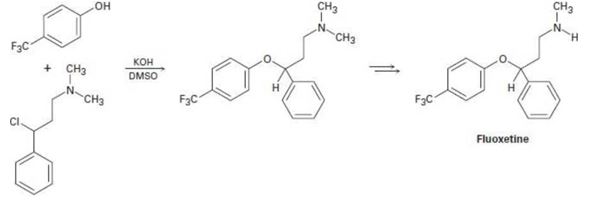
(a) The
(b) The physiologically active enantiomer of fluoxetine has (S) stereochemistry. Based on your answer in part (a), draw the structure of the alkyl chloride you would need, showing the correct stereochemistry.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Chloroform, long used as an anesthetic and now considered carcinogenic, has a heat of vaporization of 31.4 kJ/mol. During vaporization, its entropy increases by 94.2 J/mol.K. Therefore, select the alternative that indicates the temperature, in degrees Celsius, at which chloroform begins to boil under a pressure of 1 atm.
A) 28
B) 40
C) 52
D) 60
E) 72
If we assume a system with an anodic overpotential, the variation of n as a function
of current density:
1. at low fields is linear 2. at higher fields, it follows Tafel's law
Obtain the range of current densities for which the overpotential has the same value
when calculated for 1 and 2 cases (maximum relative difference of 5% compared to
the behavior for higher fields).
To which overpotential range does this correspond?
Data: i = 1.5 mA cm², T = 300°C, B = 0.64, R = 8.314 J K1 mol-1 and F = 96485 C mol-1.
Answer by equation please
Chapter 18 Solutions
Organic Chemistry
Ch. 18.1 - Name the following ethers:Ch. 18.2 - Why do you suppose only symmetrical ethers are...Ch. 18.2 - How would you prepare the following ethers using a...Ch. 18.2 - Review the mechanism of oxymercuration shown in...Ch. 18.2 - How would you prepare the following ethers? Use...Ch. 18.2 - Prob. 6PCh. 18.3 - Prob. 7PCh. 18.3 - Write the mechanism of the acid-induced cleavage...Ch. 18.3 - Why are HI and HBr more effective than HCl in...Ch. 18.4 - What product would you expect from Claisen...
Ch. 18.5 - Prob. 11PCh. 18.6 - Predict the major product of each of the following...Ch. 18.6 - Prob. 13PCh. 18.6 - Predict the major product of the following...Ch. 18.7 - 15-Crown-5 and 12-crown-4 ethers complex Na+ and...Ch. 18.8 - Prob. 16PCh. 18.8 - 2-Butene-l-thiol is one component of skunk spray....Ch. 18.9 - The 1H NMR spectrum shown is that of a cyclic...Ch. 18.SE - Give IUPAC names for the following compounds...Ch. 18.SE - Show the product, including stereochemistry, that...Ch. 18.SE - Prob. 21VCCh. 18.SE - Treatment of the following alkene with a...Ch. 18.SE - Prob. 23MPCh. 18.SE - Prob. 24MPCh. 18.SE - Predict the product(s) and provide the mechanism...Ch. 18.SE - The alkoxymercuration of alkenes involves the...Ch. 18.SE - Predict the product(s) and provide the mechanism...Ch. 18.SE - Predict the product(s) and provide the mechanism...Ch. 18.SE - Prob. 29MPCh. 18.SE - Ethers undergo an acid-catalyzed cleavage reaction...Ch. 18.SE - Treatment of 1, 1-diphenyl-l, 2-epoxyethane with...Ch. 18.SE - Fluoxetine, a heavily prescribed antidepressant...Ch. 18.SE - When 2-methyl-2, 5-pentanediol is treated with...Ch. 18.SE - Prob. 34MPCh. 18.SE - Prob. 35MPCh. 18.SE - Aldehydes and ketones undergo acid-catalyzed...Ch. 18.SE - Propose a mechanism to account for the following...Ch. 18.SE - Prob. 38APCh. 18.SE - Prob. 39APCh. 18.SE - How would you prepare the following ethers?Ch. 18.SE - Prob. 41APCh. 18.SE - tert-Butyl ethers can be prepared by the reaction...Ch. 18.SE - Treatment of trans-2-chlorocyclohexanol with NaOH...Ch. 18.SE - Predict the products of the following ether...Ch. 18.SE - Prob. 45APCh. 18.SE - Prob. 46APCh. 18.SE - Write the mechanism of the hydrolysis of cis-5,...Ch. 18.SE - Prob. 48APCh. 18.SE - Acid-catalyzed hydrolysis of a 1,...Ch. 18.SE - Prob. 50APCh. 18.SE - Epoxides are reduced by treatment with lithium...Ch. 18.SE - Prob. 52APCh. 18.SE - The red fox (Vulpes vulpes) uses a chemical...Ch. 18.SE - Anethole, C10H12O, a major constituent of the oil...Ch. 18.SE - Propose structures for compounds that have the...Ch. 18.SE - Prob. 56GPCh. 18.SE - How would you synthesize anethole (Problem 18-54)...Ch. 18.SE - How could you prepare benzyl phenyl ether from...Ch. 18.SE - Meerwein's reagent, triethyloxonium...Ch. 18.SE - Prob. 60GPCh. 18.SE - Prob. 61GPCh. 18.SE - The Zeisel method is an old analytical procedure...Ch. 18.SE - Prob. 63GPCh. 18.SE - Prob. 64GPCh. 18.SE - Prob. 65GPCh. 18.SE - Identify the reagents a-e in the following scheme:Ch. 18.SE - Propose structures for compounds that have the...Ch. 18.SE - Prob. 68GPCh. 18.SE - Prob. 69GPCh. 18.SE - Predict the product(s) if the starting materials...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Some of the theories used to describe interface structure can be distinguished by:1. the measured potential difference.2. the distribution of ions in solution.3. the calculation of charge density.4. the external Helmoltz plane.arrow_forwardWhen talking about the acidity of carboxylic acids, is it the same thing to say higher or stronger acidity?arrow_forwardUsing the following two half-reactions, determine the pH range in which $NO_2^-\ (aq)$ cannot be found as the predominant chemical species in water.* $NO_3^-(aq)+10H^+(aq)+8e^-\rightarrow NH_4^+(aq)+3H_2O(l),\ pE^{\circ}=14.88$* $NO_2^-(aq)+8H^+(aq)+6e^-\rightarrow NH_4^+(aq)+2H_2O(l),\ pE^{\circ}=15.08$arrow_forward
- Indicate the formula of the product obtained by reacting methyl 5-chloro-5-oxopentanoate with 1 mole of 4-penten-1-ylmagnesium bromide.arrow_forwardIn the two chair conformations of glucose, the most stable is the one with all the OH groups in the equatorial position. Is this correct?arrow_forwardIndicate the formula of the product obtained by reacting D-Galactose with hydroxylamine.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License