
(a)
Interpretation:
The CN stretching mode that absorbs the higher-frequency IR photons is to be indicated for given pair of compounds. The reason for it is to be explained.
Concept introduction:
We simplify the picture of molecular vibrations by considering the ball-and-spring model, which treats bonds as simple springs that connect atoms together. According to Hooke’s law, the spring vibrates at a particular frequency (
Answer to Problem 15.19P
The CN stretching mode that absorbs the higher-frequency IR photons due to strong and stiffer bond is indicated below,
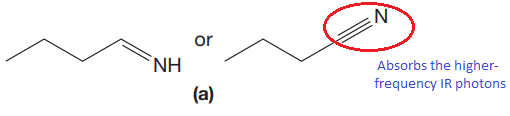
Explanation of Solution
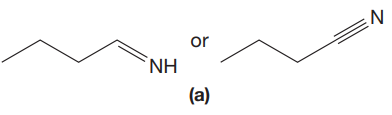
The given pair of compounds is,
The triple bonds tend to be stronger and stiffer than double bonds. A stronger and stiffer bond tends to lead to a higher vibrational frequency. In given pair of compounds, with the faster vibration, the
The CN stretching mode that absorbs the higher-frequency IR photons is shown below,

The CN stretching mode that absorbs the higher-frequency IR photons is indicated on the basis of the relationship between strength and stiffness of the bond and vibrational frequency.
(b)
Interpretation:
The CN stretching mode that absorbs the higher-frequency IR photons is to be indicated for given pair of compounds. The reason for it is to be explained.
Concept introduction:
We simplify the picture of molecular vibrations by considering the ball-and-spring model, which treats bonds as simple springs that connect atoms together. According to Hooke’s law, the spring vibrates at a particular frequency (
Answer to Problem 15.19P
The CN stretching mode that absorbs the higher-frequency IR photons due to strong and stiffer bond is indicated below,
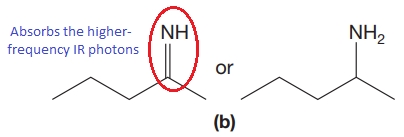
Explanation of Solution
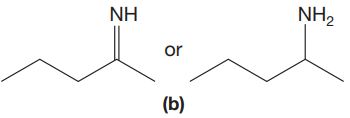
The given pair of compounds is,
The double bonds tend to be stronger and stiffer than single bonds. A stronger and stiffer bond tends to lead to a higher vibrational frequency. In given pair of compounds, with the faster vibration, the
The CN stretching mode that absorbs the higher-frequency IR photons is shown below,

The CN stretching mode that absorbs the higher-frequency IR photons is indicated on the basis of the relationship between strength and stiffness of the bond and vibrational frequency.
(c)
Interpretation:
The CN stretching mode that absorbs the higher-frequency IR photons is to be indicated for given pair of compounds. The reason for it is to be explained.
Concept introduction:
We simplify the picture of molecular vibrations by considering the ball-and-spring model, which treats bonds as simple springs that connect atoms together. According to Hooke’s law, the spring vibrates at a particular frequency (
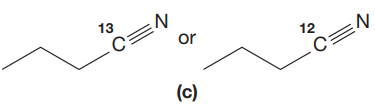
Answer to Problem 15.19P
The CN stretching mode that absorbs the higher-frequency IR photons due to lower mass is indicated below,
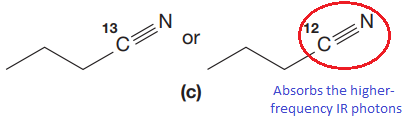
Explanation of Solution
The given pair of compounds is,
In both compounds there is
But
The CN stretching mode that absorbs the higher-frequency IR photons is shown below,

The vibrational mode that absorbs at a higher frequency in the IR region is determined on the basis of the relationship between mass and vibrational frequency.
Want to see more full solutions like this?
Chapter 15 Solutions
Organic Chemistry: Principles And Mechanisms: Study Guide/solutions Manual (second)
- Part VI. Ca H 10 O is the molecular formula of compound Tom and gives the in the table below. Give a possible structure for compound Tom. 13C Signals summarized C1 C2 C3 C4 C5 C6 C7 13C shift (ppm) 23.5 27.0 33.0 35.8 127 162 205 DEPT-90 + DEPT-135 + +arrow_forward2. Using the following data to calculate the value of AvapH o of water at 298K. AvapH o of water at 373K is 40.7 kJ/mol; molar heat capacity of liquid water at constant pressure is 75.2J mol-1 K-1 and molar heat capacity of water vapor at constant pressure is 33.6 J mol-1 K-1.arrow_forwardPart VII. Below are the 'HNMR 13 3 C-NMR, COSY 2D- NMR, and HSQC 20-NMR (Similar with HETCOR but axes are reversed) spectra of an organic compound with molecular formula C6H13 O. Assign chemical shift values to the H and c atoms of the compound. Find the structure. Show complete solutions. Predicted 1H NMR Spectrum ли 4.7 4.6 4.5 4.4 4.3 4.2 4.1 4.0 3.9 3.8 3.7 3.6 3.5 3.4 3.3 3.2 3.1 3.0 2.9 2.8 2.7 2.6 2.5 2.4 2.3 2.2 2.1 2.0 1.9 1.8 1.7 1.6 1.5 1.4 1.3 1.2 1.1 1.0 0.9 0.8 f1 (ppm)arrow_forward
- 3. Draw the expanded structural formula, the condensed structural formula, and the skeletal structural formula for 2-pentene. expanded structure: Condensed structure: Skeletal formula: 4. Draw the expanded structural formula, the condensed structural formula, and the skeletal structural formula for 2-methyl-3-heptene. expanded structure: Condensed structure: Skeletal formula: following structurearrow_forwardPart IV. Propose a plausible Structure w/ the following descriptions: a) A 5-carbon hydrocarbon w/ a single peak in its proton decoupled the DEPT-135 Spectrum shows a negative peak C-NMR spectrum where b) what cyclohexane dione isomer gives the largest no. Of 13C NMR signals? c) C5H120 (5-carbon alcohol) w/ most deshielded carbon absent in any of its DEPT Spectivaarrow_forward13C NMR is good for: a) determining the molecular weight of the compound b) identifying certain functional groups. c) determining the carbon skeleton, for example methyl vs ethyl vs propyl groups d) determining how many different kinds of carbon are in the moleculearrow_forward
- 6 D 2. (1 pt) Limonene can be isolated by performing steam distillation of orange peel. Could you have performed this experiment using hexane instead of water? Explain. 3. (2 pts) Using GCMS results, analyze and discuss the purity of the Limonene obtained from the steam distillation of orange peel.arrow_forwardPart III. Arrange the following carbons (in blue) in order of increasing chemical shift. HO B NH 2 A CIarrow_forward6. Choose the compound that will produce the spectrum below and assign the signals as carbonyl, aryl, or alkyl. 100 ō (ppm) 50 0 7. 200 150 Assign all of the protons on the spectrum below. 8. A B 4 E C 3 ō (ppm) 2 1 0 Choose the compound that will produce the spectrum below and assign the signals to the corresponding protons. OH 6 OH 3 2 1 0 4 ō (ppm)arrow_forward
- In the Thermo Fisher application note about wine analysis (Lesson 3), the following chromatogram was collected of nine components of wine. If peak 3 has a retention time of 3.15 minutes and a peak width of 0.070 minutes, and peak 4 has a retention time of 3.24 minutes and a peak width of 0.075 minutes, what is the resolution factor between the two peaks? [Hint: it will help to review Lesson 2 for this question.] MAU 300 200 T 34 5 100- 1 2 CO 6 7 8 9 0 2.4 2.6 2.8 3.0 3.2 3.4 3.6 3.8 4.0 4.2 4.4 4.6 4.8 5.0 5.2 Minutes 3.22 0.62 1.04 O 1.24arrow_forwardThe diagram shows two metals, A and B, which melt at 1000°C and 1400°C. State the weight percentage of the primary constituent (grains of C) that would be obtained by solidifying a 20% alloy of B. 1000°C a+L L+C 900°С 12 α a+C 45 1200 C L+y 140096 C+Y a+ß 800°C 700°C C+B 96 92 a+B 0 10 20 30 40 50 60 70 80 90 100 A % peso B Barrow_forward8. Choose the compound that will produce the spectrum below and assign the signals to the corresponding protons. 2 4 3 ō (ppm) OH 4 6 6 СОН 2 1 0arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,



