
a)
Interpretation:
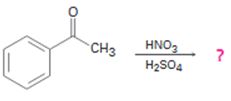
The product of the reaction in which actophenone reacts with HNO3 and H2SO4 is to be predicted. The observed regiochemistry is to be explained by drawing the resonance structures of the intermediates.
Concept introduction:
The carbonyl group is an electron withdrawing group. Hence in
To predict:
The product of the reaction in which actophenone reacts with HNO3 and H2SO4.
To explain:
The observed regiochemistry by drawing the resonance structures of the intermediates.
Answer to Problem 77AP
Actophenone reacts with HNO3 and H2SO4 to produce m-nitroacetophenone.
Explanation of Solution
The electrophile, NO2+, attacks the aromatic ring in the first step to produce a resonance stabilized carbocation (sigma complex). In the second step the carbocation deprotonates to yield the product. For the meta attack the intermediate is more stabilized as shown.
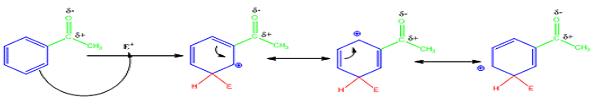
For ortho and para attack one of the resonance forms will be unstable as there will be a positive charge on the carbon to which the carbonyl group is attached.
Actophenone reacts with HNO3 and H2SO4 to produce m-nitroacetophenone.
b)
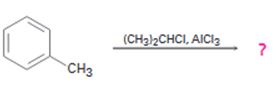
Interpretation:
The product of the reaction in which toluene reacts with isopropyl chloride in the presence of AlCl3 is to be predicted. The observed regiochemistry is to be explained by drawing the resonance structures of the intermediates.
Concept introduction:
Alkyl groups are an electron releasing groups. Hence during aromatic electrophilic substitution reactions the alkyl groups will activate the ring and orient the electrophile to the ortho and para positions.
To predict:
The product of the reaction in which toluene reacts with isopropyl chloride in the presence of AlCl3.
To explain:
The observed regiochemistry by drawing the resonance structures of the intermediates.
Answer to Problem 77AP
When toluene reacts with isopropyl chloride in the presence of AlCl3 the products obtained are o- and p-isopropylbenzenes. The p-isomer will predominate.
Explanation of Solution
The electrophile, (CH3)2CH+, attacks the aromatic ring in the first step to produce a resonance stabilized carbocation (sigma complex). In the second step the carbocation deprotonates to yield the product. For the ortho and para attacks, one of the the resonance structures has a positive charge on a carbon adjacent to the methyl group. This form is stabilized by electron release from the methyl group. Such stabilization is not possible for the meta attack. Hence a mixture of ortho-para products is produced in which the p-isomer predominates.
For ortho attack:
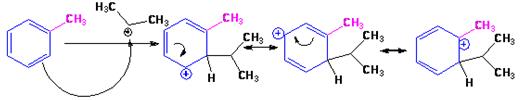
For para attack:
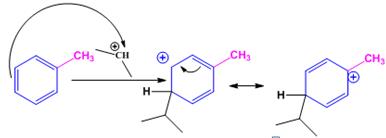
When toluene reacts with isopropyl chloride in the presence of AlCl3 the products obtained are o- and p-isopropylbenzenes. The p-isomer will predominate.
c)
Interpretation:
The product of the reaction in which benzonitrile reacts with chlorine in the presence of FeCl3 is to be predicted. The observed regiochemistry is to be explained by drawing the resonance structures of the intermediates.
Concept introduction:
The nitrile group is an electron withdrawing group. Hence in aromatic electrophilic substitution reactions the nitrile group will deactivate the ring and orient the electrophile to the meta position.
To predict:
The product of the reaction in which benzonitrile reacts with chlorine in the presence of FeCl3.
To explain:
The observed regiochemistry by drawing the resonance structures of the intermediates.
Answer to Problem 77AP
When benzonitrile reacts with chlorine in the presence of FeCl3 meta chlorobenzonitrile will be produced.
Explanation of Solution
The electrophile, Cl+, attacks the aromatic ring in the first step to produce a resonance stabilized carbocation (sigma complex). In the second step the carbocation deprotonates to yield the product. For the meta attack the intermediate is more stabilized as shown.
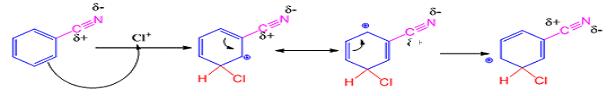
For ortho and para attack one of the resonance forms will be unstable as there will be a positive charge on the carbon to which the nitrile group is attached.
When benzonitrile reacts with chlorine in the presence of FeCl3 meta chlorobenzonitrile will be produced.
d)
Interpretation:
The product of the reaction in which methoxy benzene reacts with iodine in the presence of CuCl2 is to be predicted. The observed regiochemistry is to be explained by drawing the resonance structures of the intermediates.
Concept introduction:
Methoxy group is an electron releasing group. Hence during aromatic electrophilic substitution reactions the methoxy group will activate the ring and orient the electrophile to the ortho and para positions.
To predict:
The product of the reaction in which benzene reacts with iodine in the presence of CuCl2.
To explain:
The observed regiochemistry by drawing the resonance structures of the intermediates.
Answer to Problem 77AP
When methoxybenzene reacts with iodine in the presence of CuCl2, the products obtained are o- and p-iodobenzenes.
Explanation of Solution
Iodine itself is unreactive toward aromatic rings. CuCl2 accelerate the iodination reaction by oxidizing iodine to a more powerful by electrophilic species that reacts like I+.
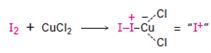
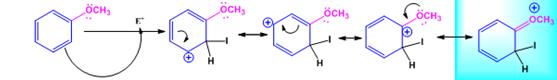
The electrophile, I+, attacks the aromatic ring in the first step to produce a resonance stabilized carbocation (sigma complex). In the second step the carbocation deprotonates to yield the product. For the ortho and para attacks, one of the the resonance structures has a positive charge on a carbon adjacent to the methoxy group. This form is stabilized by electron release from the methoxy group. Such stabilization is not possible for the meta attack. Hence a mixture of ortho-para products is produced.
Ortho attack:
Meta attack:
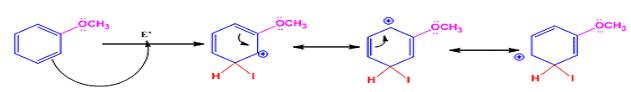
When methoxybenzene reacts with iodine in the presence of CuCl2, the products obtained are o- and p-iodobenzenes.
Want to see more full solutions like this?
Chapter 16 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- 3. For the reactions below, draw the arrows corresponding to the transformations and draw in the boxes the reactants or products as indicated. Note: Part A should have arrows drawn going from the reactants to the middle structure and the arrows on the middle structure that would yield the final structure. For part B, you will need to draw in the reactant before being able to draw the arrows corresponding to product formation. A. B. Rearrangement ΘΗarrow_forward2. Draw the arrows required to make the following reactions occur. Please ensure your arrows point from exactly where you want to exactly where you want. If it is unclear from where arrows start or where they end, only partial credit will be given. Note: You may need to draw in lone pairs before drawing the arrows. A. B. H-Br 人 C Θ CI H Cl Θ + Br Oarrow_forward4. For the reactions below, draw the expected product. Be sure to indicate relevant stereochemistry or formal charges in the product structure. a) CI, H e b) H lux ligh Br 'Harrow_forward
- Arrange the solutions in order of increasing acidity. (Note that K (HF) = 6.8 x 10 and K (NH3) = 1.8 × 10-5) Rank solutions from least acidity to greatest acidity. To rank items as equivalent, overlap them. ▸ View Available Hint(s) Least acidity NH&F NaBr NaOH NH,Br NaCIO Reset Greatest acidityarrow_forward1. Consider the following molecular-level diagrams of a titration. O-HA molecule -Aion °° о ° (a) о (b) (c) (d) a. Which diagram best illustrates the microscopic representation for the EQUIVALENCE POINT in a titration of a weak acid (HA) with sodium. hydroxide? (e)arrow_forwardAnswers to the remaining 6 questions will be hand-drawn on paper and submitted as a single file upload below: Review of this week's reaction: H₂NCN (cyanamide) + CH3NHCH2COOH (sarcosine) + NaCl, NH4OH, H₂O ---> H₂NC(=NH)N(CH3)CH2COOH (creatine) Q7. Draw by hand the reaction of creatine synthesis listed above using line structures without showing the Cs and some of the Hs, but include the lone pairs of electrons wherever they apply. (4 pts) Q8. Considering the Zwitterion form of an amino acid, draw the Zwitterion form of Creatine. (2 pts) Q9. Explain with drawing why the C-N bond shown in creatine structure below can or cannot rotate. (3 pts) NH2(C=NH)-N(CH)CH2COOH This bond Q10. Draw two tautomers of creatine using line structures. (Note: this question is valid because problem Q9 is valid). (4 pts) Q11. Mechanism. After seeing and understanding the mechanism of creatine synthesis, students should be ready to understand the first half of one of the Grignard reactions presented in a past…arrow_forward
- Propose a synthesis pathway for the following transformations. b) c) d)arrow_forwardThe rate coefficient of the gas-phase reaction 2 NO2 + O3 → N2O5 + O2 is 2.0x104 mol–1 dm3 s–1 at 300 K. Indicate whether the order of the reaction is 0, 1, or 2.arrow_forward8. Draw all the resonance forms for each of the following molecules or ions, and indicate the major contributor in each case, or if they are equivalent. (4.5 pts) (a) PH2 سمةarrow_forward
- 3. Assign absolute configuration (Rors) to each chirality center. a. H Nitz C. он b. 0 H-C. C H 7 C. ་-4 917-417 refs H 1つ ८ ડુ d. Но f. -2- 01 Ho -OH 2HNarrow_forwardHow many signals do you expect in the H NMR spectrum for this molecule? Br Br Write the answer below. Also, in each of the drawing areas below is a copy of the molecule, with Hs shown. In each copy, one of the H atoms is colored red. Highlight in red all other H atoms that would contribute to the same signal as the H already highlighted red. Note for advanced students: In this question, any multiplet is counted as one signal. Number of signals in the 'H NMR spectrum. For the molecule in the top drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box at right. No additional Hs to color in top molecule For the molecule in the bottom drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box at right. No additional Hs to color in bottom moleculearrow_forwardIn the drawing area below, draw the major products of this organic reaction: 1. NaOH ? 2. CH3Br If there are no major products, because nothing much will happen to the reactant under these reaction conditions, check the box under the drawing area instead. No reaction. Click and drag to start drawing a structure. ☐ : A คarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


