
(a)
Interpretation:
The spin and number of equivalent nuclei required to produce the given ESR spectrum are to be stated.
Concept introduction:
Electron spin resonance is the method to study the materials with an unpaired electron. An unpaired electron absorbs the microwave radiation under the strong magnetic field. The difference between two energy levels is calculated by the formula shown below.
Answer to Problem 16.22E
The spin and number of equivalent nuclei required to produce the given ESR spectrum are
Explanation of Solution
The give ESR spectrum is shown below.
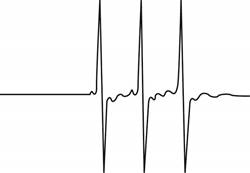
Figure 1
The number of spins in the ESR spectrum is shown below.
Where,
•
•
The total number of signal in the given ESR spectrum is
Three different peaks with same intensity indicate that
Substitute the values of
The above equation is further simplified for the value of
Therefore, the spin and number of equivalent nuclei required to produce the given ESR spectrum are
The spin and number of equivalent nuclei required to produce the given ESR spectrum are
(b)
Interpretation:
The spin and number of equivalent nuclei required to produce the given ESR spectrum are to be stated.
Concept introduction:
Electron spin resonance is the method to study the materials with an unpaired electron. An unpaired electron absorbs the microwave radiation under the strong magnetic field. The difference between two energy levels is calculated by the formula shown below.
Answer to Problem 16.22E
The spin and number of equivalent nuclei required to produce the given ESR spectrum are
Explanation of Solution
The give ESR spectrum is shown below.
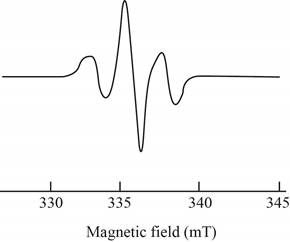
Figure 2
The number of spins in the ESR spectrum is shown below.
Where,
•
•
The total number of signal in the given ESR spectrum is
Three peaks with different intensities indicate that two equivalent nuclei are present.
Substitute the values of
The above equation is further simplified for the value of
Therefore, spin and number of equivalent nuclei required to produce the given ESR spectrum are
The spin and number of equivalent nuclei required to produce the given ESR spectrum are
(c)
Interpretation:
The spin and number of equivalent nuclei required to produce the given ESR spectrum are to be stated.
Concept introduction:
Electron spin resonance is the method to study the materials with an unpaired electron. An unpaired electron absorbs the microwave radiation under the strong magnetic field. The difference between two energy levels is calculated by the formula shown below.
Answer to Problem 16.22E
The spin and number of equivalent nuclei required to produce the given ESR spectrum are
Explanation of Solution
The give ESR spectrum is shown below.
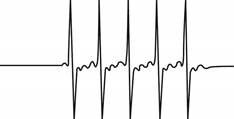
Figure 3
The number of spins in the ESR spectrum is shown below.
Where,
•
•
The total number of signal in the given ESR spectrum is
Five different peaks with same intensity represents that
Substitute the values of
The above equation is further simplified for the value of
The spin and number of equivalent nuclei required to produce the given ESR spectrum are
The spin and number of equivalent nuclei required to produce the given ESR spectrum are
(d)
Interpretation:
The spin and number of equivalent nuclei required to produce the given ESR spectrum are to be stated.
Concept introduction:
Electron spin resonance is the method to study the materials with an unpaired electron. An unpaired electron absorbs the microwave radiation under the strong magnetic field. The difference between two energy levels is calculated by the formula shown below.
Answer to Problem 16.22E
The spin and number of equivalent nuclei required to produce the given ESR spectrum are
Explanation of Solution
The give ESR spectrum is shown below.
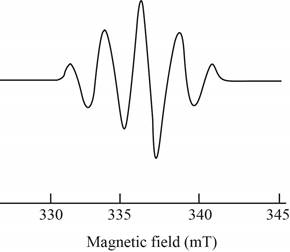
Figure 4
The number of spins in the ESR spectrum is shown below.
Where,
•
•
The total number of signal in the given ESR spectrum is
Five different peaks with different intensities indicate that four equivalent nuclei are present.
Substitute the values of
The above equation is further simplified for the value of
Therefore, the spin and number of equivalent nuclei required to produce the given ESR spectrum are
The spin and number of equivalent nuclei required to produce the given ESR spectrum are
Want to see more full solutions like this?
Chapter 16 Solutions
Physical Chemistry
- A student proposes the transformation below in one step of an organic synthesis. There may be one or more reactants missing from the left-hand side, but there are no products missing from the right-hand side. There may also be catalysts, small inorganic reagents, and other important reaction conditions missing from the arrow. • Is the student's transformation possible? If not, check the box under the drawing area. . If the student's transformation is possible, then complete the reaction by adding any missing reactants to the left-hand side, and adding required catalysts, inorganic reagents, or other important reaction conditions above and below the arrow. • You do not need to balance the reaction, but be sure every important organic reactant or product is shown. + T X O O лет-ле HO OH HO OH This transformation can't be done in one step.arrow_forwardDetermine the structures of the missing organic molecules in the following reaction: X+H₂O H* H+ Y OH OH Note: Molecules that share the same letter have the exact same structure. In the drawing area below, draw the skeletal ("line") structures of the missing organic molecules X and Y. You may draw the structures in any arrangement that you like, so long as they aren't touching. Click and drag to start drawing a structure. X Sarrow_forwardPredict the major products of this organic reaction. If there aren't any products, because nothing will happen, check the box under the drawing area instead. No reaction. HO. O :☐ + G Na O.H Click and drag to start drawing a structure. XS xs H₂Oarrow_forward
- What are the angles a and b in the actual molecule of which this is a Lewis structure? H H C H- a -H b H Note for advanced students: give the ideal angles, and don't worry about small differences from the ideal groups may have slightly different sizes. a = b = 0 °arrow_forwardWhat are the angles a and b in the actual molecule of which this is a Lewis structure? :0: HCOH a Note for advanced students: give the ideal angles, and don't worry about small differences from the ideal that might be caused by the fact that different electron groups may have slightly different sizes. a = 0 b=0° Sarrow_forwardDetermine the structures of the missing organic molecules in the following reaction: + H₂O +H OH O OH +H OH X Note: Molecules that share the same letter have the exact same structure. In the drawing area below, draw the skeletal ("line") structure of the missing organic molecule X. Click and drag to start drawing a structure.arrow_forward
- Identify the missing organic reactant in the following reaction: x + x O OH H* + ☑- X H+ O O Х Note: This chemical equation only focuses on the important organic molecules in the reaction. Additional inorganic or small-molecule reactants or products (like H₂O) are not shown. In the drawing area below, draw the skeletal ("line") structure of the missing organic reactant X. Click and drag to start drawing a structure. Carrow_forwardCH3O OH OH O hemiacetal O acetal O neither O 0 O hemiacetal acetal neither OH hemiacetal O acetal O neither CH2 O-CH2-CH3 CH3-C-OH O hemiacetal O acetal CH3-CH2-CH2-0-c-O-CH2-CH2-CH3 O neither HO-CH2 ? 000 Ar Barrow_forwardWhat would be the best choices for the missing reagents 1 and 3 in this synthesis? 1. PPh3 2 2. n-BuLi 3 Draw the missing reagents in the drawing area below. You can draw them in any arrangement you like. • Do not draw the missing reagent 2. If you draw 1 correctly, we'll know what it is. • Note: if one of your reagents needs to contain a halogen, use bromine. Explanation Check Click and drag to start drawing a structure.arrow_forward
- Predict the products of this organic reaction: NaBH3CN + NH2 ? H+ Click and drag to start drawing a structure. ×arrow_forwardPredict the organic products that form in the reaction below: + OH +H H+ ➤ ☑ X - Y Note: You may assume you have an excess of either reactant if the reaction requires more than one of those molecules to form the products. In the drawing area below, draw the skeletal ("line") structures of the missing organic products X and Y. You may draw the structures in any arrangement that you like, so long as they aren't touching. Click and drag to start drawing a structure. Garrow_forwardPredict the organic products that form in the reaction below: OH H+ H+ + ☑ Y Note: You may assume you have an excess of either reactant if the reaction requires more than one of those molecules to form the products. In the drawing area below, draw the skeletal ("line") structures of the missing organic products X and Y. You may draw the structures in any arrangement that you like, so long as they aren't touching. Click and drag to start drawing a structure. ✓ marrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning




