
Concept explainers
PRACTICE PROBLEM
1-Chloro-3-methyl-2-butene undergoes hydrolysis in a mixture of water and dioxane at a rate that is more than a thousand times that of 1-chloro-2-butene.
(a) What factor accounts for the difference in reactivity?
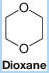
(b) What products would you expect to obtain? [Dioxane is a cyclic ether (below) that is miscible with water in all proportions and is a useful cosolvent for conducting reactions like these. Dioxane is carcinogenic (i.e., cancer causing), however, and like most ethers, it tends to form peroxides.]
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic Chemistry
Additional Science Textbook Solutions
College Physics: A Strategic Approach (3rd Edition)
Human Physiology: An Integrated Approach (8th Edition)
Organic Chemistry (8th Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Brock Biology of Microorganisms (15th Edition)
Anatomy & Physiology (6th Edition)
- Hi can you please help me solve this problem? thank youarrow_forwardAn electrode process takes place at a metal-solution interface. Indicate the current condition that must be met for Faradaic rectification to occur.arrow_forwardAt a metal-solution interface, an electron is exchanged, and the symmetry factor beta < 0.5 is found in the Butler-Volmer equation. What does this indicate?arrow_forward
