
(a)
Interpretation:
The IUPAC names of the acid chloride and carboxylate anion reactants and acid anhydride products have to be written.
Concept Introduction:
Naming of Acid Chloride:
Acid chlorides are derivatives of carboxylic acids with the formula,
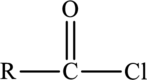
Acid chlorides are named by replacing the –ic acid ending of the common name with –yl chloride and –oic acid ending of the IUPAC name of the
Naming of Acid Anhydrides:
Acid anhydrides are compounds with the formula,
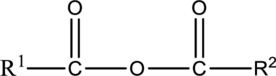
Acid anhydride is divided into symmetrical and unsymmetrical anhydrides.
Symmetrical anhydrides: contain same acyl groups. They are named by replacing acid ending of the carboxylic acid with the word anhydride.
Unsymmetrical anhydrides: contain different acyl groups. They are named by arranging the names of two-parent carboxylic acid followed by the word anhydride. The names of the carboxylic acid are arranged by size or in alphabetical order.
(b)
Interpretation:
The IUPAC names of the acid chloride and carboxylate anion reactants and acid anhydride products have to be written.
Concept Introduction:
Naming of Acid Chloride:
Acid chlorides are derivatives of carboxylic acids with the formula,
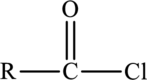
Acid chlorides are named by replacing the –ic acid ending of the common name with –yl chloride and –oic acid ending of the IUPAC name of the carboxylic acid with –oyl chloride.
Naming of Acid Anhydrides:
Acid anhydrides are compounds with the formula,
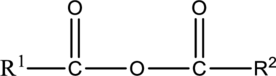
Acid anhydride is divided into symmetrical and unsymmetrical anhydrides.
Symmetrical anhydrides: contain same acyl groups. They are named by replacing acid ending of the carboxylic acid with the word anhydride.
Unsymmetrical anhydrides: contain different acyl groups. They are named by arranging the names of two-parent carboxylic acid followed by the word anhydride. The names of the carboxylic acid are arranged by size or in alphabetical order.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
General, Organic, and Biochemistry
- Using the general properties of equilibrium constants At a certain temperature, the equilibrium constant K for the following reaction is 0.84: H2(g) + 2(g) 2 HI(g) = Use this information to complete the following table. Suppose a 34. L reaction vessel is filled with 0.79 mol of HI. What can you say about the composition of the mixture in the vessel at equilibrium? There will be very little H2 and 12. ☐ x10 There will be very little HI. Neither of the above is true. What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. 2 HI(g) H₂(9)+12(9) K = What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. 2 H2(g)+212(9) 4 HI(g) K = ☐ ☑arrow_forwardPredicting the qualitative acid-base properties of salts Consider the following data on some weak acids and weak bases: base acid Κα Kb name formula name formula hydrocyanic acid - 10 HCN 4.9 × 10 pyridine C₂H₂N 1.7 × 10 9 acetic acid HCH3CO2 1.8 × 10 5 hydroxylamine HONH2 1.1 × 10¯ 8 Use this data to rank the following solutions in order of increasing pH. In other words, select a '1' next to the solution that will have the lowest pH, a '2' next to the solution that will have the next lowest pH, and so on. 0.1 M KCN solution pH choose one ✓ 0.1 M C5H5NHCI choose one ✓ 0.1 M NaCH3CO2 choose one ✓ 0.1 M HONH3Br ✓ choose one 1 (lowest) 2 3 4 (highest)arrow_forwardFor this question please solve the first question. Please explain your thought process, the steps you took, and how you would tackle a similar problem. Thank you for your help!arrow_forward
- Part C A solution that is 0.040 M in HCIO4 and 0.046 M in HCI Express your answer numerically to two decimal places. ΜΕ ΑΣΦ ? pH = Submit Request Answer Part D A solution that is 1.08% HCl by mass (with a density of 1.01 g/mL) Express your answer numerically to three decimal places. ΜΕ ΑΣΦ -> 0 ? pH =arrow_forwardPredict the equilibrium arrows for the following reaction:*see imagearrow_forwardProvide the missing information for each of the two following reacitons: *see imagearrow_forward
- Draw an example of the following functional groups: *see imagearrow_forwardAldehydes and Ketones: Show the reaction conditions, and molecules, that connect the reactant to the product. A protecting group will be needed. *see imagearrow_forwardAldehydes and Ketones: Show the reaction conditions, and molecules, that connect the reactant to the product. *see imagearrow_forward
- Provide the missing information for each of the four reactions: *see imagearrow_forward6. Chlorine dioxide (CIO) is used as a disinfectant in municipal water-treatment plants. It decomposes in a first-order reaction with a rate constant of 14 s. How long would it take for an initial concentration of 0.06 M to decrease to 0.02 M? [6 pts]arrow_forwardIf possible, replace an H atom on the a carbon of the molecule in the drawing area with a methyl group substituent, and replace an H atom on the ẞ carbon with a hydroxyl group substituent. If one of the substituents can't be added for any reason, just don't add it. If neither substituent can be added, check the box under the drawing area. en HO OHarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





