
Organic Chemistry
11th Edition
ISBN: 9781118133576
Author: T. W. Graham Solomons, Craig Fryhle
Publisher: Wiley, John & Sons, Incorporated
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 3PP
PRACTICE PROBLEM
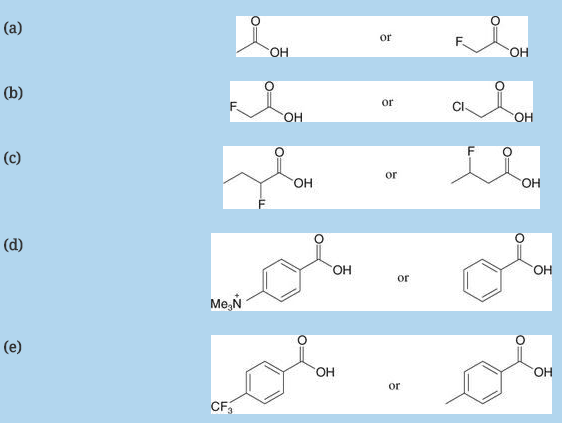
Which acid of each pair shown here would you expect to be stronger?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Using the Nernst equation to calculate nonstandard cell voltage
A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction:
MnO2 (s)+4H* (aq)+2Cr²+ (aq) → Mn²+ (aq)+2H₂O (1)+2Cr³+ (aq)
+
2+
2+
3+
Suppose the cell is prepared with 7.44 M H* and 0.485 M Cr²+ in one half-cell and 7.92 M Mn² and 3.73 M Cr³+ in the other.
Calculate the cell voltage under these conditions. Round your answer to 3 significant digits.
☐
x10
μ
Х
5
?
000
日。
Calculating standard reaction free energy from standard reduction...
Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free energy AG° for the following redox reaction.
Be sure your answer has the correct number of significant digits.
NO (g) +H₂O (1) + Cu²+ (aq) → HNO₂ (aq) +H* (aq)+Cu* (aq)
kJ
-
☐ x10
x10
olo
18
Ar
Calculating the pH of a weak base titrated with a strong acid
b
An analytical chemist is titrating 116.9 mL of a 0.7700M solution of aniline (C6H5NH2) with a 0.5300M solution of HNO3. The pK of aniline is 9.37.
Calculate the pH of the base solution after the chemist has added 184.2 mL of the HNO 3 solution to it.
Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HNO3 solution added.
Round your answer to 2 decimal places.
pH = ☐
☑
5
Chapter 17 Solutions
Organic Chemistry
Ch. 17 - Practice Problem 17.1 Give an IUPAC systematic...Ch. 17 - Prob. 2PPCh. 17 - PRACTICE PROBLEM
17.3 Which acid of each pair...Ch. 17 - Practice Problem 17.3 Write structural formulas...Ch. 17 - Practice Problem 17.4
Show how each of the...Ch. 17 - Practice Problem 17.5
Show how you could prepare...Ch. 17 - Practice Problem 17.6
(a) Which of the carboxylic...Ch. 17 - Prob. 8PPCh. 17 - Prob. 9PPCh. 17 - Practice Problem 17.9
Esters can also be...
Ch. 17 - Prob. 11PPCh. 17 - Prob. 12PPCh. 17 - Practice Problem 17.12
What products would you...Ch. 17 - Practice Problem 17.13 (a) Provide the reagents...Ch. 17 - Prob. 15PPCh. 17 - Practice Problem 17.15 Using decarboxylation...Ch. 17 - Practice Problem 17.16 Diacyl peroxides, ,...Ch. 17 - Prob. 18PCh. 17 - Give an IUPAC systematic or common name for each...Ch. 17 - Prob. 20PCh. 17 - Prob. 21PCh. 17 - 17.21 What major organic product would you expect...Ch. 17 - Prob. 23PCh. 17 - Prob. 24PCh. 17 - Prob. 25PCh. 17 - Prob. 26PCh. 17 - 17.26 What products would you expect to obtain...Ch. 17 - Write structural formulas for the major organic...Ch. 17 - 17.28 Indicate reagents that would accomplish each...Ch. 17 - Write structural formulas for the major organic...Ch. 17 - Prob. 31PCh. 17 - Prob. 32PCh. 17 - Prob. 33PCh. 17 - 17.33 On heating,...Ch. 17 - Prob. 35PCh. 17 - Prob. 36PCh. 17 - 17.36 Show how pentanoic acid can be prepared from...Ch. 17 - 17.37 The active ingredient of the insect...Ch. 17 - Prob. 39PCh. 17 - Prob. 40PCh. 17 - Give stereochemical formulas for compounds AQ:...Ch. 17 - 17.41 -Glyceraldehyde can be transformed into...Ch. 17 - Prob. 43PCh. 17 - 17.44 Cantharidin is a powerful vesicant that can...Ch. 17 - Prob. 45PCh. 17 - 17.44 Given here are the NMR spectra and carbonyl...Ch. 17 - Compound X (C7H12O4) is insoluble in aqueous...Ch. 17 - 17.45 Compound Y dissolves slowly when warmed...Ch. 17 - Prob. 49PCh. 17 - Prob. 50PCh. 17 - 17.52 Starting with 1-naphthol, suggest an...Ch. 17 - Suggest a synthesis of ibuprofen (Section 5.11)...Ch. 17 - Prob. 53PCh. 17 - Prob. 54PCh. 17 - Prob. 1LGPCh. 17 - Prob. 2LGPCh. 17 - Prob. 3LGPCh. 17 - Prob. 4LGPCh. 17 - Prob. 1QCh. 17 - 17.2 Which of the following would yield...Ch. 17 - 17.3 Which reagent would serve as the basis for a...Ch. 17 - Prob. 4QCh. 17 - Complete the following synthesis.Ch. 17 - 17.6 Which of these acids would undergo...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Plants use the process of photosynthesis to convert the energy in sunlight to chemical energy in the form of su...
Campbell Essential Biology with Physiology (5th Edition)
The glycine cleavage system is a group of four enzymes that together catalyze the following reaction: glycine+T...
Organic Chemistry (8th Edition)
Write a statement that compares the width of the meander belt of the Red River to the width of its floodplain.
Applications and Investigations in Earth Science (9th Edition)
Could an organism be a fermenter and also be both MR and V—P negative? Explain.
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
Modified True/False 9. A giant bacterium that is large enough to be seen without a microscope is Selenomonas.
Microbiology with Diseases by Body System (5th Edition)
All of the following terms can appropriately describe humans except: a. primary consumer b. autotroph c. hetero...
Human Biology: Concepts and Current Issues (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- QUESTION: Find the standard deviation for the 4 different groups 5.298 3.977 223.4 148.7 5.38 4.24 353.7 278.2 5.033 4.044 334.6 268.7 4.706 3.621 305.6 234.4 4.816 3.728 340.0 262.7 4.828 4.496 304.3 283.2 4.993 3.865 244.7 143.6 STDEV = STDEV = STDEV = STDEV =arrow_forwardQUESTION: Fill in the answers in the empty green boxes regarding 'Question 5: Calculating standard error of regression' *The images of the data showing 'coefficients for the standard curve' have been providedarrow_forwardUsing the Nernst equation to calculate nonstandard cell voltage Try Again Your answer is wrong. In addition to checking your math, check that you used the right data and DID NOT round any intermediate calculations. A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction: 2+ 2+ Sn²+ Ba(s) (aq) + Ba (s) Sn (s) + Ba²+ (aq) →>> Suppose the cell is prepared with 6.10 M Sn 2+ 2+ in one half-cell and 6.62 M Ba in the other. Calculate the cell voltage under these conditions. Round your answer to 3 significant digits. 1.71 V ☐ x10 ☑ 5 0/5 ? 00. 18 Ararrow_forward
- Question: Find both the b (gradient) and a (y-intercept) value from the list of data below: (x1 -x̄) 370.5 (y1 - ȳ) 5.240 (x2 - x̄) 142.5 (y2 - ȳ) 2.004 (x3 - x̄) 28.5 (y3 - ȳ) 0.390 (x4 - x̄) -85.5 (y4 - ȳ) -1.231 (x5 - x̄) -199.5 (y5 - ȳ) -2.829 (x6 - x̄) -256.5 (y6 - ȳ) -3.575arrow_forwardCalculating standard reaction free energy from standard reduction... Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free energy AG° for the following redox reaction. Be sure your answer has the correct number of significant digits. 3Cu+ (aq) + Cro²¯ (aq) +4H₂O (1) → 3Cu²+ (aq) +Cr(OH)3 (s)+5OH˜¯ (aq) 0 kJ ☐ x10 00. 18 Ararrow_forwardCalculating the pH of a weak base titrated with a strong acid An analytical chemist is titrating 241.7 mL of a 0.4900M solution of methylamine (CH3NH2) with a 0.7800M solution of HNO3. The pK of methylamine is 3.36. Calculate the pH of the base solution after the chemist has added 17.7 mL of the HNO3 solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HNO3 solution added. Round your answer to 2 decimal places. pH = ☑ ? 18 Ararrow_forward
- The following is two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 223.4 148.7 353.7 278.2 334.6 268.7 305.6 234.4 340.0 262.7 304.3 283.2 244.7 143.6 QUESTION: For both groups of data calculate the answers attached in the image.arrow_forwardThe following is a two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 340.0mmol/L 262.7mmol/L QUESTION: For both groups (Regular & Salt Reduced tomato sauce) of data provide answers to the following calculations below: 1. Standard Deviation (Sx) 2. T Values (t0.05,4) 3. 95% Confidence Interval (mmol/L) 4. [Na+] (mg/100 mL) 5. 95% Confidence Interval (mg/100 mL)arrow_forwardIf we have leucine (2-amino-4-methylpentanoic acid), alanine (2-aminopropanoic acid) and phenylalanine (2-amino-3-phenylpropanoic acid), indicate the tripeptides that can be formed (use the abbreviated symbols Leu., Ala and Phe).arrow_forward
- Briefly state why trifluoroacetic acid is more acidic than acetic acid.arrow_forwardExplain why acid chlorides are more reactive than amides in reactions with nucleophiles.arrow_forwardCalculating the pH of a weak base titrated with a strong acid An analytical chemist is titrating 101.7 mL of a 0.3500M solution of piperidine (C5H10NH) with a 0.05700M solution of HClO4. The pK of piperidine is 2.89. Calculate the pH of the base solution after the chemist has added 682.9 mL of the HClO solution to it. 4 Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HClO solution added. 4 Round your answer to 2 decimal places. pH = .11 00. 18 Ararrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY