
Concept explainers
Synthesize each compound from benzene and any other organic or inorganic reagents.
a. 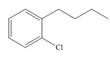 c.
c. 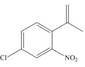 e.
e. 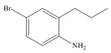 g.
g. 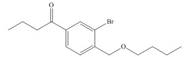
b. 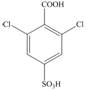 d.
d. 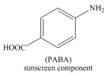 f.
f. 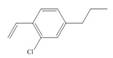 h.
h. 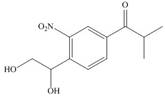
(a)
Interpretation: The synthesis of given compound from benzene and any other organic or inorganic reagent is to be stated.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.63P
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown below:
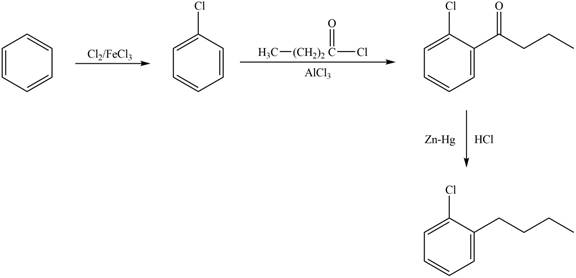
Explanation of Solution
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown in Figure 1.
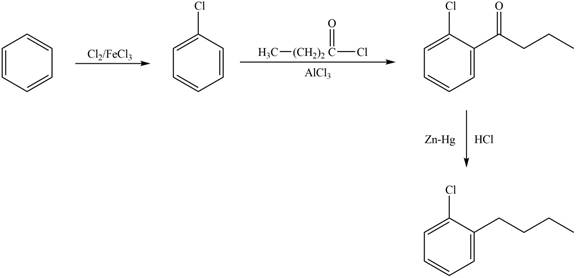
Figure 1
The synthesis of given compound takes place by chlorination of benzene, Friedel-Craft acylation and clemmensen reduction at last step.
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown in Figure 1.
(b)
Interpretation: The synthesis of given compound from benzene and any other organic or inorganic reagent is to be stated.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.63P
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown below:
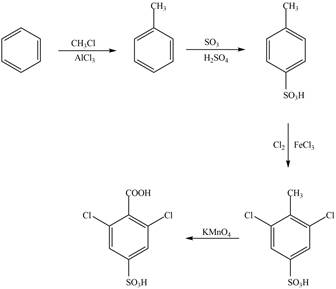
Explanation of Solution
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown in Figure 2.
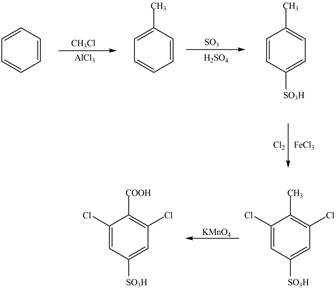
Figure 2
The synthesis of given compound take place in four steps: Friedel-Craft alkylation, sulfonation, chlorination and at last oxidation.
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown in Figure 2.
(c)
Interpretation: The synthesis of given compound from benzene and any other organic or inorganic reagent is to be stated.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.63P
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown below:
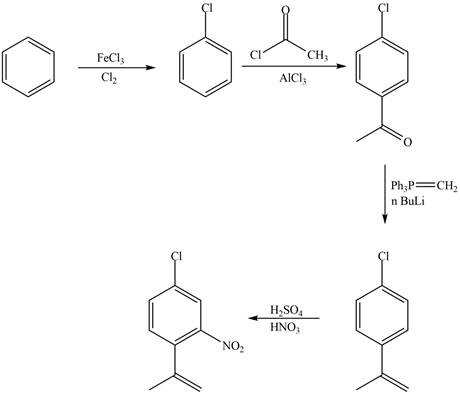
Explanation of Solution
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown in Figure 3.
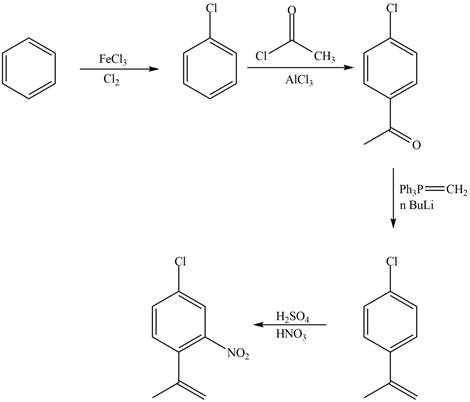
Figure 3
The first, second, third and fourth step involved in the synthesis of given compound is chlorination, Friedel-Craft acylation, Wittig reaction and nitration, respectively.
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown in Figure 3.
(d)
Interpretation: The synthesis of given compound from benzene and any other organic or inorganic reagent is to be stated.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.63P
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown below:
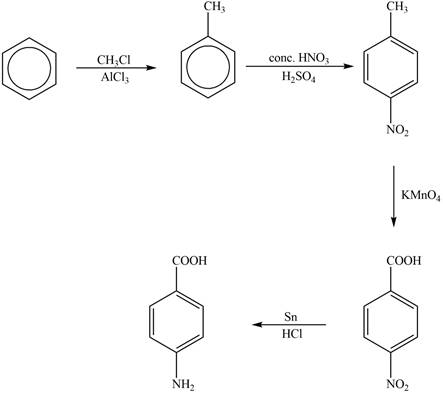
Explanation of Solution
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown in Figure 4.
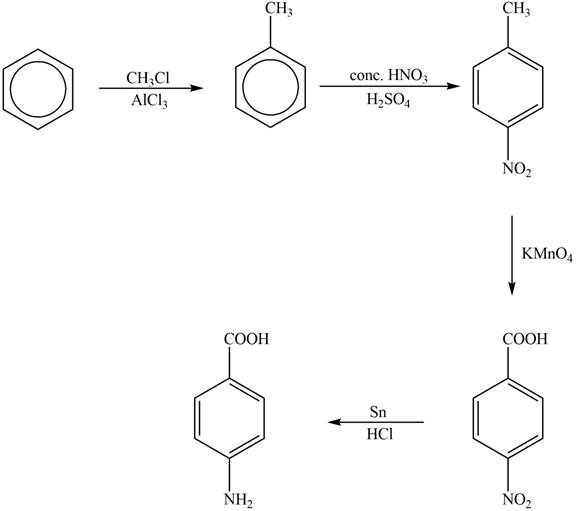
Figure 4
The first step of the synthesis is Friedel-Craft alkylation reaction. The product of this reaction undergoes nitration reaction. In next step, the
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown in Figure 4.
(e)
Interpretation: The synthesis of given compound from benzene and any other organic or inorganic reagent is to be stated.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.63P
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown below:
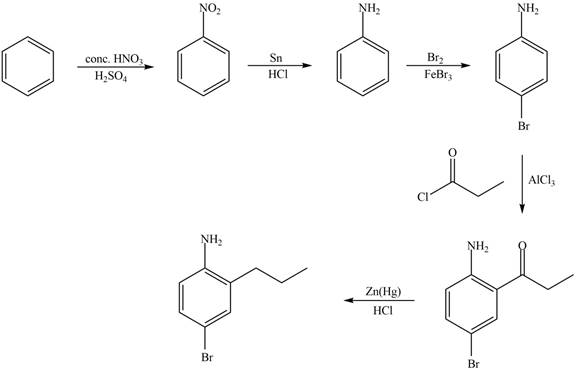
Explanation of Solution
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown in Figure 5.
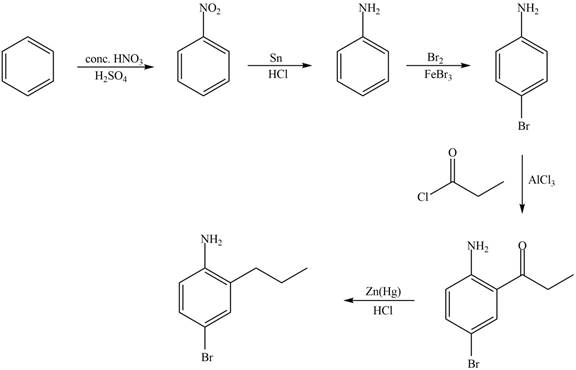
Figure 5
Benzene undergoes nitration on reaction with
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown in Figure 5.
(f)
Interpretation: The synthesis of given compound from benzene and any other organic or inorganic reagent is to be stated.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.63P
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown below:
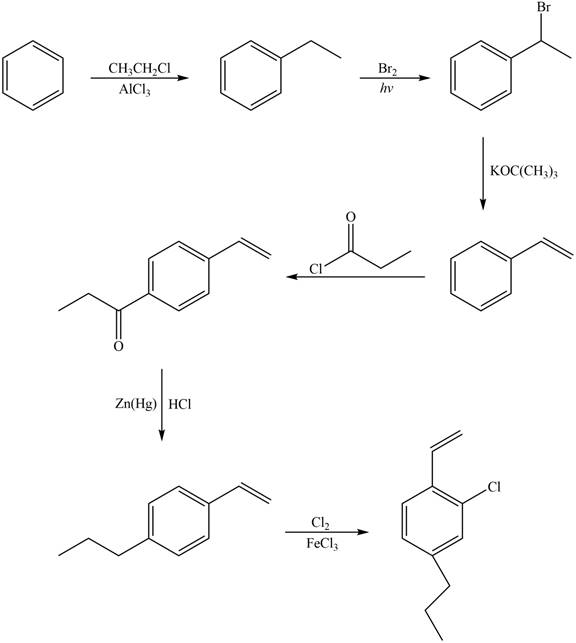
Explanation of Solution
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown in Figure 6.
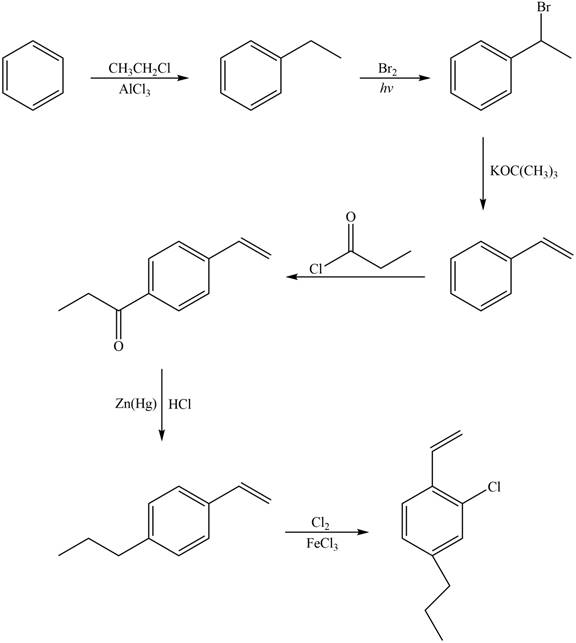
Figure 6
Benzene on Friedel-Craft alkylation with ethylchloride gives ethylbenzene. This product undergoes bromination in presence of light. The next step involves the abstraction of bromine on reaction with tertiary butoxide. This leads to the formation of
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown in Figure 6.
(g)
Interpretation: The synthesis of given compound from benzene and any other organic or inorganic reagent is to be stated.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.63P
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown below:
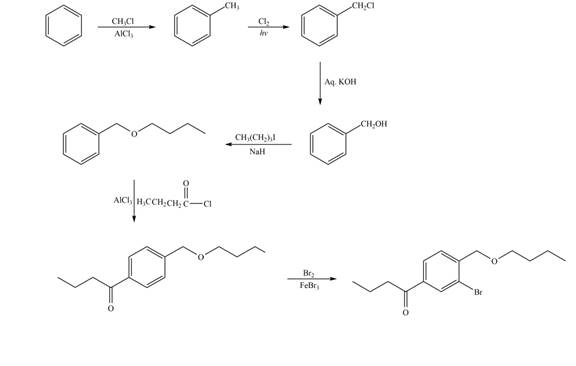
Explanation of Solution
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown in Figure 7.
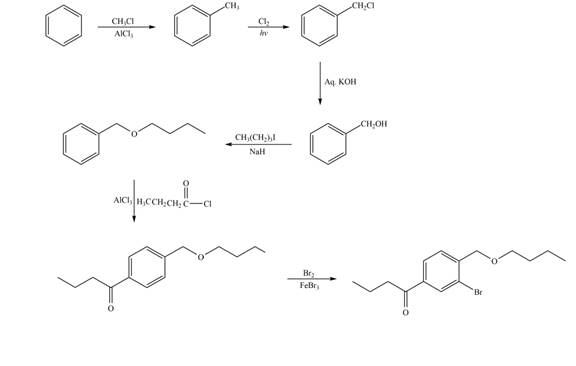
Figure 7
The first two steps involved in the synthesis of given compound are Friedel-Craft alkylation followed by chlorination in the presence of light. The chlorine group is replaced by hydroxyl group on reaction with
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown in Figure 7.
(h)
Interpretation: The synthesis of given compound from benzene and any other organic or inorganic reagent is to be stated.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.63P
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown below:
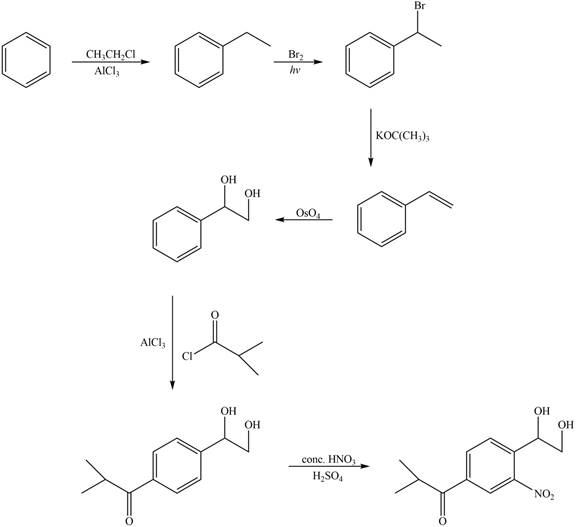
Explanation of Solution
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown in Figure 8.

Figure 8
Benzene on Friedel-Craft alkylation with ethylchloride gives ethylbenzene. This product undergoes bromination in presence of light. The next step involves the abstraction of bromine on reaction with tertiary butoxide. This leads to the formation of
The synthesis of given compound from benzene and any other organic or inorganic reagent is shown in Figure 8.
Want to see more full solutions like this?
Chapter 18 Solutions
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
- Please correct answer and don't used hand raiting and don't used Ai solutionarrow_forwardPlease correct answer and don't used hand raitingarrow_forwardThe vibrational contribution isa) temperature independent for internal energy and heat capacityb) temperature dependent for internal energy and heat capacityc) temperature independent for heat capacityd) temperature independent for internal energyarrow_forward
- Quantum mechanics. Explain the basis of approximating the summation to an integral in translational motion.arrow_forwardQuantum mechanics. In translational motion, the summation is replaced by an integral when evaluating the partition function. This is correct becausea) the spacing of the translational energy levels is very small compared to the product kTb) the spacing of the translational energy levels is comparable to the product kTc) the spacing of the translational energy levels is very large compared to the product kTarrow_forwardDon't used Ai solutionarrow_forward
- Please correct answer and don't used hand raiting don't used Ai solutionarrow_forwardIf the viscosity of hydrogen gas (at 0oC and 1 atm) is 8.83x10-5 P. If we assume that the molecular sizes are equal, calculate the viscosity of a gas composed of deuterium.arrow_forwardIf the viscosity of hydrogen gas (at 0oC and 1 atm) is 8.83x10-5 P. If we assume that the molecular sizes are equal, calculate the viscosity of a gas composed of deuterium.arrow_forward
- Laser. Indicate the relationship between metastable state and stimulated emission.arrow_forwardThe table includes macrostates characterized by 4 energy levels (&) that are equally spaced but with different degrees of occupation. a) Calculate the energy of all the macrostates (in joules). See if they all have the same energy and number of particles. b) Calculate the macrostate that is most likely to exist. For this macrostate, show that the population of the levels is consistent with the Boltzmann distribution. macrostate 1 macrostate 2 macrostate 3 ε/k (K) Populations Populations Populations 300 5 3 4 200 7 9 8 100 15 17 16 0 33 31 32 DATO: k = 1,38×10-23 J K-1arrow_forwardDon't used Ai solutionarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





