
Thermodynamics: An Engineering Approach
9th Edition
ISBN: 9781260048766
Author: CENGEL
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 15.7, Problem 67P
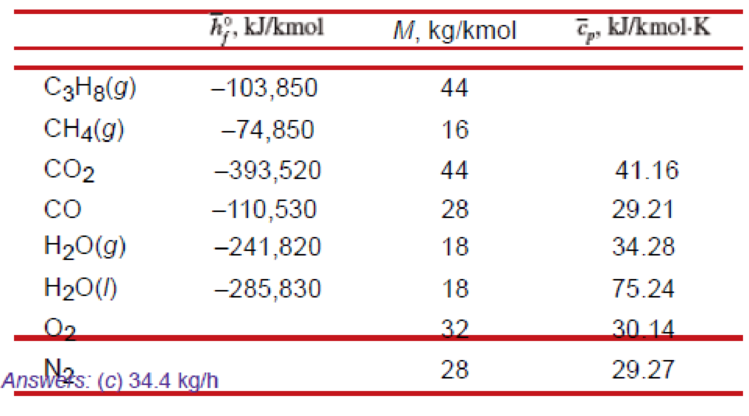
A gaseous fuel mixture that is 40 percent propane (C3H8) and 60 percent methane (CH4) by volume is mixed with the theoretical amount of dry air and burned in a steady-flow, constant-pressure process at 100 kPa. Both the fuel and air enter the combustion chamber at 298 K and undergo a complete combustion process. The products leave the combustion chamber at 398 K. Determine
- (a) the balanced combustion equation,
- (b) the amount of water vapor condensed from the products, and
- (c) the required airflow rate, in kg/h, when the combustion process produces a heat transfer output of 97,000 kJ/h.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A gaseous fuel with 80% butane, 15% nitrogen and 5% oxygen (on a mole basis) is burned to completion with 120 percent theoretical air that enters the combustion chamber at 30°C and 100 kPa. Determine the volume flow rate of air required to burn fuel at a rate of 2 kg/min.
Methanol (CH3OH) is burned with 50% excess air. Write the balanced reaction equations
for complete and actual combustion, and then determine (a) the actual air-to-fuel ratio,
and (b) the enthalpy of the complete combustion of Methanol at 25°C and 1 atm
assuming H₂O in the products is in the liquid form.
N₁
hc=Hp-HR=Nphp-ENRIR
TABLE 32
Standard enthalpies of formation
Species
State!
0₁
Gas
Gav
Ga
Gas
Gas
Gas
H₁
C.
Co,
но
1,0
CO
Useful relations:
Mair
123 ful
C,H,
CH,OH
CH,OH
Liqind
Gas
Gas
Gas
Gas
Liquid
Gas
Liquid
AF
C₂B₁
сн..
At 298.15 K (25 C) and 1 aus
Ah,, MJ/kmol
0
0
0
0
-393.52
241.83
285.84
110.54
-74.87
-101.85
-201.17
238.58
208.45
249.35
Liquid octane is burned completely with 75% excess air. Determine the air-fuel ratio for this combustion process.
Chapter 15 Solutions
Thermodynamics: An Engineering Approach
Ch. 15.7 - What are the approximate chemical compositions of...Ch. 15.7 - How does the presence of N2 in air affect the...Ch. 15.7 - Prob. 3PCh. 15.7 - Prob. 4PCh. 15.7 - Is the airfuel ratio expressed on a mole basis...Ch. 15.7 - How does the presence of moisture in air affect...Ch. 15.7 - Prob. 7PCh. 15.7 - Prob. 8PCh. 15.7 - Prob. 9PCh. 15.7 - Are complete combustion and theoretical combustion...
Ch. 15.7 - What does 100 percent theoretical air represent?Ch. 15.7 - Consider a fuel that is burned with (a) 130...Ch. 15.7 - What are the causes of incomplete combustion?Ch. 15.7 - Which is more likely to be found in the products...Ch. 15.7 - Methane (CH4) is burned with the stoichiometric...Ch. 15.7 - Prob. 16PCh. 15.7 - n-Butane fuel (C4H10) is burned with the...Ch. 15.7 - Prob. 18PCh. 15.7 - Propane (C3H8) is burned with 75 percent excess...Ch. 15.7 - Propane fuel (C3H8) is burned with 30 percent...Ch. 15.7 - In a combustion chamber, ethane (C2H6) is burned...Ch. 15.7 - Prob. 22PCh. 15.7 - Prob. 23PCh. 15.7 - Ethane (C2H6) is burned with 20 percent excess air...Ch. 15.7 - Octane (C8H18) is burned with 250 percent...Ch. 15.7 - Prob. 26PCh. 15.7 - A fuel mixture of 60 percent by mass methane (CH4)...Ch. 15.7 - Prob. 28PCh. 15.7 - A certain natural gas has the following volumetric...Ch. 15.7 - Prob. 30PCh. 15.7 - A gaseous fuel with a volumetric analysis of 45...Ch. 15.7 - Prob. 33PCh. 15.7 - The fuel mixer in a natural gas burner mixes...Ch. 15.7 - Prob. 35PCh. 15.7 - Prob. 36PCh. 15.7 - Determine the fuelair ratio when coal from...Ch. 15.7 - Prob. 38PCh. 15.7 - Prob. 39PCh. 15.7 - Prob. 40PCh. 15.7 - Prob. 41PCh. 15.7 - When are the enthalpy of formation and the...Ch. 15.7 - Prob. 43PCh. 15.7 - Prob. 44PCh. 15.7 - Prob. 45PCh. 15.7 - Prob. 46PCh. 15.7 - Prob. 48PCh. 15.7 - Repeat Prob. 1546 for liquid octane (C8H18).Ch. 15.7 - Ethane (C2H6) is burned at atmospheric pressure...Ch. 15.7 - Reconsider Prob. 1550. What minimum pressure of...Ch. 15.7 - Calculate the HHV and LHV of gaseous n-octane fuel...Ch. 15.7 - Prob. 53PCh. 15.7 - Consider a complete combustion process during...Ch. 15.7 - Prob. 56PCh. 15.7 - Prob. 57PCh. 15.7 - Prob. 58PCh. 15.7 - Propane fuel (C3H8) is burned with an airfuel...Ch. 15.7 - Prob. 60PCh. 15.7 - Prob. 61PCh. 15.7 - Prob. 62PCh. 15.7 - Octane gas (C8H18) at 25C is burned steadily with...Ch. 15.7 - Liquid ethyl alcohol [C2H5OH(l)] at 25C is burned...Ch. 15.7 - Prob. 66PCh. 15.7 - A gaseous fuel mixture that is 40 percent propane...Ch. 15.7 - A constant-volume tank contains a mixture of 120 g...Ch. 15.7 - Prob. 70PCh. 15.7 - Prob. 71PCh. 15.7 - Prob. 72PCh. 15.7 - A fuel is completely burned first with the...Ch. 15.7 - Prob. 74PCh. 15.7 - Prob. 75PCh. 15.7 - What is the adiabatic flame temperature of methane...Ch. 15.7 - Octane gas (C8H18) at 25C is burned steadily with...Ch. 15.7 - Acetylene gas (C2H2) at 25C is burned during a...Ch. 15.7 - Ethyl alcohol [C2H5OH(g)] is burned with 200...Ch. 15.7 - Prob. 81PCh. 15.7 - Prob. 82PCh. 15.7 - Reconsider Prob. 1582. The combustion products are...Ch. 15.7 - Express the increase of entropy principle for...Ch. 15.7 - Prob. 85PCh. 15.7 - What does the Gibbs function of formation gf of a...Ch. 15.7 - Liquid octane (C8H18) enters a steady-flow...Ch. 15.7 - Prob. 88PCh. 15.7 - Reconsider Prob. 1588. The automobile engine is to...Ch. 15.7 - Benzene gas (C6H6) at 1 atm and 77F is burned...Ch. 15.7 - Prob. 91PCh. 15.7 - n-Octane [C8H18(l)] is burned in the...Ch. 15.7 - A steady-flow combustion chamber is supplied with...Ch. 15.7 - Prob. 94RPCh. 15.7 - Prob. 95RPCh. 15.7 - Prob. 96RPCh. 15.7 - Prob. 97RPCh. 15.7 - Prob. 98RPCh. 15.7 - Prob. 99RPCh. 15.7 - n-Butane (C4H10) is burned with the stoichiometric...Ch. 15.7 - A gaseous fuel mixture of 60 percent propane...Ch. 15.7 - Calculate the higher and lower heating values of...Ch. 15.7 - Prob. 103RPCh. 15.7 - Methane gas (CH4) at 25C is burned steadily with...Ch. 15.7 - A 6-m3 rigid tank initially contains a mixture of...Ch. 15.7 - Propane gas (C3H8) enters a steady-flow combustion...Ch. 15.7 - Determine the highest possible temperature that...Ch. 15.7 - Liquid propane [C3H8(l)] enters a combustion...Ch. 15.7 - Prob. 109RPCh. 15.7 - Prob. 110RPCh. 15.7 - Prob. 111RPCh. 15.7 - A steam boiler heats liquid water at 200C to...Ch. 15.7 - Repeat Prob. 15112 using a coal from Utah that has...Ch. 15.7 - Liquid octane (C8H18) enters a steady-flow...Ch. 15.7 - Prob. 115RPCh. 15.7 - Consider the combustion of a mixture of an...Ch. 15.7 - Prob. 117RPCh. 15.7 - A fuel is burned steadily in a combustion chamber....Ch. 15.7 - A fuel is burned with 70 percent theoretical air....Ch. 15.7 - Prob. 126FEPCh. 15.7 - One kmol of methane (CH4) is burned with an...Ch. 15.7 - The higher heating value of a hydrocarbon fuel...Ch. 15.7 - Acetylene gas (C2H2) is burned completely during a...Ch. 15.7 - An equimolar mixture of carbon dioxide and water...Ch. 15.7 - A fuel is burned during a steady-flow combustion...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Similar questions
- Q1. Propane (C3H8) gas enters a steady-flow adiabatic combustion chamber at 25°℃ and 1 atm. It is burned with 300 percent excess air that also enters at 25°C and 1 atm. Assuming complete combustion, determine (a) the temperature of the products, (b) the entropy generation, and (c) the reversible work and exergy destruction. Assume that To = 298 K and the products leave the combustion chamber at 1 atm pressure.arrow_forwardLiquid propane (C3H8) enters a combustion chamber at 25°C at a rate of 0.07 kg/min where it is mixed and burned with 40 percent excess air that enters the combustion chamber at 7°C. An analysis of the combustion gases reveals that all the hydrogen in the fuel burns to H,0 but only 75 percent of the carbon burns to CO, with the remaining 25 percent forming CO. determine (a) the balanced equation for actual combustion process and (b) the mass flow rate of air.arrow_forwardinternal combustion enginearrow_forward
- Liquid propane (C 3 H 8 ) enters a combustion chamber at 25 °C at a rate of 0.05 kg/min where it is mixed and burned with theoretical air that enters the combustion chamber at 7 °C. an analysis of combustion gases reveals that all the hydrogen in the fuel burns to H 2 O but only but only 90% of carbon burn to CO 2 with the remaining 10% forming Co if the exit temperature of combustion gases is 1500 K (a) the mass flow rate of air and (b) the rate of heat transfer from the combustion chamberarrow_forwardAcetylene (C2H2) is burned with the stoichiometric amount of air during a combustion process. Assume complete combustion. Part A Determine the air-fuel ratio on a mass basis. Part B Determine the air-fuel ratio on a mole basis. Part C What-if scenario: What would the air to fuel ratio on a mass basis be if propene (C3H6) was burned instead of acetylene?arrow_forwardH.W.3.5 Propylene (C3H6) is burned with 50 percent excess air during a combustion process. Assuming complete combustion and a total pressure of 105 kPa, determine (a) the air-fuel ratio and (b) the temperature at which the water vapor in the products will start condensing (c) the product analysis based on volume and mass.arrow_forward
- C4Hs is burned in an engine with a fuel-rich air-fuel ratio. Dry analysis of the exhaust gives the following volume percents: CO2 = 14.95%, C4H8 0.75%, CO = 0%, H2 = 0%, O2 = 0%, with the rest being N2. Higher heating value of this fuel is QHHV = 46.9 MJ/kg. Write the balanced chemical equation for one mole of this fuel at these conditions. Calculate; a) Air-fuel ratio. b) Equivalence ratio. c) Lower heating value of fuel. [MJ/kg] d) Energy released when one kg of this fuel is burned in the engine with a combustion efficiency of 98%. [MJ]arrow_forwardOctane gas (C8H18) and atmospheric air are supplied to a combustion chamber at 25°C at the stoichiometric air/fuel ratio. The combustion takes place adiabatically under steady-flow condition. The change in velocity and the work transfer can be assumed negligible. The Enthalpy of Combustion of octane gas at 25°C is – 5,116,180 kJ/kmol (when H2O in the combustion products is in vapor phase). Determine with aid of the Table on Page 26, the final temperature of the combustion products.arrow_forwardThere is a well-insulated open combustion chamber that ethane (C2H6) goes into at 77°F and 1 atm. The theoretical amount of air also enters at 220°F and 1 atm. The combustion is complete. Determine: a) The temperature of the combustion products coming out (°F)arrow_forward
- Parrow_forwardThe reactant side has the following volumetric analysis for fuels burned simultaneously: CH₄= 68% and C₂H6= 32%. Assume a complete combustion with 15% excess air at 101.325 kPa, what is the partial pressure of water vapor in kPa.arrow_forwardEthane (C2H6) is burned with atmospheric air and the volumetric analysis of the dry products of combustion yields the following: 10% CO2, 1% CO, 3% 02 and 86% N2. Develop the combustion equation and determine (a) the percentage of the excess air (b) the air fuel ratio. (Solve for 100 kmol of dry products).arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning
Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning

Refrigeration and Air Conditioning Technology (Mi...
Mechanical Engineering
ISBN:9781305578296
Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill Johnson
Publisher:Cengage Learning
Extent of Reaction; Author: LearnChemE;https://www.youtube.com/watch?v=__stMf3OLP4;License: Standard Youtube License